Mineral and bone metabolism disorders are common complications in hemodialysis patients that present significant geographical variability.
ObjectivesThe objective of this study was to assess these disorders for the first time in hemodialysis patients from Peru.
MethodsThe study included 1551 hemodialysis patients from 55 centers affiliated with the Social Health System of Peru in the city of Lima. Demographic data, comorbidities, treatments and biochemical parameters were collected from each patient. Serum calcium, phosphorus and PTH levels were categorized according to the recommended ranges in the KDOQI and KDIGO guidelines.
ResultsThe mean age of the patients was 59.5±15.6 years, with a mean time on hemodialysis of 58.0±54.2 months. All patients were dialyzed with a calcium concentration in the dialysis fluid of 3.5mEq/l and 68.9% of patients were prescribed phosphate-binding agents (98.4% of them calcium carbonate). A high percentage of patients showed serum calcium above, and serum phosphorus below, the recommended ranges in the KDOQI guidelines (32.8% and 37.3%, respectively). More than half of the patients had serum PTH values below the recommended ranges of both the KDOQI and KDIGO guidelines (56.4% and 51.6%, respectively).
ConclusionsPatients included in this study were younger than those from other studies and showed both hypophosphataemia and suppressed PTH, probably due to an excessive calcium overload through dialysis fluid and the use of calcium-containing phosphate binding agents.
Las alteraciones del metabolismo óseo y mineral son complicaciones frecuentes de los pacientes de hemodiálisis que presentan una gran variabilidad geográfica.
ObjetivosEl objetivo del presente estudio fue evaluar por primera vez dichas alteraciones en pacientes de hemodiálisis de Perú.
MétodosEl estudio incluyó 1.551 pacientes de hemodiálisis de 55 centros concertados con el seguro social de salud de Perú, pertenecientes a la ciudad de Lima. De cada paciente se recogieron datos demográficos, comorbilidades, tratamientos y parámetros bioquímicos. Los valores de calcio, fósforo y PTH fueron categorizados según los rangos recomendados en las guías KDOQI y KDIGO.
ResultadosLa edad media de los pacientes fue de 59,5±15,6, con tiempo medio en hemodiálisis de 58,0±54,2 meses. Todos los pacientes se dializaban con una concentración de calcio en el líquido de diálisis de 3,5mEq/l y el 68,9% recibían captores de fósforo (98,4% carbonato de calcio). Se observó un alto porcentaje de pacientes con calcio sérico por encima y fósforo sérico por debajo de los rangos recomendados en las guías KDOQI (32,8% y 37,3% respectivamente). Más de la mitad de los pacientes tenían valores de PTH por debajo de los rangos recomendados, tanto en KDOQI como en KDIGO (56,4% y 51,6% respectivamente).
ConclusionesLos pacientes incluidos en el presente estudio se caracterizaron por ser más jóvenes que los de otros estudios y por tener hipofosforemia y PTH suprimida, probablemente debido a una excesiva sobrecarga de calcio a través del líquido de diálisis y el empleo de captores de fósforo con calcio.
Chronic Kidney Disease-Mineral and Bone Disorder refers to a disorder that is expressed by of biochemical, skeletal and/or extraskeletal calcifications.1 A number of studies have shown an association between this disorder and an increased risk of mortality.2–12 In order to normalize and improve the management of these disorders, different clinical practice guidelines have been developed such as KDOQI13 from the National Kidney Foundation, KDIGO14 from the International Society of Nephrology and guides from Spanish15 and Argentinean16 nephrology societies, among others.
Since the venue of these guidelines, studies have been sought to know the situation of these alterations in different countries and regions; however, in Latin America there are only few studies. An exception is the CORES study, published in 2008,17 which included data from 6 Latin American countries but not from Peru.
The objective of this cross-sectional study was to evaluate the demographic characteristics, comorbidities, treatments and different biochemical parameters of patients in CKD5D treated in 55 private hemodialysis centers in the city of Lima that had a contract with the country's social health insurance (EsSalud). This study is a first step to better understand the reality of these dialysis centers in Lima in order to implement strategies aimed at improving the treatment of these patients.
Patients and methodsThis is an observational cross-sectional study that includes 1551 patients on hemodialysis from 55 private centers in the city of Lima, Peru. The data were collected between November and December 2014. The hemodialysis centers included in this analysis were those centers that obtained in 2014 a contract with EsSalud for conventional hemodialysis care of patients insured by EsSalud who could not be treated in the hemodialysis units of the national health system. This contract established quality standards in terms of personnel, facilities and service conditions (see document in supplementary material).
