Renal sodium and water retention with resulting extracellular volume expansion and redistribution are hallmark features of heart failure syndromes. However, congestion assessment, monitoring, and treatment represent a real challenge in daily clinical practice. This document reviewed historical and contemporary evidence of available methods for determining volume status and discuss pharmacological aspects and pathophysiological principles that underlie diuretic use.
La retención renal de sodio y agua con la consiguiente expansión y redistribución del volumen de líquido extracelular constituye una de las principales características fisiopatológicas de la insuficiencia cardiaca. No obstante, la detección, monitorización y manejo de la congestión continúa representando un verdadero desafío para el clínico. En el presente documento se revisa literatura histórica y contemporánea acerca de los métodos disponibles para evaluar la congestión desde una perspectiva clínica e integradora, y se discuten aspectos farmacológicos y principios fisiopatológicos fundamentales para el uso óptimo de la terapia con diuréticos.
The relevance of this review is based on:
- -
Congestion is a key pathophysiological phenomenon largely responsible for the clinical manifestations and morbidity and mortality associated with heart failure.
- -
Limited diagnostic performance of classic symptoms and signs in the assessment of congestion.
- -
Poor understanding of the complex pathophysiology of congestion, a fact that explains the great variability in this regard.
- -
Largely empirical therapeutic approach. In this sense, and from a pathophysiological and clinical point of view, we propose a detailed and updated review of the different agents and therapeutic strategies used.
- -
The increasingly prevalent role of cardiorenal syndrome.
Heart failure (HF) is an increasing public health problem. The incidence, prevalence and, morbidity and mortality associated with this syndrome is high and constitutes the paradigm of the chronic patient who suffers from frequent decompensations.1 Most of these decompensations are attributable to congestion.2 In this scenario, diuretics are the mainstay of treatment.3 However, there is great uncertainty about how, how much and where to administer the diuretics.
The objective of this document is to inquire into clinical, pathophysiological and pharmacological aspects from an integrating perspective that contributes to a better understanding of congestion and the optimization of its treatment.
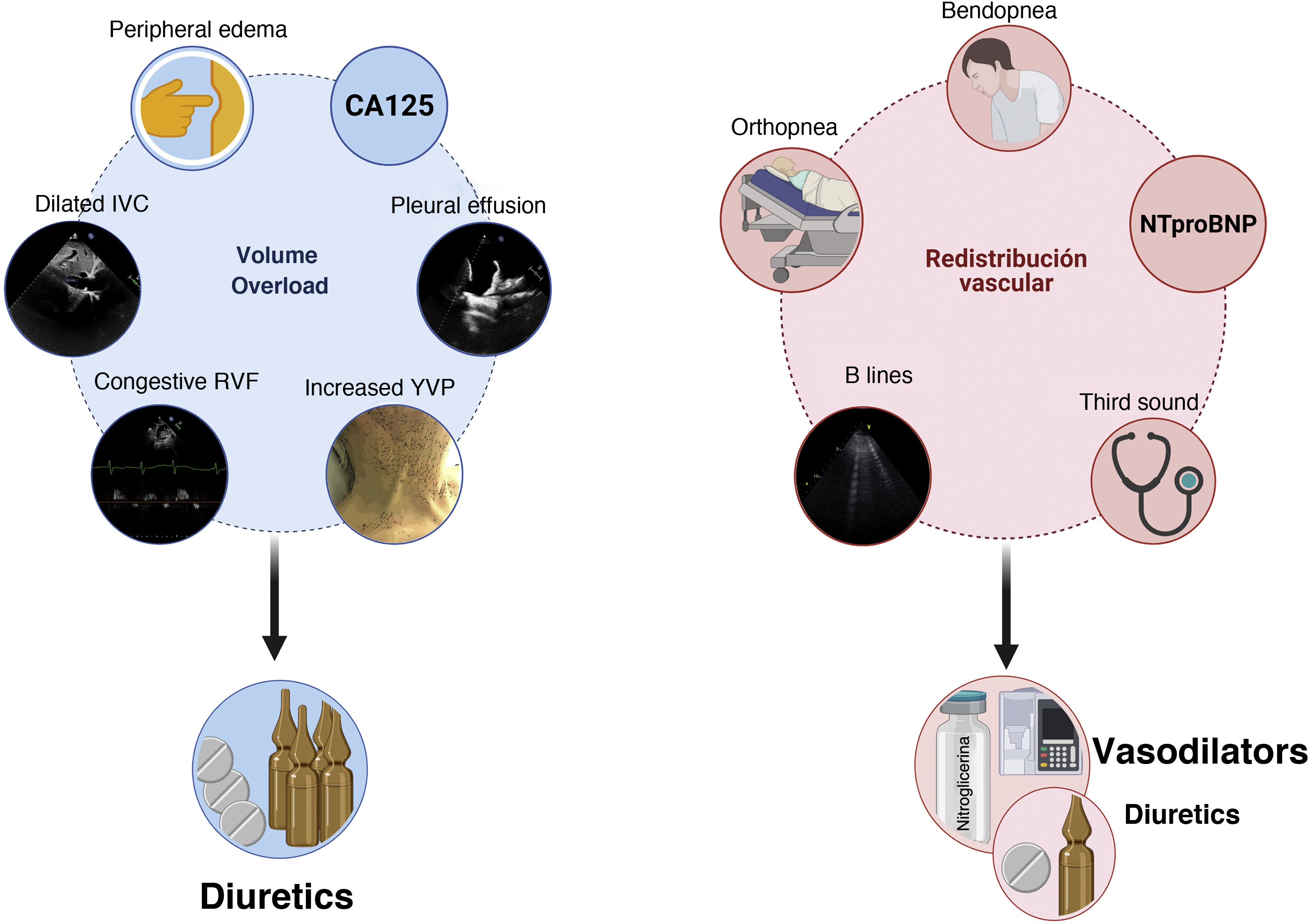
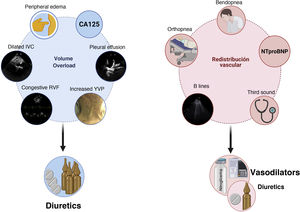
Diagnosis and characterization of congestion in HFIn HF, congestion is defined as the accumulation of fluid in the intravascular and extravacular compartment as a result of increased filling pressures. However, congestion is not a synonymous of volume overload.4 The concept of vascular redistribution suggests that increased venous tone (due to neurohormonal activation, myocardial ischemia, hypertensive episodes, medication changes, etc.) can precipitate a rapid redistribution of fluid from a peripheral venous reservoir (e.g., splanchnic bed) to the central cardiopulmonary circulation, increasing the intravascular hydrostatic pressure without increasing total blood volume.5 In this scenario, vasodilator therapy could be more appropriate than aggressive diuretic intervention (Fig. 1). In contrast, volume overload is a more latent phenomenon that results from the avidity of sodium and water in the renal tubule, and from the imbalance between the hydrostatic and oncotic pressures of the intravascular and interstitial compartment.6 Although most patients with decompensated HF present a combination of both, congestion and volume overload, identifying the predominant phenotype will determine the most appropriate therapeutic strategy (Fig. 1). It is noteworthy that the traditional symptoms and signs to assess congestion offer a limited diagnostic accuracy for the characterization and quantification of its severity.7,8
Integration of clinical methods, biomarkers and imaging techniques to distinguish between congestion due to volume overload vs. vascular redistribution.
CA125: carbohydrate antigen 125; RVF: renal venous flow; NTproBNP: N-terminal fragment of B-type natriuretic peptide; JVP: jugular venous pressure; IVC: inferior vena cava.
