Various genetic and environmental factors are involved in urolithiasis. The 2 most common metabolic abnormalities are the increase in urinary calcium and low urinary citrate excretion. The ratio calculated between the concentrations of both substances is a good risk marker for the formation of calcium stones.
ObjectivesTo determine whether the risk of urinary calcium stone formation changes throughout the day in the same patient.
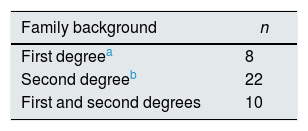
MethodsWe studied 56 children (23 V, 33 M) to check if they had prelithiasis. Calcium, citrate, and creatinine concentrations were determined in two urine samples collected one before dinner and the other in the morning. It was collected if they had ultrasound stones and if there was a history of urolithiasis in first and/or second degree relatives.
ResultsIn 25 patients (44.6%), renal ultrasound was positive for lithiasis (stones [n=9] and microlithiasis [n=16]). Forty of the 56 families (71.4%) had a history of urolithiasis. The percentage of abnormal urinary calcium (28.6%) concentrations and an abnormal calcium/citrate ratio (69.6%) was higher in the first urine of the day. The calcium/citrate ratio was the only studied parameter that was related to a family history of urolithiasis. There were no differences in urinary parameters between patients with and without ultrasound-confirmed kidney stones.
ConclusionsUrinary concentrations of calcium and the calcium/citrate ratio vary throughout the day. Urine produced at night has a higher risk of urinary calcium stone formation.
En la urolitiasis intervienen diversos factores genéticos y ambientales. Las 2 anomalías metabólicas más frecuentes son el incremento en la eliminación urinaria de calcio y la reducción en la de citrato. El cociente calculado entre las concentraciones de ambas sustancias es un buen marcador de riesgo de formación de cálculos cálcicos.
ObjetivosDeterminar si el riesgo litógeno en la orina de un mismo paciente cambia a lo largo del día.
MétodosSe estudiaron 56 niños (23V, 33M) para comprobar si eran portadores de prelitiasis. Se determinaron las concentraciones de calcio, citrato y creatinina en 2 muestras de orina recogidas, una, antes de cenar, y la otra, por la mañana, al levantarse. Se anotó si tenían cálculos ecográficos y si existían antecedentes de urolitiasis en los familiares de primer y/o segundo grado.
ResultadosEn 25 pacientes (44,6%) la ecografía renal fue positiva para litiasis (cálculos [n=9] y microcálculos [n=16]). En 40 de las 56 familias (71,4%) existían antecedentes de urolitiasis. El porcentaje mayor de valores anormales de la concentración urinaria de calcio (28,6%) y del cociente calcio/citrato (69,6%) correspondió a la primera orina del día. Este último parámetro fue el único entre los estudiados que se relacionó con los antecedentes familiares de urolitiasis. No se comprobaron diferencias en los parámetros urinarios al comparar a los pacientes con presencia o ausencia de litiasis renal ecográfica.
ConclusionesLas concentraciones urinarias de calcio y del cociente calcio/citrato se modifican a lo largo del día. Las orinas formadas durante la noche son más litógenas.
In 1939, Randall described that kidney stones present symptoms after a period of stone growth, so there is a period of variable duration that he called “symptomatic silence”.1 This period of time without symptoms is proper, although not exclusive, of the pediatric age and is known as prelithiasis period.2–4 Children on prelithiasis may show various symptoms such as macro or microscopic hematuria, urinary infection, incontinence and other voiding disorders, dysuria, emission of turbid urine or repetitive abdominal pain.3,4
For years it has been known that the urine contains substances that favor or inhibit stone formation. The two metabolic abnormalities more frequently detected, both in the prelithiasis and lithiasic period are the increase in urinary calcium excretion and the reduction of citrate. Since 1964, it has been known that a urine calcium/citrate ratio greater than 0.33mg/mg is a risk marker for calcium containing stones formation.5 This data has been subsequently confirmed in several articles.6–8
For the first time in the pediatric literature, we wanted to study if a group of children were on a situation of prelithiasis by analyzing changes in urine composition when collected at two different periods of the day.
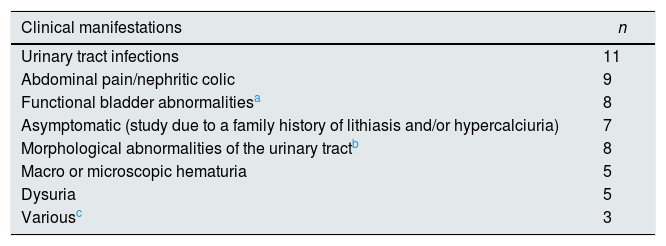
Patients and methodsTransversal study in 56 children (23M, 33F) who were evaluated to determine if they were in a situation of prelithiasis. The patients attended at the Pediatric Nephrology outpatient clinic of the University Hospital of Nuestra Señora de Candelaria between January and November 2015. Average age was 10.1±3.5 years (range: 3.5–18 years). None of them received medication. The clinical characteristics by which they were studied are shown in Table 1.