Data collected included: demographic data (age, gender and time on dialysis), comorbidities (diabetes, parathyroidectomy and previous transplant), treatments (calcium concentration in the dialysate, dialysis hours per week, type of phosphate binders) type of vitamin D, treatment with erythropoietin stimulating agents) and biochemical parameters (calcium, phosphorus, PTH, alkaline phosphatase, albumin, hemoglobin). All biochemical parameters were measured in the same central laboratory. Calcium was measured by a colorimetric method (NM-BAPTA) and phosphate by the ammonium molybdate method using a P 800 Roche equipment. The kit used for the determination of PTH was Elecsys 2010 from Roche (PTH Biointact 1–84) in a Cobas 601 device from Roche.
The calcium, phosphorus and PTH values were categorized according to the ranges recommended in the KDOQI guidelines (calcium corrected for albumin: 8.4–9.5mg/dl, phosphate: 3.5–5.5mg/dl and PTH: 150–300pg/ml)13 and KDIGO (calcium: normal values of 8.5–10.2mg/dl, phosphate: normal values of 3.0–4.5mg/dl and PTH: 2–9 times the maximum value of normality 130–585pg/ml).14 In addition, specific analyzes were made in which the population was categorized by quartiles of PTH and phosphate and according to the diagnosis of diabetes, tertiles of time in dialysis, prescription of phosphate binders and use of active vitamin D.
Population of patients was described as percentage. Mean and standard deviation or median and interquartile range were used according to the characteristics of each variable. The comparison between different groups was performed using Chi square, Student's t test, one-way analysis of variance and/or Wilcoxon test. All statistical analyzes were performed with software R version 3.0.1 (R Foundation for Statistical Computing, Vienna, Austria).
ResultsThe mean age of the patients was 59.5±15.6, with a mean time in hemodialysis of 58.09±54.2 months; the percentage of diabetic patients was 30.4%. The demographic characteristics and comorbidities of the population are shown in Table 1. It is important to note that all patients were dialyzed with a calcium concentration in the dialysis fluid of 3.5mEq/l. Phosphate binders was prescribed in 68.9% of patients, mostly calcium based phosphate binders (98.4% calcium carbonate and 1.6% sevelamer).
Description of patients according to their demographic characteristics, comorbidities, treatments and biochemical parameters.
| Age (years) (n, media±SD) | 1,551 | 59.5±15.6 |
| Sex (N, % men) | 1,549 | 53.9 |
| Time on dialysis (months) (n, media±SD) | 1,481 | 58.0±54.2 |
| Diabetes (n, %) | 1,545 | 30.4 |
| Parathyroidectomy (n, %) | 1,551 | 0.1 |
| Previous transplant (n, %) | 1,551 | 1.4 |
| Dialysis hours per week (hours) (n, media±SD) | 1,551 | 10.5±0.2 |
| Calcium concentration in the dailysis fluid | 1,551 | 100 |
| 2.5mEq/l (n, %) | 0 | 0 |
| 3.0mEq/l (n, %) | 0 | 0 |
| 3.5mEq/l (n, %) | 1,551 | 100 |
| Prescription of active vitamin D (n, %) | 572 | 36.9 |
| Calcitriol (n, %) | 552 | 35.6 |
| Alfacalcidol (n, %) | 0 | 0 |
| Paricalcitol (n, %) | 20 | 1.3 |
| Prescription of phosphate binders | 1,069 | 68.9 |
| Calcium (n, %) | 1,051 | 67.8 |
| Sevelamer (n, %) | 17 | 1.1 |
| Lanthanum (n, %) | 1 | 0.1 |
| Calcium (n, mean±SD) (mg/dl) | 1,551 | 9.4±0.7 |
| Calcium corrected by albumin (n, mean±SD) (mg/dl) | 1,551 | 9.3±0.7 |
| Phosphate (mg/dl) (n, mean±SD) | 1,551 | 4.2±1.7 |
| PTH (pg/ml) (n, median [IQR]) | 1,547 | 124.4 [240.3] |
| PTH (pg/ml) (n, mean±SD) | 1,547 | 262.9±411.0 |
| Alkaline phosphatase (IU/l) (n, median [IQR]) | 1,551 | 114.0 [73.0] |
| Alkaline phosphatase (UI/l) (n, mean±SD) | 1,551 | 153.6±128.4 |
| Albumin (g/dl) (n, mean±SD) | 1,551 | 4.0±0.4 |
| Hemoglobin (g/dl) (n, mean±SD) | 1,551 | 10.9±1.9 |
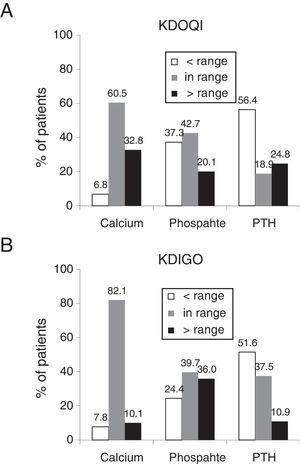