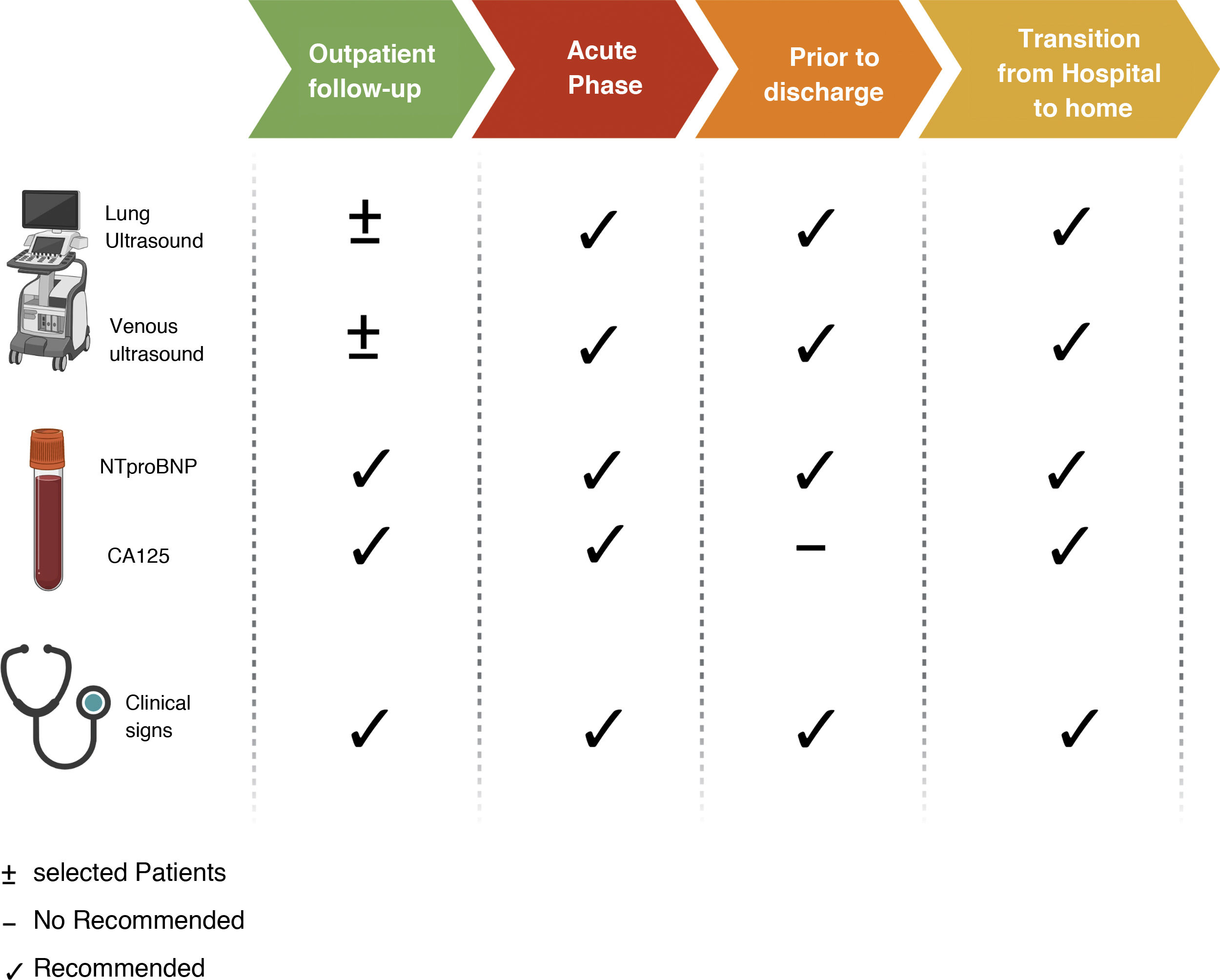
Right heart catheterization is the most specific method to assess the degree of intravascular congestion. However, outside the field of critical care or for specific purposes such as hemodynamic evaluation prior to implantation of ventricular assist devices or prior to heart transplantation, invasive pressure measurement is not routinely used. Similarly, despite the fact that remote monitoring of pulmonary arterial pressure through a wireless device implanted in the pulmonary artery has shown very promising clinical results in selected patients,10,11 the cost of these devices is the main limitation for its use. Therefore, it is necessary to integrate clinical parameters, biomarkers and imaging techniques (Table 1).
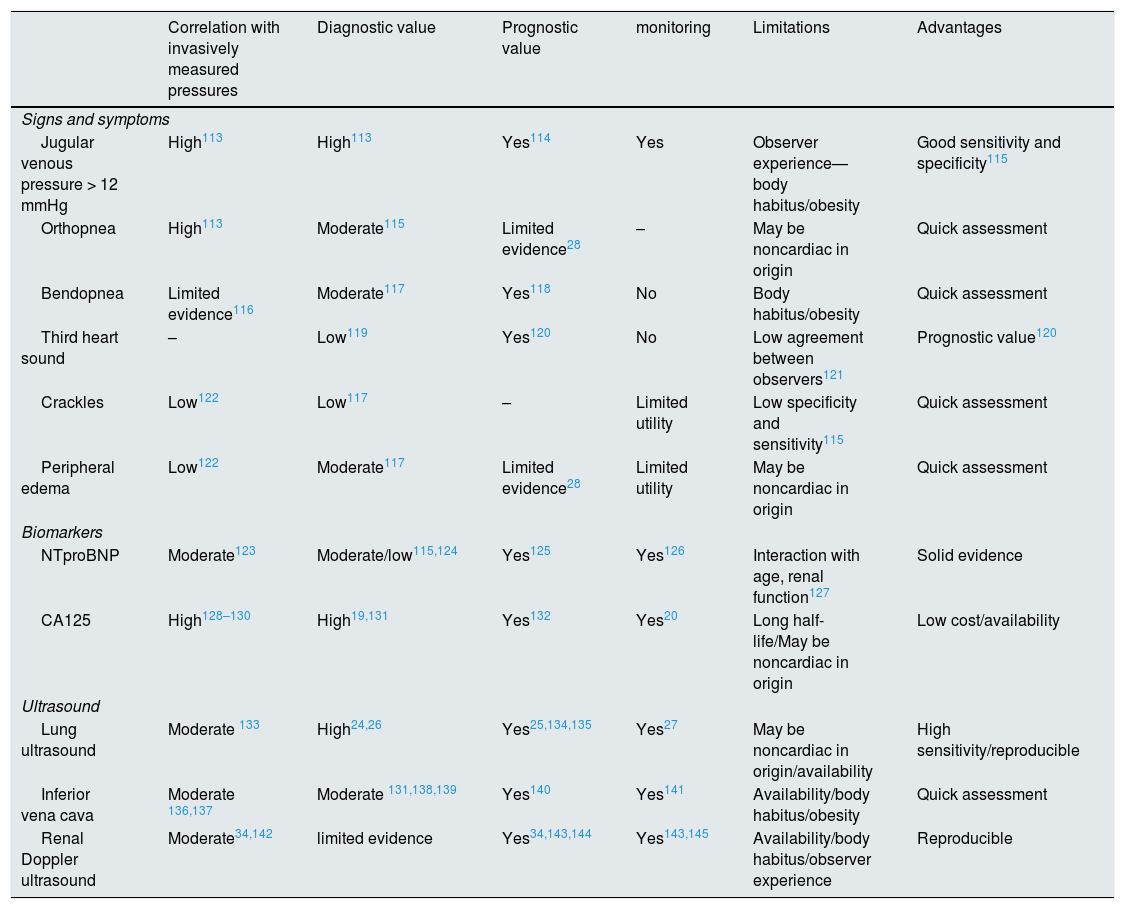
Clinical, ultrasound and biomarker methods to assess congestion in patients with heart failure: evidence and limitations.
