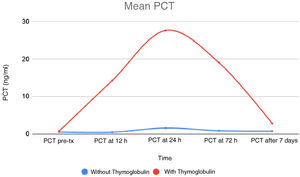
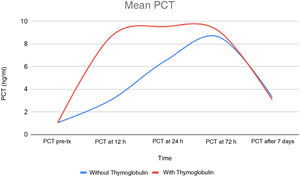
Although an increase in acute phase reactants has been observed in kidney transplant recipients treated with OKT3, there is limited information on whether other immunosuppressive drugs can cause similar abnormalities. The aim of this study was to explore whether Thymoglobulin treatment could induce a non-infectious increase in procalcitonin (PCT) in these patients.
To answer this question, a prospective observational study was conducted, including 33 consecutive patients, recipients of cadaveric donor kidney transplantation (KT). In 14 of these patients, Thymoglobulin was added to the immunosuppression protocol to treat acute rejection (four patients), as rejection prophylaxis in hyperimmunised patients (five patients) or to reduce the dose of calcineurin inhibitors in patients at high risk of graft rejection (five patients). Patients were monitored between days 7 and 30 after KT. Quantitative levels of PCT and C-reactive protein (CRP) were measured before immunosuppressive treatment and at 12 h, 24 h, 72 h and 7 days after it was started. Thymoglobulin treatment was shown to significantly increase PCT levels in these patients (Figs. 1 and 2). It is striking that only one of these patients had a urinary tract infection due to Enterococcus spp., while in two patients sepsis was initially suspected due to them having PCT levels >100, but this was ruled out as neither of them had any haemodynamic abnormalities or other signs or symptoms of active infection. The rest of the patients treated with Thymoglobulin had increased PCT levels without infectious symptoms or elevation of other parameters that might indicate infection (for example, leucocytosis, increased erythrocyte sedimentation rate, abnormal clotting). In contrast, PCT levels were normal in the remaining 19 patients who did not receive Thymoglobulin.
PCT is one of the most valuable and commonly tested biomarkers in cases of sepsis of bacterial or fungal origin.1 During infections, plasma PCT levels can exceed normal values by as much as 1000-fold,2 such that in sepsis, a correlation has been established between PCT value and sepsis severity.3
High-dose immunosuppressive regimens (anti-rejection therapy, induction therapy) and, in particular, the administration of pan-T-cell antibodies (Thymoglobulin) used in transplant patients, are associated with an increased risk of bacterial and fungal infections that are often difficult to diagnose.4 PCT monitoring has also proven to be of great interest in transplant patients. Similar to the general population, despite immunosuppressive therapy and the potential impact on the natural clearance rate of PCT under abnormal renal function, PCT increases in cases of bacterial or fungal infections in KT recipients.5
In our cohort, it appears that the use of Thymoglobulin resulted in a temporary increase in PCT levels, with a peak 24 h after administration. PCT levels do not appear to correlate with the Thymoglobulin dose (even a single 100-mg dose of Thymoglobulin increases PCT). We found that CRP levels also increased in parallel to the PCT levels in these patients, going unnoticed in most cases and secondary to surgery or the inflammatory process of acute rejection. However, CRP levels reached >20 mg/dl in patients with higher PCT levels.
This same increase has also been reported in a patient diagnosed with metastatic melanoma treated with RAF inhibitors and MEK1 and MEK2 kinase inhibitors (dabrafenib and trametinib).6 Other authors have observed increased PCT and fever in patients with solid organ cancer (without infection), and patients with solid organ cancer with PCT in the normal range and positive cultures.7–9 While increased PCT is higher and more specific in bacterial and fungal infections, it is also synthesised systemically in other scenarios such as surgery, pancreatitis and severe trauma, possibly by intestinal translocation of lipopolysaccharides or other bacterial products. This mechanism could be the cause of the increased levels observed in patients treated with immunomodulators.10
In conclusion, these data support the evidence that increased PCT levels in kidney transplant patients treated with Thymoglobulin may not be the ideal indicator of infection. It would be prudent to confirm this statement by assessing other clinical signs or symptoms before iniciating any treatment.