The percentage of patients who had serum calcium and phosphorus within the ranges recommended in the KDOQI clinical practice guidelines was 60.5% and 42.7% respectively (Fig. 1A) and 82.1% and 39.7% according to the KDIGO guidelines (Fig. 1B), while those with higher serum calcium and serum phosphorus lower than the recommended range by KDOQI were 32.8% and 37.3% respectively (Fig. 1A). With respect to PTH, more than half of the patients had values below the recommended ranges, both in KDOQI and in KDIGO (56.4% and 51.6% respectively [Fig. 1A and B]). The patients who had the 3 biochemical parameters (calcium, phosphorus and PTH) within the ranges recommended by the KDOQI and KDIGO guidelines represented 4.5% and 12.1% respectively.
Percentage of patients in the ranges recommended by the KDOQI (A) and KDIGO (B) guidelines.
The ranges recommended by the KDOQI guidelines are calcium corrected for albumin: 8.4–9.5mg/dl, serum phosphate: 3.5–5.5mg/dl and PTH: 150–300pg/ml13 and by the KDIGO guidelines, normal values of serum calcium: 8.5–10.2mg/dl, serum phosphate: normal values 3.0–4.5mg/dl and PTH: 2–9 times the maximum value of normality 130–585pg/ml.14
Tables 1 and 2 of the supplementary material describe the patients according to the quartiles of PTH and phosphorus. The age, percentage of men and diabetics decreased as PTH increased, while the time on dialysis increased (Table 1 of the supplementary material). No relevant differences were observed in the different quartiles of serum phosphate, except for the increase in the levels of serum albumin with the phosphate (Table 2 of the supplementary material).
The diabetic patients were older (Table 3 of the supplementary material) with a shorter dialysis time and lower PTH values as compared with non-diabetics. The values of calcium, phosphate and albumin did not show differences between both groups (Table 3 of the supplementary material).
The results related to time on dialysis are shown in Table 4 of the supplementary material in which it is observed that the patients with the longest time on dialysis were younger, with less proportion of men and diabetics and they had higher serum PTH levels.
The patients on active vitamin D prescription were younger and had higher values of PTH compared to those who were not prescribed vitamin D. The values of calcium, phosphorus and albumin were not different between both groups (Table 2).
Characteristics of patients according to the prescription of active vitamin D.
| All (n=1551) | No (n=979) | Yes (n=572) | P value | |
|---|---|---|---|---|
| Age (years) (mean±SD) | 59.5±15.6 | 60.2±15.6 | 58.3±15.6 | 0.018 |
| Gender (% men) | 53.9 | 55.5 | 51.2 | 0.1 |
| Diabetes (%) | 30.4 | 30.0 | 31.2 | 0.7 |
| Time on dialysis (months) (mean±SD) | 58.0±54.2 | 59.6±53.5 | 55.3±55.1 | 0.1 |
| PTH (pg/ml) (mean [IQR]) | 124.4 [240.2] | 113.7 [194.8] | 157.0 [278.8] | <0.001 |
| PTH (pg/ml) (median±SD) | 262.9±411.0 | 237.8±367.4 | 306.0±473.7 | 0.003 |
| Calcium (mg/dl) (mean± SD) | 9.4±0.7 | 9.3±0.7 | 9.4±0.7 | 0.6 |
| Corrected calcium (mg/dl) (mean±SD) | 9.3±0.7 | 9.3± 0.7 | 9.3±0.7 | 0.8 |
| Phosphate (mg/dl) (mean±SD) | 4.2±1.7 | 4.2±1.7 | 4.2±1.7 | 0.8 |
| Alkaline phosphatase (IU/l) (median [IQR]) | 114.0 [73.0] | 115.0 [72.3] | 114.0 [76.8] | 0.4 |
| Alkaline phosphatase (UI/l) (mean±SD) | 153.6±128.4 | 155.8±134.6 | 149.8±116.9 | 0.4 |
| Albumin (g/dl) (mean±SD) | 4.0±0.4 | 4.0±0.4 | 4.0±0.4 | 0.6 |
| Hemoglobin (g/dl) (mean±SD) | 10.9±1.9 | 10.9±1.9 | 10.9±1.9 | 0.8 |
Table 3 shows the characteristics of the patients according to the prescription of phosphate binders. The patients who were prescribed phosphate binders were younger, had a higher percentage of diabetics and had been on dialysis for less period of time. There were no differences in serum phosphate between the two groups, nor in the values of calcium, PTH, alkaline phosphatase and albumin.