| Correlation with invasively measured pressures | Diagnostic value | Prognostic value | monitoring | Limitations | Advantages | |
|---|---|---|---|---|---|---|
| Signs and symptoms | ||||||
| Jugular venous pressure > 12 mmHg | High113 | High113 | Yes114 | Yes | Observer experience—body habitus/obesity | Good sensitivity and specificity115 |
| Orthopnea | High113 | Moderate115 | Limited evidence28 | – | May be noncardiac in origin | Quick assessment |
| Bendopnea | Limited evidence116 | Moderate117 | Yes118 | No | Body habitus/obesity | Quick assessment |
| Third heart sound | – | Low119 | Yes120 | No | Low agreement between observers121 | Prognostic value120 |
| Crackles | Low122 | Low117 | – | Limited utility | Low specificity and sensitivity115 | Quick assessment |
| Peripheral edema | Low122 | Moderate117 | Limited evidence28 | Limited utility | May be noncardiac in origin | Quick assessment |
| Biomarkers | ||||||
| NTproBNP | Moderate123 | Moderate/low115,124 | Yes125 | Yes126 | Interaction with age, renal function127 | Solid evidence |
| CA125 | High128–130 | High19,131 | Yes132 | Yes20 | Long half-life/May be noncardiac in origin | Low cost/availability |
| Ultrasound | ||||||
| Lung ultrasound | Moderate 133 | High24,26 | Yes25,134,135 | Yes27 | May be noncardiac in origin/availability | High sensitivity/reproducible |
| Inferior vena cava | Moderate 136,137 | Moderate 131,138,139 | Yes140 | Yes141 | Availability/body habitus/obesity | Quick assessment |
| Renal Doppler ultrasound | Moderate34,142 | limited evidence | Yes34,143,144 | Yes143,145 | Availability/body habitus/observer experience | Reproducible |
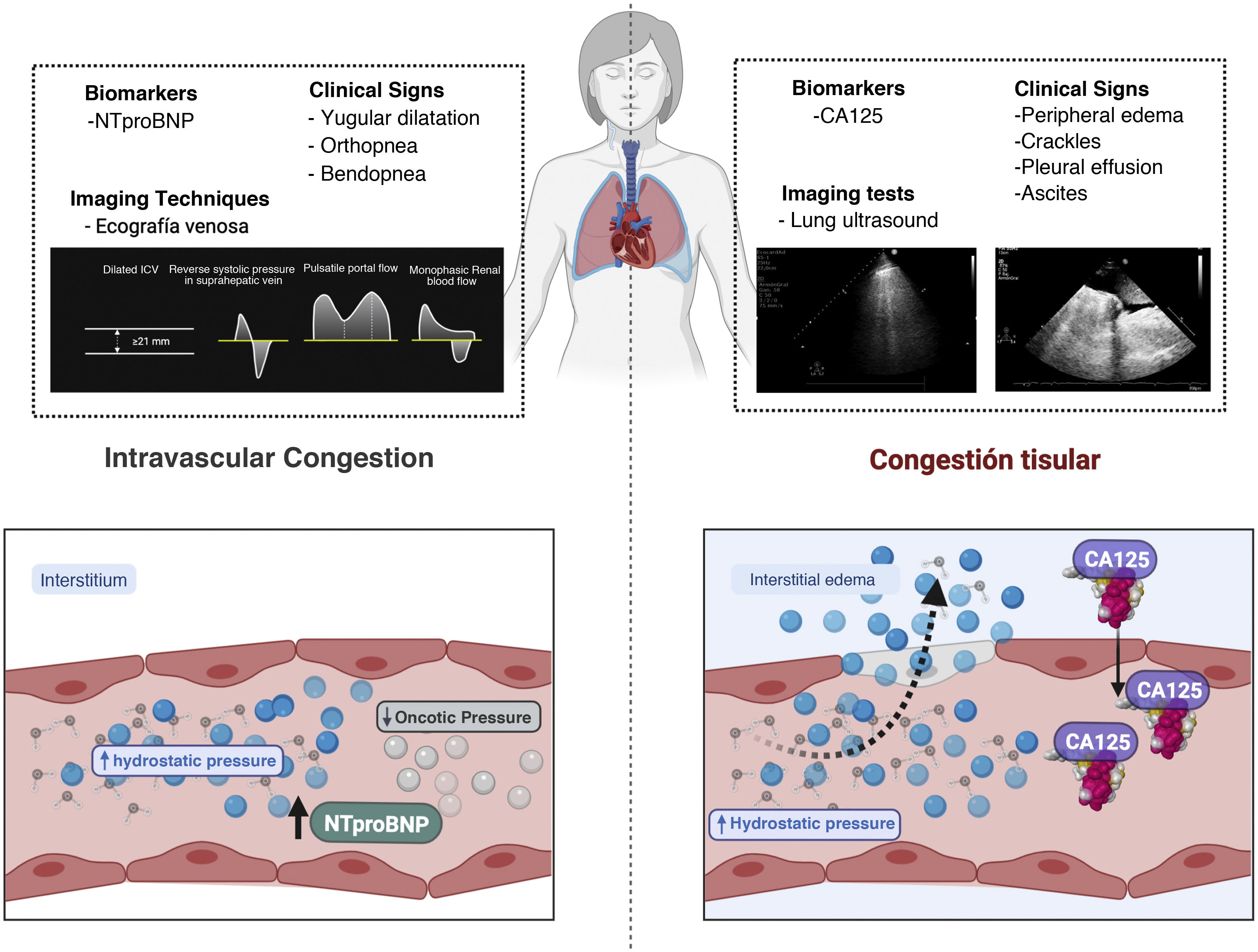
Jugular venous distention, orthopnea, and bendopnea are clinical variables associated with increased central venous pressure and provide acceptable diagnostic performance .12 Likewise, the presence of high levels of natriuretic peptides in an appropriate clinical context suggests an increase in cardiac filling pressures.13,14 Regarding imaging techniques, venous ultrasound has emerged as a useful and non-invasive tool. Beyond the measurement of the diameter of the inferior vena cava as an indirect parameter of central venous pressure, the analysis of Doppler venous waveforms in the portal vein, hepatic veins and intrarenal veins provides additional information on the distensibility of the venous system in response to venous congestion15 (Fig. 2).
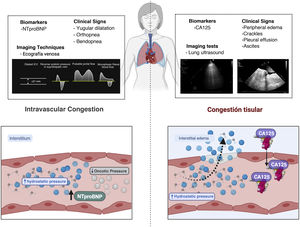
Multiparametric assessment of congestion. The figure shows the clinical signs, biomarkers and imaging techniques that have been established as markers of congestion. Some are more indicative of tissue congestion, while others are more indicative of intravascular congestion.
CA125: carbohydrate antigen; NTproBNP: N-terminal fragment of B-type natriuretic peptide.
Most symptoms and signs used to assess interstitial congestion have moderate specificity and low sensitivity for identifying tissue congestion. Therefore, it is essential to integrate clinical information with biomarkers and imaging techniques (Fig. 2).
Carbohydrate antigen 125 (CA125)Carbohydrate antigen 125 (CA125) is a glycoprotein synthesized by coelomic epithelial cells in places such as the pleura, pericardium, and peritoneum.16 Although it has traditionally been used for monitoring and risk stratification in ovarian cancer, elevated plasma concentrations of CA125 have been identified in other entities related to hydropic states, such as HF.17 Although the pathophysiological mechanism linked to HF is not fully understood, one of the most accepted theories suggests that there is activation of mesothelial cells in response to increased hydrostatic pressure, mechanical stress, and inflammatory cytokines.18 Recent evidence has shown the association of CA125 with clinical parameters of systemic congestion, and a positive correlation with various surrogate biomarkers of inflammation and congestion.19 Two clinical trials in patients with acute congestive HF have evaluated a diuretic strategy guided by plasma concentrations of CA125 versus standard of care (guided by symptoms and signs) and the results are promising suggesting the usefulness of this biomarker to optimize the intensity of depleting treatment.20,21
Lung ultrasoundLung ultrasound has emerged as a very useful tool for the evaluation of pulmonary interstitial congestion. The amount of water in the lungs corresponds to the degree of echogenicity found on ultrasound.22 In the case of interstitial pulmonary edema, the ultrasound beam reflects off the edematous interlobar septa and produces reverberation artifacts called B lines.23 The number of B lines is indicative of the degree of pulmonary interstitial congestion. In patients with dyspnea, ≥3 B lines in at least two zones per hemithorax (of 6–8 zones evaluated in total) identifies patients with acute HF with greater sensitivity (94–97%) and specificity (96–97%) than physical examination and chest x-ray.24 Likewise, a higher number of B lines at discharge after hospitalization for acute HF or in outpatients with chronic HF identifies those with a higher risk of HF readmission and death. A recent clinical trial has also shown that pulmonary ultrasound-guided diuretic treatment can reduce the number of decompensations in patients with HF.
Additionally, taking into account the dynamic nature and prognostic relevance of residual congestion,28 integrating clinical parameters, biomarkers and imaging techniques (Appendix A Supplementary material 1) provide relevant information to decision making .29
Impact of congestion on glomerular filtrationThe increase in central venous pressure is transmitted to the renal venous system and, therefore, influences the glomerular filtration rate (GFR).30,31 Renal filtration pressure (from which glomerular capillary hydrostatic pressure is derived) depends on renal perfusion pressure (RPP) and renal blood flow (RBF). In turn, RPP depends on mean arterial pressure (MAP) and renal venous pressure (RVP), and RBF depends on renal arterial pressure (RAP), RVP, and intrarenal vascular resistance (RVR)32:
- □
RPP = MAP − RVP
- □
RBF = RAP − RVP/RVR
In patients with chronic HF, RBF and GFR remain almost constant within a relatively wide RPP range (80−180 mmHg). This is due to renal autoregulation and tubule-glomerular feedback mechanisms that modify pre- and postglomerular resistance in order to maintain filtration pressure.33 However, in the setting of acute HF, autoregulatory mechanisms are altered and filtration pressure depends largely on the balance between MAP and RVP.33 Therefore, the increase in RVP (intrarenal afterload) can significantly reduce RBF. Likewise, since the kidney is an encapsulated organ, the increase in RVP produces mechanical compression on the interstitium and intratubular compartment, which further reduces the glomerular transcapillary hydrostatic pressure gradient and, therefore, represents an additional mechanism through which renal venous hypertension can compromise GFR.34 Likewise, renal venous congestion produces inflammation, oxidative stress and renal ischemia, causing intrinsic tubular damage.34
Impact of congestion on sodium and water reabsorptionUnder physiological conditions, changes in glomerular filtration rate are balanced by equivalent changes in tubular reabsorption (glomerulotubular balance).35 Thus, depending on the filtration fraction (FF), changes in hydrostatic and oncotic pressure in the renal interstitium and peritubular capillaries will determine the reabsorption of Na+ and water in the proximal tubule. It is important to highlight that glomerulotubular balance is not influenced by neurohumoral activation, but rather by Starling's forces that operate locally in the microcirculation of the proximal nephron.36 Renal venous congestion causes an increase in hydrostatic pressure in the tubular lumen, in the interstitium, and at the level of the peritubular capillaries.37 However, the increase in hydrostatic pressure stimulates lymphatic drainage in the renal interstitium (resulting in a reduction in interstitial oncotic pressure) while the peritubular capillaries are virtually impermeable to plasma proteins (peritubular oncotic pressure remains high).38 Thus, the resulting colloid osmotic pressure gradient between the interstitium and peritubular capillary stimulates Na+ and water reabsorption directly. Likewise, the increase in sodium reabsorption at the proximal level reduces the flow of sodium and chloride towards the macula densa which, together with the reduction in RBF, further increases neurohormonal activation, generating a vicious cycle.
