After kidney transplantation, there is an overall increase in weight, which may increase the risk of chronic kidney disease (CKD) and graft loss. But, not all patients gain weight, and the impact on the graft of this different evolution has not been well studied. The objective was to determine the causes of this different evolution and its effect on the graft.
Patients and methodsRetrospective single-center cohort study of 201 patients followed up after transplantation, analyzing the determinants of the variation in weight at one year using logistic regression, and its effect on graft survival at the end of follow-up using Cox regression.
ResultsGlobally, there was an average weight gain of 4.5 kg in the first year, but 26.6% lost weight. 37.2% increased their BMI, while 9.5% decreased it. The determinants of the different evolution of weight were age (OR for every 10 years: 0.6, p = 0.002), previous dialysis modality (ref. hemodialysis) (OR 0.3, p = 0.003), and BMI before transplantation (OR 0.9, p = 0.017). The different evolution of weight did not influence the final situation of the graft. The BMI at one year did influence as a continuous variable (HR 1.3, p = 0.003), and obesity, with a worse evolution (HR 7.0, p = 0.025).
ConclusionsAlthough not all patients gain weight after kidney transplantation, the different evolution of weight does not influence graft survival.
Tras el trasplante renal se produce de manera global un incremento del peso pudiendo aumentar el riesgo de enfermedad renal crónica (ERC) y de pérdida del injerto. Pero no todos los pacientes ganan peso, y la repercusión sobre el injerto de esta diferente evolución, no está bien estudiado. El objetivo fue determinar las causas de esta diferente evolución y su efecto sobre el injerto.
Pacientes y métodosEstudio de cohortes retrospectivo unicéntrico de 201 pacientes seguidos tras el trasplante, analizando los determinantes de la variación del peso al año mediante regresión logística, y su efecto sobre la pérdida del injerto al final del seguimiento mediante regresión de Cox.
ResultadosGlobalmente se produjo durante el primer año un aumento de peso de 4,5 kg de media, pero un 26,6% perdieron peso. El 37,2% aumentó su índice de masa corporal (IMC), mientras que el 9,5% lo disminuyó. Los determinantes de la diferente evolución del peso fueron la edad (OR por cada 10 años: 0,6; p = 0,002), la modalidad de diálisis previa (ref. hemodiálisis) (OR: 0,3; p = 0,003) y el IMC previo al trasplante (OR: 0,9; p = 0,003). La diferente evolución del peso no influyó en la pérdida del injerto. Sí influyeron el IMC al año como variable continua (HR: 1,3; p = 0,003) y la obesidad, con peor evolución (HR: 7,0; p = 0,025).
ConclusionesAunque no todos los pacientes ganan peso tras el trasplante renal, la diferente evolución del peso no influye en la supervivencia del injerto.
- –
Although most patients gain weight after kidney transplantation, there is a significant percentage of patients who lose it.
- –
Age, the type of dialysis and the BMI prior to the transplant are the determinants of weight gain or loss.
- –
Weight gain or loss itself is not associated with graft loss.
- –
The BMI reached one year after the transplant influences graft survival, presenting a greater risk of graft loss in patients with a BMI greater than 28.5 kg/m2.
Several studies have shown that obesity is a risk factor for the development of chronic kidney disease (CKD).1–3 Several factors are involved in its development and, although obese patients have a higher risk of developing diabetes mellitus (DM),4 a known cause of CKD, the risk of CKD onset and progression goes beyond that caused by DM itself.4,5
Various studies have demonstrated the existence of an increase in weight and, therefore, in the body mass index (BMI), after kidney transplantation. This weight gain occurs during the first year, and fundamentally during the first 6 months after the transplant.6 Although this increase in BMI would be associated with an increased risk of impaired graft function, as occurs in the non-transplanted population, the effect of obesity on the evolution of the transplant has not been sufficiently elucidated.7 On the one hand, there are studies that affirm that obesity does not influence the evolution of the graft,8 while the majority affirm that they find a negative effect on the function of the transplant9 demonstrating even worse renal function already in the overweight range.10,11 This weight gain occurs mainly at the expense of an increase in adipose tissue, especially visceral fat, with the consequent development of insulin resistance, increasing the risk of diabetes.12
Most studies describe an overall weight gain after kidney transplantation,6 however, in daily clinical practice we see patients who not only do not gain weight, but even lose it. The determinants of this weight gain or loss, or its impact on the evolution of the graft have not been sufficiently evaluated.