Characteristics of patients according to the prescription of phosphate binders.
| All (n=1,551) | No (n=482) | Yes (n=1,069) | P value | |
|---|---|---|---|---|
| Age (years) (mean±SD) | 59.5±15.6 | 60.9±15.7 | 58.9±15.6 | 0.021 |
| Sex (% men) | 53.9 | 57.8 | 52.2 | 0.046 |
| Diabetes (%) | 30.4 | 26.6 | 32.2 | 0.030 |
| Time on dialysis (months) (Average±DS) | 58.0±54.2 | 63.5±51.5 | 55.7±55.1 | 0.01 |
| PTH (pg/ml) (median [IQR]) | 124.4 [240.2] | 125.9 [260.3] | 123.8 [223.5] | 0.6 |
| PTH (pg/ml) (mean±SD) | 262.9±411.0 | 262.9±55.9 | 262.9±433.7 | 1.0 |
| Calcium (mg/dl) (mean±SD) | 9.4±0.7 | 9.4±0.7 | 9.3±0.7 | 0.4 |
| Corrected calcium (mg/dl) (mean±SD) | 9.3±0.7 | 9.3±0.7 | 9.3±0.7 | 0.4 |
| Phosphorus (mg/dl) (mean±SD) | 4.2±1.7 | 4.2±1.7 | 4.2±1.7 | 0.5 |
| Alkaline phosphatase (IU/l) (median [IQR]) | 114.0 [73.0] | 116.0 [70.8] | 114.0 [77.0] | 0.5 |
| Alkaline phosphatase (IU/l) (mean±SD) | 153.6±128.4 | 158,6136.0 | 151.3±24.7 | 0.3 |
| Albumin (g/dl) (mean±SD) | 4.0±0.4 | 4.0±0.4 | 4.0±0.4 | 0.9 |
| Hemoglobin (g/dl) (mean±SD) | 10.9±1.9 | 10.9±2.0 | 10.9±1.8 | 0.6 |
The present study is the first large-scale study (55 centers, 1551 patients) that describes part of the dialysis population of Lima, Peru. According to data from the Ministry of Health of Peru in June 2015,18 there were 12,773 patients on hemodialysis, of which 5400 (42%) were in the capital, Lima. Therefore, the patients included in the present study (1551) represent approximately 29% of the dialysis population of Lima and 12% of the total of the country. As relevant general common features, it should be noted that in this group of patients a high percentage of patients with suppressed PTH and hypophosphatemia were observed.
Abnormalities in bone and mineral metabolism of chronic kidney disease are present to a variable degree in all hemodialysis patients with a large geographical variability, due in part to the different possibilities of access to dialysis treatment due to socio-economic factors and the differences of health coverage systems. All these factors are able to influence the demographic, etiopathogenic factors and evolution of abnormalities of bone and mineral metabolism. The patients of the present study were younger than those of the COSMOS studies (64.8±14.2)19 and DOOPS11 (61.7±14.5), but similar to those of CORES,8 the latter performed with patients from 6 countries in Latin America in which patients from Peru were not included. Other aspects such as the percentage of diabetics (30.4%) was similar to COSMOS19 (29.5%), DOOPS11 (35.1%) and the French Phosphate and Calcium Observatory (FPCO)20 (27.3%). The time on dialysis was also similar (58.0±54.2 months), COSMOS19 (50.4±52.8 months) and DOOPS11 (56.4±67.2 months).