Congestion treatmentUnder physiological conditions, extracellular volume homeostasis remains constant as a result of the tight control of various counterregulatory mechanisms that determine the rate of reabsorption/excretion of sodium and water in the renal tubule. From a quantitative point of view, a normal glomerular filtration rate (125 mL/min/1.73 m2) supplies approximately 25,000 mmol Na+/day to the renal tubule. Even so, more than 99% of the filtered sodium is reabsorbed and only a small amount is finally eliminated in the urine (approximately 100 mmol/L/day). Thus, minimal changes in the ratio between filtrated Na+ and the fraction that is reabsorbed in the renal tubule can exert a profound influence on the net Na+ balance.
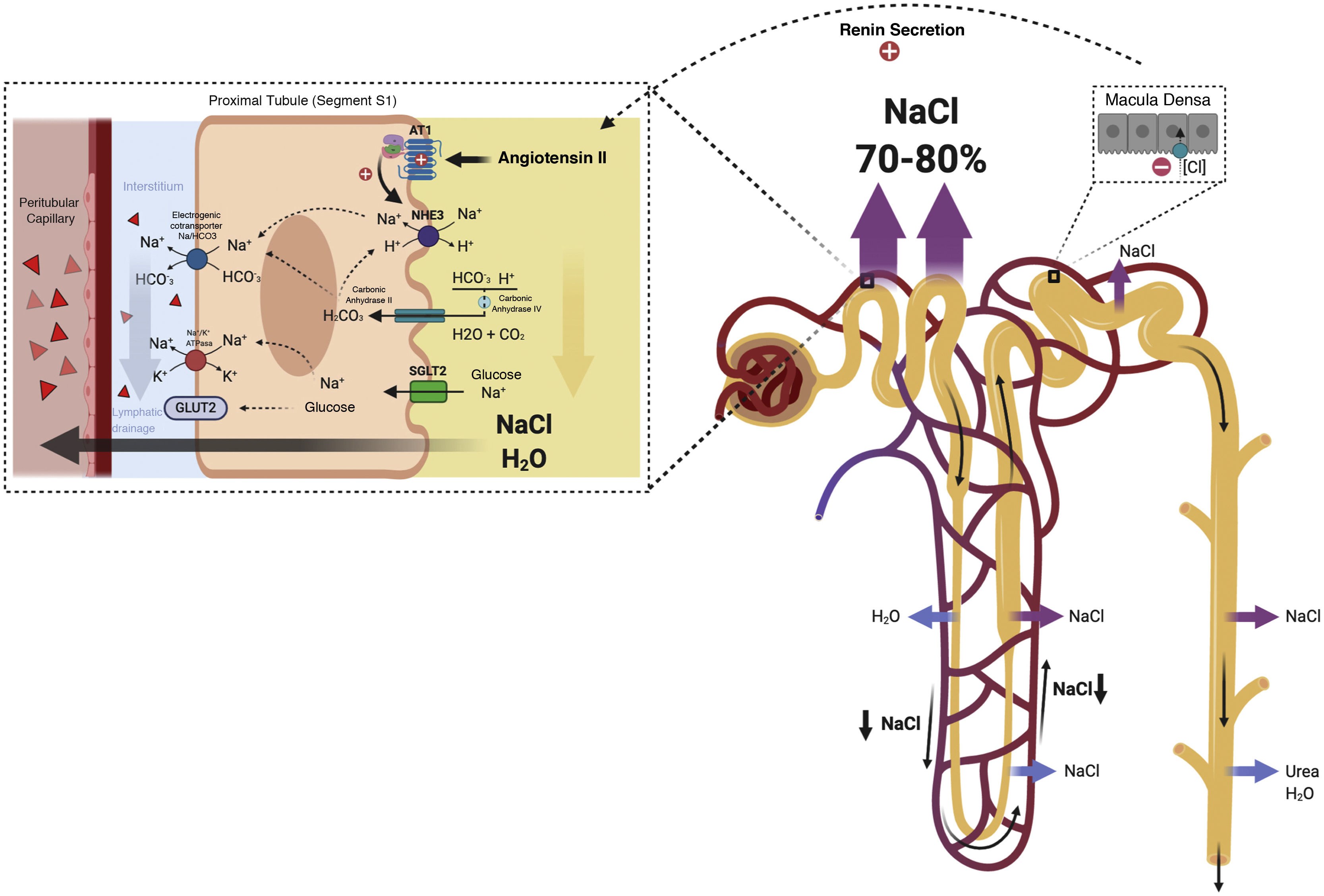
Proximal diureticsThe main function of the renal proximal tubule is the nearly isosmotic reabsorption of approximately 70% of the glomerular ultrafiltrate. This includes the reabsorption of 65–75% of the filtered Na+.39 Na+ reabsorption in the proximal tubule occurs through paracellular and transcellular mechanisms mediated fundamentally by glomerulotubular balance and by neurohumoral influence.35
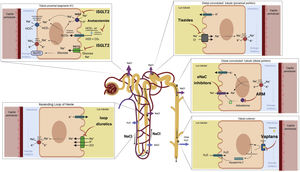
In patients with decompensated HF, neurohumoral activation exerts direct effects on epithelial transport in the proximal tubule by stimulating cotransporters involved in transcellular Na+ reabsorption (Na+/H+ exchanger isoform 3 [NHE3], electrogenic cotransporter Na+/HCO3− and Na–K–ATPase)40 (Fig. 3). Likewise, the reduction in the RBF produces an increase in the filtration fraction and, therefore, an increase in the proximal reabsorption of Na+ mediated by the glomerulotubular balance. All these mechanisms generate a vicious cycle that enhances the reabsorption of Na+ in the proximal tubule, favors the activation of the renin-angiotensin-aldosterone axis and contributes to the resistance of diuretics that act more distally in the nephron (Fig. 4).
Proximal tubule. Neurohormonal activation and intraglomerular and peritubular hemodynamic changes facilitate Na and water reabsorption in the proximal tubule. Additionally, increased lymphatic flow washes out interstitial proteins and decreases oncotic pressure in the renal interstitium, further promoting passive Na reabsorption.