Initial clinical manifestations of the patients included in the study.
| Clinical manifestations | n |
|---|---|
| Urinary tract infections | 11 |
| Abdominal pain/nephritic colic | 9 |
| Functional bladder abnormalitiesa | 8 |
| Asymptomatic (study due to a family history of lithiasis and/or hypercalciuria) | 7 |
| Morphological abnormalities of the urinary tractb | 8 |
| Macro or microscopic hematuria | 5 |
| Dysuria | 5 |
| Variousc | 3 |
Calcium, citrate and creatinine concentrations were determined in two urine samples, one collected before dinner, and the second one in the morning (first urine of the day). The presence of calculi or micro calculi detected by ultrasound was noted as well as the evidence of family history of urolithiasis in their first or second degree relatives.
Urinary tests and renal ultrasoundThe urine collected at night was stored in a refrigerator. Both urine samples were homogenized, acidified and tempered to dissolve the presumably formed crystals. Creatinine was determined by a colorimetric kinetic test based on the Jaffé method (Creatinine Jaffé Gen.2, Roche). Calcium was quantified by photometry (Calcium Gen.2, Roche). The citraturia was measured by means of a colorimetric enzymatic technique (LTA). All these determinations were performed in a Cobas c501 analyzer (Roche Diagnostics Analyzer). To avoid interobserver variability one pediatric radiologist performed renal ultrasounds using MyLab25Gold. There were considered calculi if the size was greater than 3mm with a sonic shadow, and the microcalculi were particles in the renal calyces less than 3mm in diameter and in general without shadow cones.9
Normal valuesThe results were evaluated according to the criteria of Grases et al., which establish that a urine is potentially lithogenic if the calcium concentration is greater than 27mg/dl, the citrate is less than 230mg/l or the calcium/citrate ratio is greater than 0.33ng/mg.7,10
Statistic analysisThe Kolmogorov–Smirnov test was used to examine the distribution of the simple, then a basic statistic test was performed. All variables evaluated showed a normal distribution. Student's t-test was used to evaluate differences between quantitative variables either for independent or related samples. Fisher's exact test was used to determine the difference between qualitative variables. Probability values less than 0.05 were considered significant. These analysis were performed with the statistical package SPSS (SPSS v. 19.0, SPSS Inc., USA).
ResultsIn 25 patients (44.6%) renal ultrasound showed lithiasis. In 9 had calculi and the remaining 16 were microcalculi. The most frequent clinical onset was in urinary tract infection (n=11), abdominal pain (n=9) and functional bladder alterations (n=8) (Table 1). In 40 of the 56 families (71.4%) there was a family history of urolithiasis (Table 2).
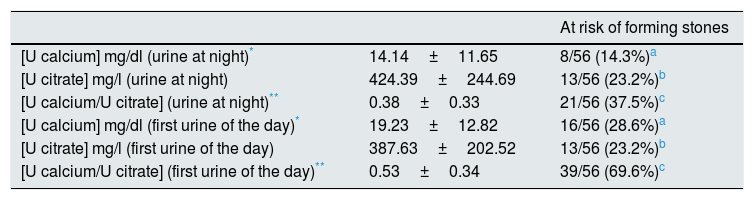
Table 3 shows the urinary values corresponding to the urine collected at night (before dinner) and the first urine of the day. The highest percentage of abnormal values of urinary calcium concentration (28.6%) and calcium/citrate ratio (69.6%) were observed in the first urine of the day. Both parameters were statistically different in the two urine samples (Table 3).
Results obtained in the urine collected at night (before dinner) and in the first urine of the day.
| At risk of forming stones | ||
|---|---|---|
| [U calcium] mg/dl (urine at night)* | 14.14±11.65 | 8/56 (14.3%)a |
| [U citrate] mg/l (urine at night) | 424.39±244.69 | 13/56 (23.2%)b |
| [U calcium/U citrate] (urine at night)** | 0.38±0.33 | 21/56 (37.5%)c |
| [U calcium] mg/dl (first urine of the day)* | 19.23±12.82 | 16/56 (28.6%)a |
| [U citrate] mg/l (first urine of the day) | 387.63±202.52 | 13/56 (23.2%)b |
| [U calcium/U citrate] (first urine of the day)** | 0.53±0.34 | 39/56 (69.6%)c |
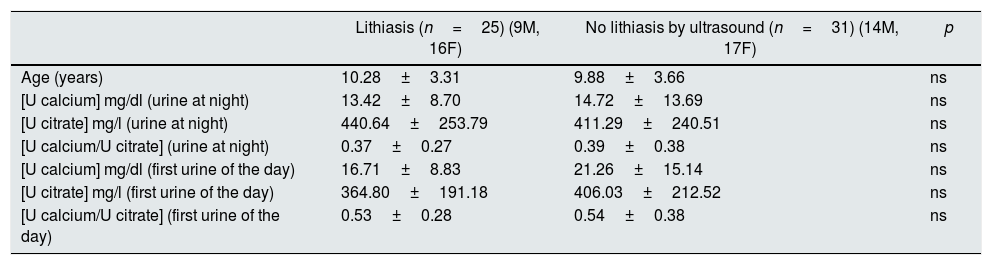
There were no differences in age or urinary parameters in patients with and without lithiasis by ultrasound (Table 4). However, 19 of the 25 cases (76%) with ultrasound lithiasis presented some lithogenic risk marker; in 17 of these 19 patients, the calcium/citrate ratio in the first urine of the day was elevated either a single risk factor (n=6) or associated to other parameters (n=11).