A very distinct characteristic of these patients that probably had a great influence on the observed results is that they were dialyzed in their totality with a calcium concentration in the dialysis fluid of 3.5mEq/l, much higher than that reported in other cohorts.17,19,21 Such high concentrations of calcium in the dialysis fluid produces a positive net calcium balance,22,23 which causes a transient elevations (not always detectable) of calcium and suppression of PTH, which are 2 of the most striking findings of the present study. In addition, the use of 3.5mEq/l of calcium in the dailysate is far from the recommendations of all clinical practice guidelines, ranging between 2.5 and 3.0mEq/l.13–16 Another important difference to interpret the results lies in the weekly dialysis hours of the patients in this study (10.5±0.2h), below than the usual standards and not reaching on average the 4hours per session.17,24
The use of phosphate binders and active vitamin D (68.9% and 36.9% respectively) was also reduced as compared with other studies in Europe, the United States, Australia, Canada and Japan (COSMOS24 – 85.1% and 48%, DOOPS11 – 90.2% and 52%, FPCO20 – 80% and 48.2%). The results obtained in the 55 centers of Lima are also different from the CORES study in Latin America, however this comparison is not adequate because there is only data from patients treated with active vitamin D (45.5%).8
Although in all the aforementioned international studies the main phosphate binder was calcium based phosphate binder, the percentage of prescription was lower, since other types of binders were also used in addition to the calcium based phosphate binders. By contrast, in this Lima study basically all patients (98.4%) received calcium-containing phosphate binders. The high calcium concentration in the dialysis fluid and the almost exclusive use of calcium phosphate binders explains that the mean value of serum calcium observed in the present study (9.4mg/dl) was greater than that observed in the other studies as FPCO (8.9mg/dl),20 COSMOS (9.1±0.7)5 and DOOPS (9.3±0.9mg/dl)11 and therefore the percentage of patients with serum calcium higher than the ranges recommended in KDOQI13 and KDIGO14 and also greater than that observed in COSMOS.19
As already mentioned, in this study there was a high percentage of patients with very low PTH (Fig. 1A and B), with mean values much lower than those observed in other studies,11,17,19 probably due to the marked suppression of PTH secondary to the large load of oral calcium and through dialysis fluids. Although PTH levels were associated with factors such as age, sex, time on dialysis and diabetes (Table 1 of the supplementary material), these variables can not explain the low levels observed in the present study. A relevant difference of the present study with respect to others was a younger age, which could justify higher PTH but not lower. The association of low bone activity coinciding with a permanent overload of calcium are 2 factors that favor calcifications in soft tissues, especially arteries and heart valves.25
Another noticeable finding in this study was the high percentage of patients with low serum phosphorus, more than the percentages observed in other studies.8,11,19 Hyperphosphataemia is common in hemodialysis because dialysis is not usually sufficiently effective to eliminate the phosphate incorporated through the diet and the phosphate mobilized from bone, mainly if the PTH is elevated.26 In the present study, we do not observe a high percentage of patients with high serum phosphate, and although we do not have data on protein intake, the observed phosphate levels could suggest a low protein intake and a higher degree of malnutrition in these patients; in fact, low phosphorus levels were associated with lower levels of albumin, the only nutritional indicator in the study (Table 2 of the supplementary material). Against this possibility, the average albumin levels observed were even higher than those observed in other studies. This would allow us to speculate that while hypophosphatemia is usually responsible for low levels of PTH, the opposite can also occur when there are important sustained causes of PTH suppression, such as those observed in this study. The sustained suppression of PTH, resulting in low bone turnover, decreases the phosphate efflux from bone, and this reduction of phosphorus contribution to the bloodstream may be as important as a low phosphorus diet in the genesis of hypophosphatemia in this type of patients.
As it is already known, diabetes, like advanced age, is associated with low remodeling and with decreased PTH. In this study the presence of diabetes was also associated with lower PTH values. These results coincide with other studies such as CORES (patients from Latin America)8 and others5,6 in which a high percentage of diabetics show low PTH.
In this cohort of patients from Lima, the percentage of patients receiving VDRA (active vitamin D) was lower than that observed in other cohorts,17 therefore it is not possible to explain the high percentage of patients with very low PTH by an excess of VDRA use. By contrast, patients receiving VDRA had higher PTH values and were younger, as was observed in other studies.17,27
Finally, and in relation to the control of hyperphosphatemia, the guidelines recommend the use of phosphate binders to maintain serum phosphate levels within the recommended ranges,13,14 but guidelines do not specify the type of binder to be used. In general, it is advisable to avoid the use of calcium-containing binders in the presence of vascular calcifications, hypercalcemia, adynamic bone disease or persistently low PTH levels. In the present study, as expected, due to the high prevalence of normal and low phosphatemia, phosphate binders were prescribed to less than 70% of patients, in contrast to 80–95% in other studies.21,24 Contrary to what was expected in this study, serum phosphorus levels were not higher in the group of patients with prescription of phosphate binders, a fact that differs from that observed in other studies21,24,28 and allows us to suspect that the prescription criteria of phosphorus binders were possibly not homogeneous.