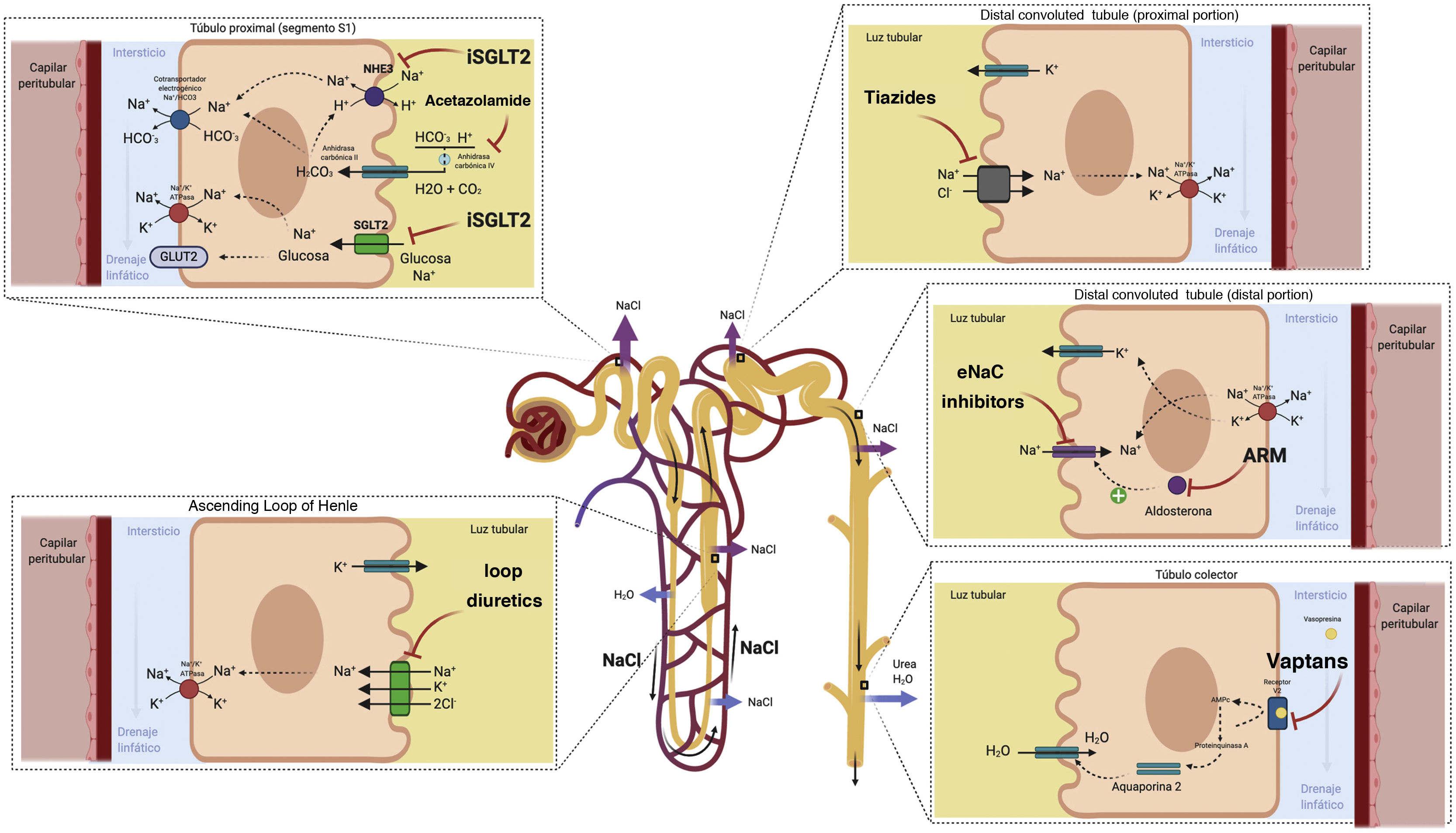
Acetazolamide acts by inhibiting carbonic anhydrase and thus blocks the reabsorption of bicarbonate and Na+ in the proximal tubule.41 Although the diuretic and natriuretic capacity of acetazolamide on its own is poor, it could play a role as an "enhancer" of diuretic efficacy if used in combination with diuretics that act more distally in the renal tubule by increasing distal delivery of Na+. This concept is supported by small observational studies42–44 and by a small randomized study that included 24 patients with refractory congestion in whom the administration of acetazolamide was associated with an improvement in the fractional excretion of Na+.45
Thus, until the evidence is stronger, acetazolamide is recommended as a second-line drug. Since it can cause metabolic acidosis, periodic evaluation of renal function, serum electrolytes, and blood pH is recommended.
Sodium-glucose cotransporter 2 inhibitorsSodium-glucose cotransporter 2 (SGLT2) inhibitors are hypoglycemic drugs that have been consistently shown to reduce HF hospitalizations in patients with type 2 diabetes mellitus,46 in stable patients with HF and depressed ejection fraction (diabetics and non-diabetics),47 and in diabetic patients with a recent episode of HF regardless of ejection fraction.48
The SGLT2 cotransporter is located in the S1 segment of the proximal convoluted tubule of the nephron and reabsorbs approximately 90% of filtered glucose. Tubular glucose reabsorption is coupled to Na+ reabsorption (one Na+ molecule for each glucose molecule) following an electrochemical gradient of higher concentration in the tubular lumen and lower concentration inside the tubular epithelial cell. In addition, the SGLT2 contransporter is located adjacent to the renal Na+/hydrogen exchanger (NHE3), which is largely responsible for Na+ reabsorption in the proximal tubule. SGLT2 inhibition appears to exert a cross-reaction with the NHE3 exchanger, enhancing natriuresis by a mechanism independent of glucose reabsorption inhibition.49 Although the natriuretic effect of SGLT2 inhibition appears to be weak in monotherapy, recent evidence suggests a synergistic effect when combined with loop diuretics by increasing Na+ delivery to the thick loop of Henle.50 Furthermore, the release of renin mediated by loop diuretics produces an upregulation of the SGLT2 cotransporter, enhancing Na + flow from the proximal tubule to more distal parts after its inhibition.49 Another interesting aspect derives from its potential capacity to produce a significant increase in the excretion of electrolyte-free water, mediated mainly by an osmotic effect.51–53 This effect could favor the decongestion of the interstitium without associating relevant changes in intravascular volume.
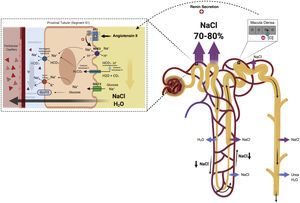
Loop diureticsAlthough only one third of the volume filtered by the glomerulus reaches the loop of Henle, this segment is especially important for the maintenance of homeostasis of extracellular volume and the concentration of urine.
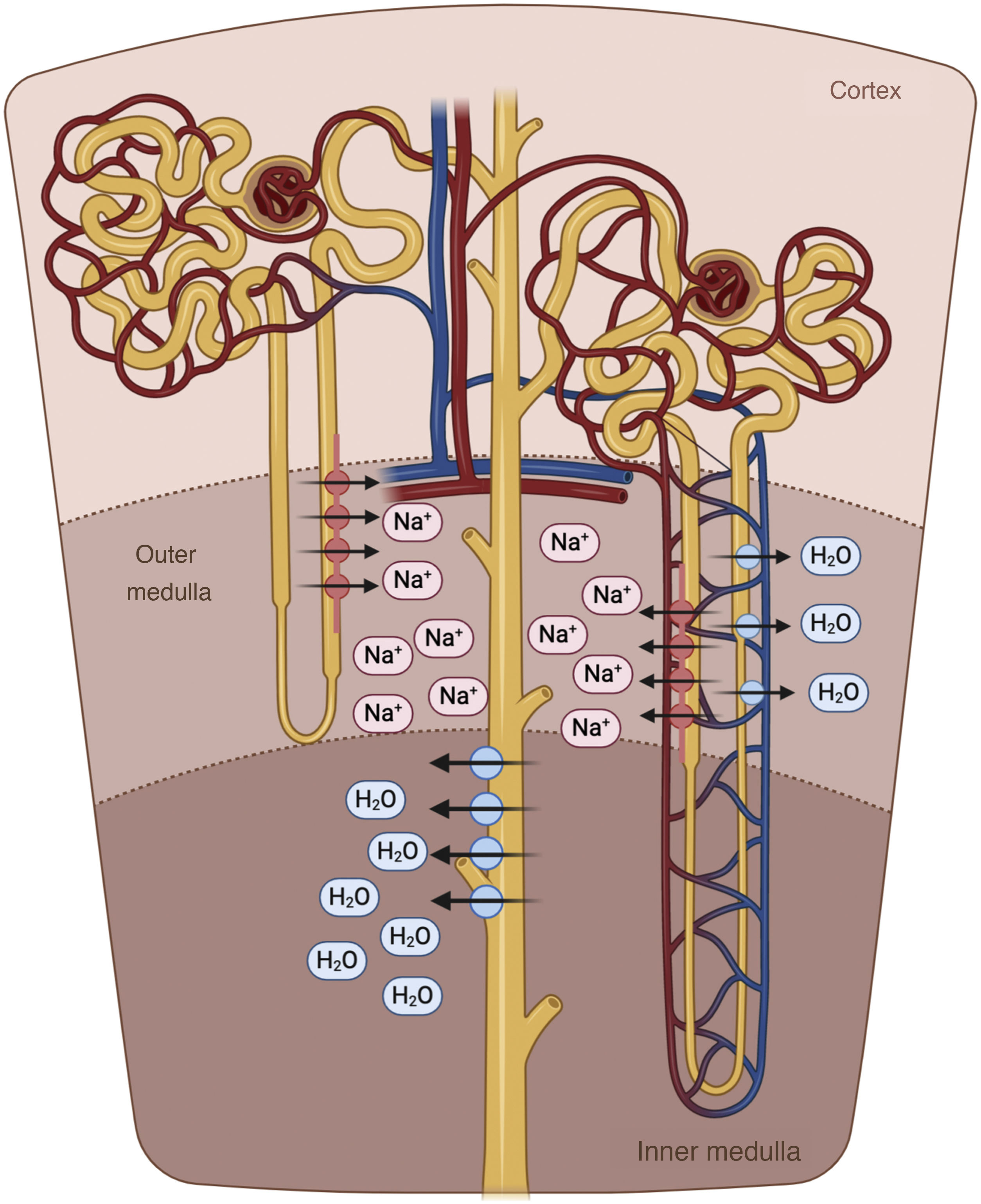
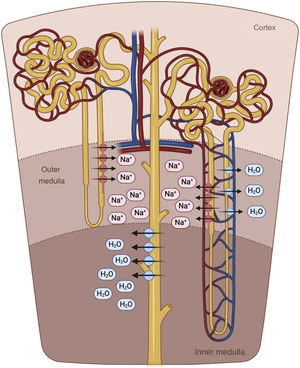
The descending limb of the loop of Henle is extremely permeable to water and less permeable to ions; the tonicity of the tubular fluid increases progressively as the loop of Henle descends from the renal cortex towards the inner part of the medulla.54 The loop of Henle becomes impermeable to water in its ascending limb, and the high tubular flow of NaCl in this tubular segment activates the Na+/K+/2Cl− cotransporter (NKCC2) in the thick limb, which dilutes the fluid and generates the necessary osmotic gradient in the interstitium of the outer medulla for vasopressin-dependent reabsorption of water in the collecting duct 55 (Fig. 5).