Age and urinary parameters separated according to the presence or absence of lithiasis by ultrasound.
| Lithiasis (n=25) (9M, 16F) | No lithiasis by ultrasound (n=31) (14M, 17F) | p | |
|---|---|---|---|
| Age (years) | 10.28±3.31 | 9.88±3.66 | ns |
| [U calcium] mg/dl (urine at night) | 13.42±8.70 | 14.72±13.69 | ns |
| [U citrate] mg/l (urine at night) | 440.64±253.79 | 411.29±240.51 | ns |
| [U calcium/U citrate] (urine at night) | 0.37±0.27 | 0.39±0.38 | ns |
| [U calcium] mg/dl (first urine of the day) | 16.71±8.83 | 21.26±15.14 | ns |
| [U citrate] mg/l (first urine of the day) | 364.80±191.18 | 406.03±212.52 | ns |
| [U calcium/U citrate] (first urine of the day) | 0.53±0.28 | 0.54±0.38 | ns |
F, female; ns, not significant; M, male.
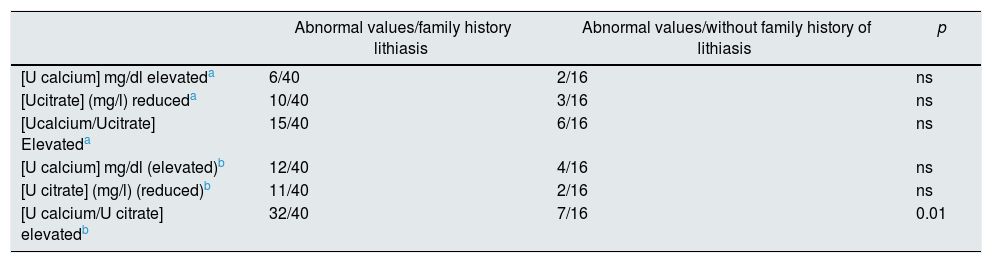
In addition, the variables were analyzed according to the family history of lithiasis. It was observed that the calcium/citrate ratio from the first urine of the day was the only parameter associated with a family history of lithiasis (Table 5). The presence of ultrasound calculi was not related to the family history of urolithiasis.
Relationship between urinary variables determined or calculated and family history of renal lithiasis.
| Abnormal values/family history lithiasis | Abnormal values/without family history of lithiasis | p | |
|---|---|---|---|
| [U calcium] mg/dl elevateda | 6/40 | 2/16 | ns |
| [Ucitrate] (mg/l) reduceda | 10/40 | 3/16 | ns |
| [Ucalcium/Ucitrate] Elevateda | 15/40 | 6/16 | ns |
| [U calcium] mg/dl (elevated)b | 12/40 | 4/16 | ns |
| [U citrate] (mg/l) (reduced)b | 11/40 | 2/16 | ns |
| [U calcium/U citrate] elevatedb | 32/40 | 7/16 | 0.01 |
ns, not significant.
The most important components of the kidney stones structure are known since the late eighteenth and early nineteenth centuries from the initial studies by Antoine François Fourcroy and Louis Nicolas Vauquelin.11,12 Apart from calculi made of cystine, uric acid and magnesium ammonium phosphate (struvite), in the rest of calculi it is commonly observed the presence of the calcium ion, either as phosphate (hydroxyapatite, brushite), oxalate monohydrate (whewellite), oxalate dihydrate (weddellite) or carbonate. Thus calcium is one of the most important promoters in the generation of kidney stones.