The patients included belong to 55 private centers that were dialyzed with the common quality standards required by the National Health System of Peru, but account for only about 1/3 of the patients who are dialyzed in Lima, and therefore do not represent the total hemodialysis population of Lima or Peru as a whole, this is a limitation of the study. However, an important strength of the study is that the biochemical parameters of the 1551 patients were analyzed in a centralized lab and with the same methodology. Another important positive aspect is that this analysis represents the first medium-large-scale study aiming to know and evaluate the parameters of bone and mineral metabolism in a dialysis population in Peru.
In summary, patients from this cohort of patients from Lima are characterized by being younger than other populations of hemodialysis patients studied,11,19,20 with elevated calcium concentrations in the dialysis fluid, which the exclusive use of calcium phosphate binders thus receiving a large load of calcium that justify high prevalence of patients with suppressed PTH. The later justifies a low bone remodeling that could, together with a possible low intake of phosphorus (unproven in this study), be responsible for the high prevalence of hypophosphatemia observed in this cohort. The work provides information that corresponds to approximately 1/3 of the patients in Lima and a little more than 10% of patients from Peru, but it does not allow us to generalize the results to the whole country. However, we consider that it represents a significant contribution to the knowledge of the current scenario of hemodialysis, at least of an important part of the patients of Lima. This allows to have enough data to review management strategies of patients on hemodialysis in order to reduce the alterations of bone and mineral metabolism in hemodialysis.
Financial supportThe UGC Metabolismo Oseo of the Central University Hospital of Asturias receives funding from the Carlos III Health Institute (ISCIII), ISCIII Retic REDinREN (RD06/0016/1013, RD12/0021/1023 and RD16/0009/0017), ISCIII (ICI14/00107), European Regional Development Fund (ERDF), IICA State Plan + Dica + 2013 2013–2016, Science, Technology and Innovation Plan 2013–2017 of the Principality of Asturias (GRUPIN14-028) Instituto de Investigación Sanitaria del Principado de Asturias (ISPA).
Conflicts of interestNone that could affect the objectivity of the study.
To Sara Huamaní Cucchi and Angie Vanesa Cáceres Huamaní for their contribution to the study, to all the private hemodialysis centers of the capital city of Lima-Peru and to the National Center for Renal-EsSalud Health. Dialysis centers participating in the study: Benedict XVI, C.D. Callao, C.D. Comas SAC, C.D. Renal Life, C.E. San Judas, C.N. Cipreses, Caminos del Inca, CD Jesús María, CD. Benedict X, CD. Tingo María, CD. Zárate, Cenper SAC, Renal Life Dialysis Center, SAC Renal Disease Prevention Center, Salu Center, SAC Renal Health Center, Renal Disease Center, Centro Nefrológico SA, Villa Sur Nephrological Center, San Marcelo SA Renal Center, INC. de Servicios Múltiples, S.A.C. Nordial, Grau Renal Disease Clinic, SAC Kidney Clinic, Santa Elena EIRL Comprehensive Clinic, Jesús María CM, Comas SAC, Cono Norte, Davita SA, Dialysis and Transplantation Institute SAC, Interdial, Peruvian Medical Investments SAC, Nephro Continent, Nephro Care, Nordial, Om Dial SAC, Peruvian America, Plus Life Cañete, Plusvida, Pronefros SAC, Renal Care, Renexa, Riobranco Inversiones Medicas SAC, San Fernando, San Juan Masias, San Miguel, Santa Ana, Santa Ena, Santa Patricia, Santa Rosa, St. Thomas Aquinas, Serv. of Support Diag. and Terapéutico San Miguel SA, Servibendesa SA, Servicios Espec. San Camilo S.A.C., Ventanilla, Villa María.Supplementary data
Supplementary data associated with this article can be found, in the online version, at doi:10.1016/j.nefroe.2018.03.008.
Please cite this article as: Méndez-Chacón P, Riccobelli N, Dionisi MP, Sánchez-Álvarez E, Bardales-Viguria F, Méndez-Chacón Rodríguez C, et al. Influencia de la sobrecarga de calcio sobre el metabolismo óseo y mineral en 55 centros de hemodiálisis de Lima. Nefrologia. 2018;38:279–285.