Loop of Henle. The descending limb of the loop of Henle is extremely permeable to water. In contrast, the thick ascending part is impermeable to water, and the high tubular flow of NaCl in this segment of the renal tubule activates the Na + /K + /2Cl − cotransporter (NKCC2) in the thick part, which dilutes the luminal fluid. and generates the necessary osmotic gradient in the interstitium of the renal medulla for vasopressin-dependent reabsorption of water in the collecting duct.
In patients with HF, natriuresis and free water excretion are compromised by multiple factors.56 The loop of Henle is fundamentally involved in three of them. First, the increased reabsorption of water and NaCl in the proximal tubule decreases the volume of filtrate reaching the loop of Henle. This point is especially important given that the NKCC2 cotransporter requires adequate concentrations of chloride (Cl−) for the reabsorption of Na+ and potassium.57 Second, neurohormonal activation produces an upregulation of the cotransporter NKCC2, which increases the active reabsorption of Na+ in the thick portion of the loop of Henle and, therefore, the tonicity of the medullary interstitium.58 Third, hypoperfusion of the vasa recta as a result of intrarenal vasoconstriction and venous congestion reduces renal medullary solute clearance, impairing the ability of the kidneys to dilute urine and excrete free water.59
Loop diuretics are the cornerstone in the treatment of congestion since they exert a powerful inhibitory effect on the NKCC2 cotransporter. Consequently, they increase the amount of NaCl reaching the distal nephron and thus interfere with the generation of the osmotic gradient in the renal medullary interstitium, decreasing the reabsorption of free water in the collecting tubule (resulting in the production of hypotonic urine). However, despite its high diuretic and natriuretic efficacy, there are a number of pharmacokinetic and pharmacodynamic considerations that must be taken into account.
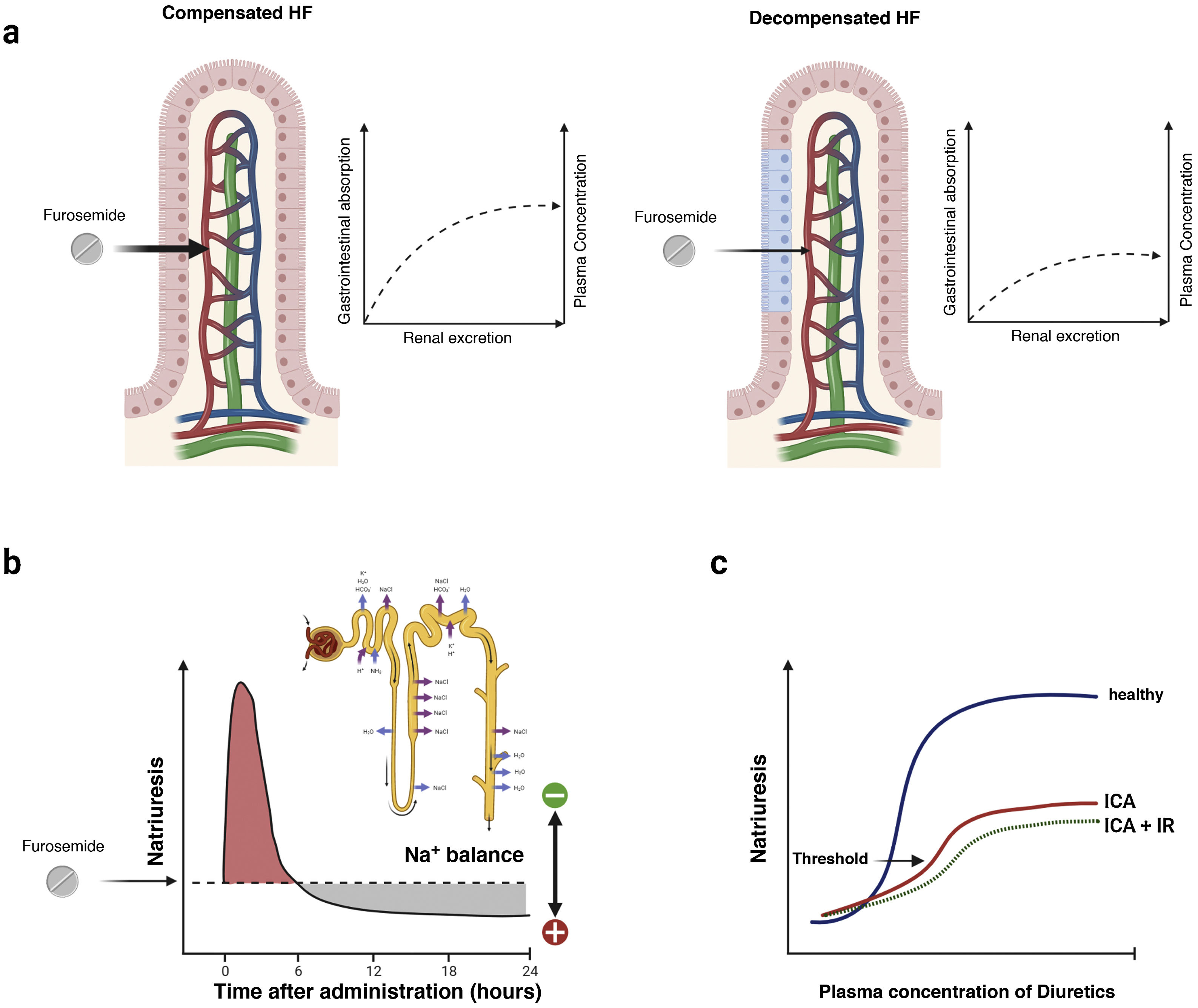
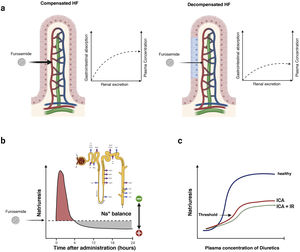
Gastrointestinal absorption and bioavailability of the oral presentationLoop diuretics are absorbed relatively rapidly by the gastrointestinal tract (onset of action in 30−60 min). However, the individual bioavailability of oral furosemide varies between 10 and 100% (mean bioavailability of 50%).60 This variation has been attributed to differences in gastric emptying and blood flow, as well as the impact of venous congestion and intestinal edema61 (Fig. 6). In contrast, the absorption and bioavailability of torasemide is more stable (>80%) and is less influenced by intestinal congestion.62 However, dose bioequivalence must be taken into account (40 mg of furosemide is equivalent to 20 mg of torasemide).
Pharmacokinetic and pharmacodynamic characteristics of furosemide. a) Absorption-dependent kinetics. Furosemide exhibits a unique pharmacokinetic property, the rate of elimination of the drug is significantly faster than the rate of absorption. Therefore, plasma levels are highly dependent on absorption rates, and any impairment in the absorption process leads to significant reductions in the half-life of this agent. b) Postdiuretic sodium retention. Loop diuretics are characterized by short half-lives. Therefore, the initial natriuresis generally decreases progressively in the 3 to 6 h after its administration. During this time, the nephron reabsorbs sodium avidly and can generate a positive balance. c) Natriuretic threshold. The administered dose must exceed a certain threshold to be effective. Thus, it is not surprising that an empirically selected dose may be ineffective. This point is also greatly influenced by the erratic bioavailability of oral furosemide.