The citric acid, as the oxalic acid, was discovered by Scheele2; its ability to inhibit of crystallization was already appreciated In the mid-nineteenth century. Spiller observed that citric acid had a special ability to maintain calcium in solution. In the presence of citrate, calcium was not precipitated by carbonate, phosphate or oxalate; when some of these precipitates were formed, they could be brought back to solution with the addition of citrate.13 In the twentieth century, it was shown that citraturia could be a marker of body acid–base balance. Thus, Östberg described, for the first time, that the elimination of citrate in human urine increases in the presence of metabolic alkalosis and, conversely, it decreases in cases of acidosis.14 Subsequently, it was learned that hypocitraturia is one of the main causes of stone formation.15 Thereafter, the treatment with citrate would become one of the most effective methods to prevent stone formation.16,17 Even in 1960, Bibus and Luis Cifuentes demonstrated that the pharmacological treatment with citrate allowed the uric acid stones to disappear.18
Urinary ratios or indices calculated in relation to creatinine are a simple functional test of great utility in the pediatric setting, given the difficulty of collecting hourly urine samples and they do not require concomitant blood extraction.19 However, it should be remembered that values of the same parameter may be different according to the age – they are high in infants and young children – and the values may vary depending on the time of day in which the sample is collected. In our study, the urinary ratios corresponding to calcium and citrate were not calculated due to the difficulty in defining the normality values in the 2 types of samples collected and because it was not the objective of the study.
Urine is a metastable system containing several substances with the ability to crystallize and, therefore, solid particles may be generated. Normally, these substances are in supersaturated conditions, so the ability by which the crystallization process begins depends on the degree of supersaturation, the presence of favoring substances (heterogeneous nucleants) and the levels of crystallization inhibitors.7 If the concentrations of promoter and inhibitor factors are balanced, crystal formation does not occur. Crystal formation occurs if there is an unfavorable combination between the substances that favor and inhibit crystallization. The calcium/citrate ratio estimates this disbalance in a simple manner and offers the great advantage that it is not influenced by age, sex or the time of day of collection of the sample.8 DeFoor et al. noted that the calcium/citrate ratio from a 24h urine collection was the best parameter to evaluate the risk of recurrent calcium stones in children.20
Our study confirms that the composition of the urine varies throughout the day (Table 3), as it happens with other parameters, reason why the 24h urine behaves like a totum revolutum that does not let to identify the important changes that occur in the urinary composition throughout the different time periods of the day, especially during the night.2,21 In our study we confirmed that the highest percentage of abnormal values of urinary calcium concentration and calcium/citrate ratio are recorded in the first urine of the day. Bergsland et al. confirmed that subjects with idiopathic hypercalciuria excrete more calcium at night in the same amount of urine as compared with controls, with the consequent increase in the risk of stone formation. These authors indicated that the difference in the quantification of supersaturation in patients with idiopathic hypercalciuria vs controls was 7.65 for oxalate and 1.78 for calcium phosphate.22 This elevation in the calcium concentration of the nocturnal urine should be related to the increase in bone resorption described in idiopathic hypercalciuria,23–25 which is added to the physiological increase of bone resorption at night.26,27
In our study, citraturia decreased slightly but not significantly in the first urine of the day (Table 3). This is despite the tendency to a situation of metabolic acidosis, due to the nocturnal fasting, caused by the degradation, of the fatty acids a acetyl-CoA and the consequent formation of ketone bodies. As indicated above, citraturia decreases in situations of acidosis.14,28 This acidotic tendency is corrected by circadian physiological mechanisms of regulation of the acid–base equilibrium that begin with the activation of the Na+H+exchanger (NHE3: sodium-hydrogen antiporter 3) that is expressed in the renal proximal tubule.29,30 This slight reduction of the aforementioned citraturia perhaps contributed to the increase in the value of the calcium/citrate ratio observed in the first urine of the day. It was striking to observe that calcium/citrate ratio was the only parameter related to the family history of urolithiasis (Table 5). This fact illustrate that, outside of the daily interferences characteristic of the diet, the urine formed at night reveals the influence of the genetic conditions that favor lithiasis.
It should not be a surprise that the presence of lithiasis by ultrasound was not related to age, urinary parameters (Table 4) or family history of urolithiasis. The mechanism of stone formation is complex and depends on many environmental and genetic factors. It is known, that urine contains some others inhibitors of crystallization, that are independent of the diet.31 However, the results obtained in our study may be limited by the sample size.
The practical matter of this study is the consideration that the lithogenic activity is not uniform throughout the day. At night there is maximum renal water conservation, also in lithiasic or prelithiasic patients the calcium/citrate ratio is increased; therefore this nocturnal period should be the initial therapeutic objective for preventive dietary or pharmacological intervention.
Conflicts of interestThe authors declare that they have no conflicts of interest.
Please cite this article as: García Nieto VM, Pérez Bastida XI, Salvador Cañibano M, García Rodríguez VE, Monge Zamorano M, Luis Yanes MI. Cuantificación del riesgo de formación de cálculos cálcicos en la orina correspondiente a 2 momentos del día en un grupo de niños estudiados para descartar prelitiasis. Nefrologia. 2018;38:267–272.