Loop diuretics are characterized by relatively short half-lives. The initial natriuresis generally decreases progressively in the 3–6 h following its administration.63 After this time, the nephron avidly reabsorbs sodium, resulting in “postdiuretic sodium retention”64 (Fig. 6b). In this sense, dosages that maintain stable plasma concentrations would be more recommended in patients with a higher degree of congestion.
Natriuretic thresholdThe dose administered must exceed a certain threshold to be effective. Although most healthy people will respond to 20 mg of oral furosemide, the natriuresis threshold shifts up and to the right in patients with decompensated HF, even more so if they have concomitant renal dysfunction58,65 (Fig. 6c).
The braking phenomenon and structural remodeling of the nephronA feature that complicates the effectiveness of diuretic therapy derives from the structure of the nephron itself. Sodium excretion during diuretic therapy reflects a balance between inhibition of reabsorption at the primary site of action and stimulation of reabsorption at other sites in the nephron ("braking phenomenon").66 Although this process is physiological, these mechanisms contribute to diuretic resistance. In addition, chronic treatment with loop diuretics is associated with remodeling and hypertrophy of the distal convoluted and collecting duct, which increases the capacity of the distal nephron to reabsorb sodium and water.
Route of administrationThe optimal route of administration of furosemide is not well established. Contiuous infusion offers the theoretical advantage of avoiding the sodium reabsorption peak and reducing sudden changes in intravascular volume.67 Although this strategy has not been shown to reduce rehospitalizations or mortality, a meta-analysis that included a total of 923 patients from 12 studies, observed a greater reduction in weight using this strategy, without being associated with ionic disorders or renal function deterioration.68
Administration of furosemide together with hypertonic salineThe rationale for administering furosemide together with saline resides in its osmotic capacity, which favors vascular refill from the interstitium. This contributes to plasma volume expansion and counteracts the deleterious effect of intravascular depletion caused by diuretics. Clinical trials69–71 and observational studies72–74 in refractory patients have shown its effectiveness in terms of decongestion, preserving renal function, and even reducing adverse events during follow-up. However, there is a wide heterogeneity in the form of preparation and the dose of furosemide used in the different studies (Table 2).
Method of administration intravenous furosemide and hypertonic saline.
| Authors | n | Furosemide dose | Preparation | |
|---|---|---|---|---|
| Paterna S. et al. 57 | 1771 | 2011 | 500−1000 mg/12 h | 150 ml NaCl (1.4−4.6%) in 30 min |
| Tuttolmondo A. et al. 58 | 150 | 2011 | 125−1000 mg/12 h | 150 ml NaCl (1.4−4.6) in 60 min |
| Issa V. et al. 59 | 32 | 2011 | 120 mg/24 h | 100 ml NaCl (7.5%) in 1 h |
| Lafrenière G. et al. 61 | 47 | 2012 | 250 mg/12 h | 150 ml NaCl (3%) in 1 h |
| Torres M. et al. 62 | 51 | 2019 | 125 mg/24 h | 100 ml NaCl (2.4%) in 30−60 min |
*n: number of patients; min: minutes.
The consensus document on the use of diuretics in patients with HF and congestion of the European Association of Heart Failure 3 places the loop diuretic as the first line of treatment. In addition, in patients admitted for HF who present decreased initial natriuresis or insufficient diuresis, it is recommended to double the dose of loop diuretics up to high doses of furosemide (400−600 mg). This recommendation is based on clinical trials that have evaluated diuretic strategies in acute HF, in which high doses of furosemide were associated with greater resolution of congestion without being associated with adverse events during follow-up.68,68,75,76 (Appendix C Supplemental material 2).
The CARRESS-HF study evaluated veno-venous ultrafiltration vs. a standardized protocol for pharmacological treatment in patients with acute HF and impaired renal function.77 This protocol recommended perfusion of furosemide (10–20 mg/h, preceded by a bolus) and adding a thiazide if the patient continued to have congestion and his daily diuresis was less than 3 L. After evaluating 188 patients, pharmacological treatment was shown to be as effective as veno-venous ultrafiltration in resolving congestion, and produced less deterioration in renal function.
The DOSE-AHF study compared high-dose intravenous furosemide (2.5 times the baseline oral diuretic) vs. low doses (same dose of oral diuretic) in 308 patients with acute HF.75 The high-dose group obtained a greater resolution of the congestive data despite a greater percentage of worsening of renal function. However, a post-hoc analysis showed that this deterioration was associated with fewer adverse events.78
Distal tubule diureticsThiazidesThiazide diuretics exert their action in the initial part of the distal convoluted tubule, inhibiting the sodium-chloride cotransporter. Although practically 90% of the reabsorption of sodium in patients with HF occurs at a more proximal level,66 in patients treated chronically with loop diuretics there is a greater supply of sodium and a greater avidity for its reabsorption in the distal nephron.
The use of thiazides alone is not very effective in generating natriuresis in patients with HF; however, its addition to treatment with loop diuretics generates a significant increase in fractional excretion of sodium.66 Furthermore, thiazides maintain their natriuretic effect even in the presence of advanced renal failure.79 Observational studies have shown improvement in decongestion when added to furosemide in patients with advanced HF80,81; however, hydro-electrolytic disorders and renal alterations are relatively frequent and may be significant (metabolic alkalosis, hypokalemia, hyponatremia, hypomagnesemia, and renal failure), so these drugs must be used under strict control and monitoring. Hyponatremia occurs because distal natriuresis is greater than the urinary volume excreted, producing hypertonic urine. This same hypochloremic metabolic alkalosis induced by loop and/or thiazide diuretics can generate resistance to their diuretic effect, by mechanisms such as chloride depletion, reduction of drug concentration in tubular lumen and activation of renin.82
The most widely used thiazide diuretics in our environment are chlorthalidone and hydrochlorothiazide. Chlorthalidone has a longer half-life, 45−60 h vs. the 6−15 h of hydrochlorothiazide.3 It should be noted that the natriuretic effect of hydrochlorothiazide is achieved with doses approximately 1.5–2 times higher than those of chlorthalidone.83
There have been no randomized clinical trials that have shown the benefit of thiazides in HF. Intravenous chlorothiazide and metolazone have shown similar efficacy to tolvaptan in a clinical trial on 60 patients with HF and congestion refractory to diuretics, improving diuretic efficiency and generating rapid weight loss.84
Given the experience of using these drugs and their natriuretic potency in combination with loop diuretics, they are usually the treatment of first choice in patients with congestion refractory to high doses of loop diuretics.
Mineralocorticoid receptor antagonistsThe deleterious neurohormonal activation in HF leads to a state of hyperaldosteronism, which is also exacerbated during depletive treatment. At the renal level, aldosterone promotes sodium reabsorption by inducing the expression of epithelial sodium channels in the distal nephron.66
Mineralocorticoid receptor antagonists (MRA) act at the renal level in the distal nephron, inhibiting the effects of aldosterone and therefore modulating the activity and expression of sodium and potassium channels.
Spironolactone and eplerenone are the two most commonly used drugs. Both have been shown to reduce the risk of death and rehospitalization due to HF,85 and have a class I indication in the Clinical Practice Guidelines for the treatment of HF with reduced ejection fraction. However, the doses used in RALES or EMPHASIS-HF (mean daily doses of 26 mg of spironolactone and 40 mg of eplerenone, respectively)85 have little diuretic effect. In patients with HF and preserved ejection fraction, a decrease in physical signs of congestion has been observed with low-dose spironolactone (25 mg/day).86
Spironolactone at doses ≥100 mg/day is capable of inducing natriuresis, improving signs of congestion, and reducing the need for loop diuretics in patients with HF and refractory congestion.87 The main clinical trial that has evaluated the potential benefit of using high doses of MRA was the ATHENA-HF trial, which randomized 360 patients with acute HF to receive spironolactone at a dose of 100 mg daily vs. placebo (or 25 mg/day) for 96 h.88 No differences were observed in NT-proBNP levels (primary endpoint), nor in a combined endpoint of death and HF decompensation events. There were also no differences in secondary surrogate endpoints of diuretic efficacy. It is noteworthy that the drug was used for a short period of time. A pharmacokinetic substudy found that at 48 h many patients receiving the drug de novo had not reached adequate pharmacological levels.89
The main side effect of these drugs is hyperkalaemia,89 and for this reason they can be an especially attractive option in the presence of hypokalaemia.90
Diuretics with effect on the collecting ductVasopressin antagonists (AVP) exert their mechanism of action in the renal collecting duct, counteracting the action of antidiuretic hormone. In HF there are inappropriately high levels of arginine vasopressin which, at the renal level, through the stimulation of V2 receptors, promote the expression of aquaporin-2 channels, generating reabsorption of free water and therefore contributing to fluid overload and dilutional hyponatremia.66
The most widely used drug is tolvaptan, a V2 receptor antagonist that inhibits the expression of aquaporin-2 channels and, therefore, induces the excretion of free water. By inducing aquaresis but not natriuresis, plasma osmolality is increased, being effective for the correction of dilutional hyponatremia in HF.
Short-term treatment with tolvaptan has been shown to improve physical signs of congestion and induce volume loss in patients with acute HF.92 In the short term, it can generate diuretic efficacy comparable to thiazides administered concomitantly with loop diuretics. The EVEREST clinical trial randomized 4133 patients with HF with reduced ejection fraction hospitalized for HF decompensation to receive tolvaptan or placebo. Although treatment with tolvaptan improved dyspnea and signs of congestion, it had no effect on morbidity and mortality. A post-hoc analysis showed a possible benefit in reducing events in patients with a higher degree of hyponatremia (Na < 130 mEq/L).93,94
The recommended dose of tolvaptan is 30 mg/day; however, a post-marketing study in Japan, which included 3349 patients with acute HF and resistance to diuretic treatment, showed that low doses of tolvaptan (7.5 mg daily) were as effective and safer than higher doses.95 They do not produce a significant drop in blood pressure, hydroelectrolytic alterations or worsening of renal function that may even improve.91 However, high doses have been associated with liver toxicity, so liver function should be periodically evaluated. Currently its high cost is a limitation for its use.
Intra and extracorporeal ultrafiltrationExtracorporeal ultrafiltration is a method of extracting extracellular fluid that is indicated in patients refractory to intensive diuretic treatment.9 Although randomized clinical trials show heterogeneous and non-definitive results,96–100 it seems useful to consider this therapeutic option in patients refractory to the treatment.101 As an alternative to the extracorporeal ultrafiltration, continuous ambulatory peritoneal dialysis emerges as a potentially useful method of long term ultrafiltration in the outpatient setting. In this sense, there are various groups that identify a striking symptomatic improvement and a reduction in hospitalizations in patients with refractory HF.102–105
Sacubitril/ValsartanNatriuretic peptides (NP) improve RPP by reducing preglomerular vascular resistance, increasing the filtration surface achieved by the relaxation of mesangial cells, and stimulate diuresis and natriuresis by direct glomerular mechanisms (tubulo-glomerular balance) and mediated by cGMP activation.106 However, its biological function is compromised in HF patients due to degradation mediated by neprilysin activity. Sacubitril/valsartan combines the benefits derived from the inhibition of the renin-angiotensin-aldosterone system with the reduction of NP degradation as a result of neprilysin inhibition. This combination has been shown to reduce cardiovascular morbidity and mortality,107,108 reduce adverse cardiac remodeling109 and slow the progression of kidney damage in patients with HF with reduced ejection fraction.110 Additionally, treatment with sacubitril/valsartan is associated with a greater reduction in clinical signs of congestion111 and a less demand for intensification of outpatient diuretic treatment.112 Therefore, it is an interesting therapeutic option to maintain euvolemia in this subgroup of patients.
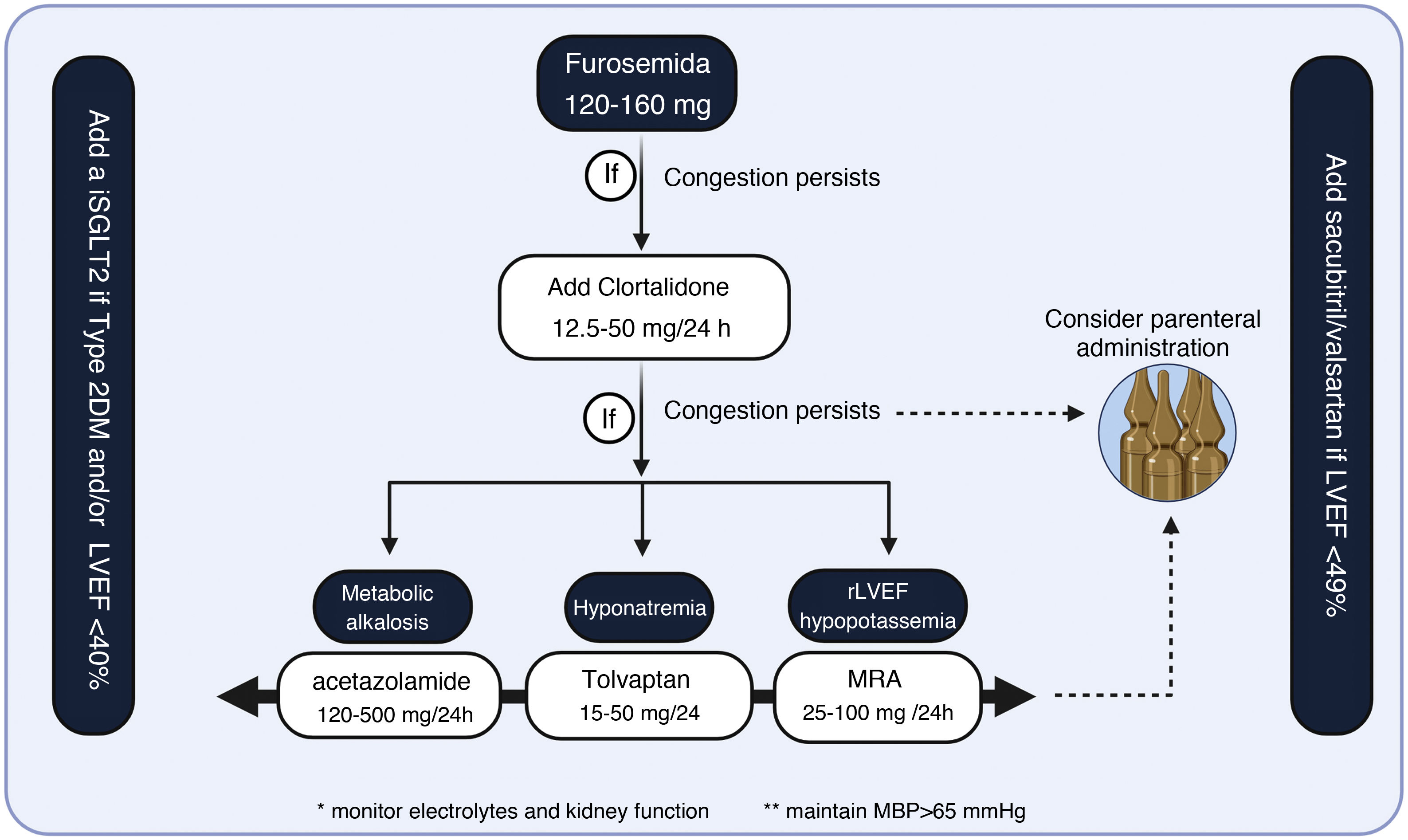
Therapeutic regimen for the management of congestion in HFIn the absence of proven evidence strategies that have demonstrated the suitability of the optimal diuretic strategy, we propose the following diagnostic/therapeutic scheme
- 1
Multiparametric quantification of congestion using common clinical parameters, noninvasive imaging techniques, and biomarkers.
- 2
Identification of the congestion profile of the patient with HF. Patients with vascular redistribution as the predominant phenotype will require less aggressive diuretic strategies. In contrast, those with volume overload (predominantly systemic and extravascular congestion phenotypes) will require more aggressive strategies.
- 3
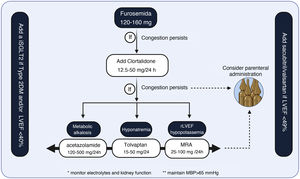
The first step in diuretic treatment should be loop diuretics. In case of refractoriness to high loop diuretic doses (120-160 mg), we propose the following algorithm (Fig. 7.
None.
Conflict of interestsThe authors declare that they have no conflict of interest.
The following is Supplementary data to this article:
Supplemental material 2. Maximum dose of intravenous furosemide
*in 72 h
¥ Technical file: https://cima.aemps.es/cima/dochtml/ft/65540/FichaTecnica_65540.html.
Please cite this article as: de la Espriella R, Santas E, Zegri Reiriz I, Luis Górriz J, Cobo Marcos M, Núñez J. Cuantificación y tratamiento de la congestión en insuficiencia cardíaca: una visión clínica y fisiopatológica. Nefrologia. 2022;42:145–162.