The objective of our study was to analyze the determinants of weight gain or loss after kidney transplantation and its impact on the evolution of the graft, as well as on the appearance of new diabetic patients per year, and the development of cardiovascular events (CVD) in our patient population.
Patients and methodA retrospective cohort study was perfrmed on all kidney transplant patients from January 2013 to April 2018 who had at least 12 months of follow-up after transplantation in our center. Living donor transplants were not performed during the follow-up period, and patients with double transplants were excluded.
The following variables were collected: age at the time of transplantation, sex, dialysis modality prior to transplantation, CKD diagnosis, previous diagnosis of DM, development of DM one year after transplantation (development of DM from novo during the first year after transplantation), clinical history of cardiovascular disease (ischemic heart disease, ischemic stroke, or peripheral arterial disease), number of HLA incompatibilities, type of immunosuppression, type of donation, previous kidney transplant, delayed graft function (considered as the need for at least one dialysis session in the immediate post-transplant period), development of acute rejection during the first year, age of the donor, development of major cardiovascular events during the follow-up period (acute myocardial infarction, ischemic stroke or symptomatic peripheral arterial disease), and cumulative steroid dose during the first year. This steroid dose was calciulated ba adding the daily dose of oral prednisone administered during the first, the doses of methylprednisolone administered during the peri-transplant period (500 mg prior to the transplant and 125 mg 24 h after the transplant), the doses of steroids administered in case of acute rejection (500 mg in a single daily bolus for 3 consecutive days), and the doses administered prior to the administration of thymoglobulin when it was considered. To unify the dosage of steroids, the doses of methylprednisolone have been transformed by their equivalence in prednisone, at a ratio of methylprednisolone:prednisone of 1:1.25.
It was recorded the time (months) on dialysis prior to the transplant, as well as the follow-up time (months) after the transplant. The graft situation at the end of the follow-up was recorded as functioning graft vs. return to dialysis due to graft loss, censoring deaths with a functioning graft.
Height and weight were collected at the time of transplantation, at 3, 6, 9, and 12 months and at the end of follow-up. Thus, the BMI was calculated using the formula BMI = weight (kg)/height (m2) and the body surface area using the Dubois & Dubois formula (m2). The BMI stages were established according to the WHO references using the stage of reference the interval 18.5–25 kg/m2; obesity was defined as a BMI equal to or greater than 30 kg/m2. Weight gain or loss at one year and at the end of follow-up was calculated using the difference between the weight at one year or at the end of follow-up and the weight at the time of transplantation. A binary variable was created based on weight gain or loss. Other binary variables were generated using as cut-off points a BMI greater or less than 30 kg/m2, greater or less than 25 kg/m2, and according to the value of BMI that was associated with a greater risk of graft loss.
Variables are expressed as mean and standard deviation (SD) for quantitative variables or as median and interquartile range (IQR) according to whether there was a normal distribution of the variable as analyzed using the Kolmogorov–Smirnov test; and as number and percentage for qualitative variables. The significance of the change in weight during the study period was analyzed using the non-parametric Wilcoxon Test, while the change in BMI was assessed using the non-parametric McNemar test. The student’s t test or the Mann–Whitney U test was used to compare continuous variables between groups, depending on the case. Categorical variables were compared by Chi-square or Fisher’s exact test as appropriate. The relationship between continuous variables was made using the Pearson or Spearman correlation coefficient, depending on the case. Using multivariate logistic regression, the determinants of weight gain or loss were established using the “successive steps backwards” method. The Cox proportional hazards regression model, adjusted for possible confounding variables, was used to analyze the influence of weight gain or loss after transplantation on graft loss at the end of follow-up, and the effect of the BMI value on the final evolution. Graft survival based on the weight gain or loss as well as the BMI stage, was evaluated using the Kaplan–Meier method using log-rank to establish the presence of statistically significant differences. The influence of weight gain or loss, or the BMI adjusted for the confounding variables, in the development of new diabetics one year after the transplant was analyzed by multivariable logistic regression. The Cox proportional hazards regression model was used to analyze the influence of post-transplant weight gain or loss, adjusted for possible confounding variables, on the occurrence of a major cardiovascular event (CVD) at the end of the follow-up period. A p < 0.05 was considered statistically significant.
ResultsThere were 201 patients collected, of which 17 (8.5%) returned to dialysis during the study period; the median follow-up was 47 months. Twenty-one patients (10.4%) died with a functioning graft. The median age was 56.1 years (IQR: 17.9), with 67.5% males. The characteristics of the sample are shown in Table 1.
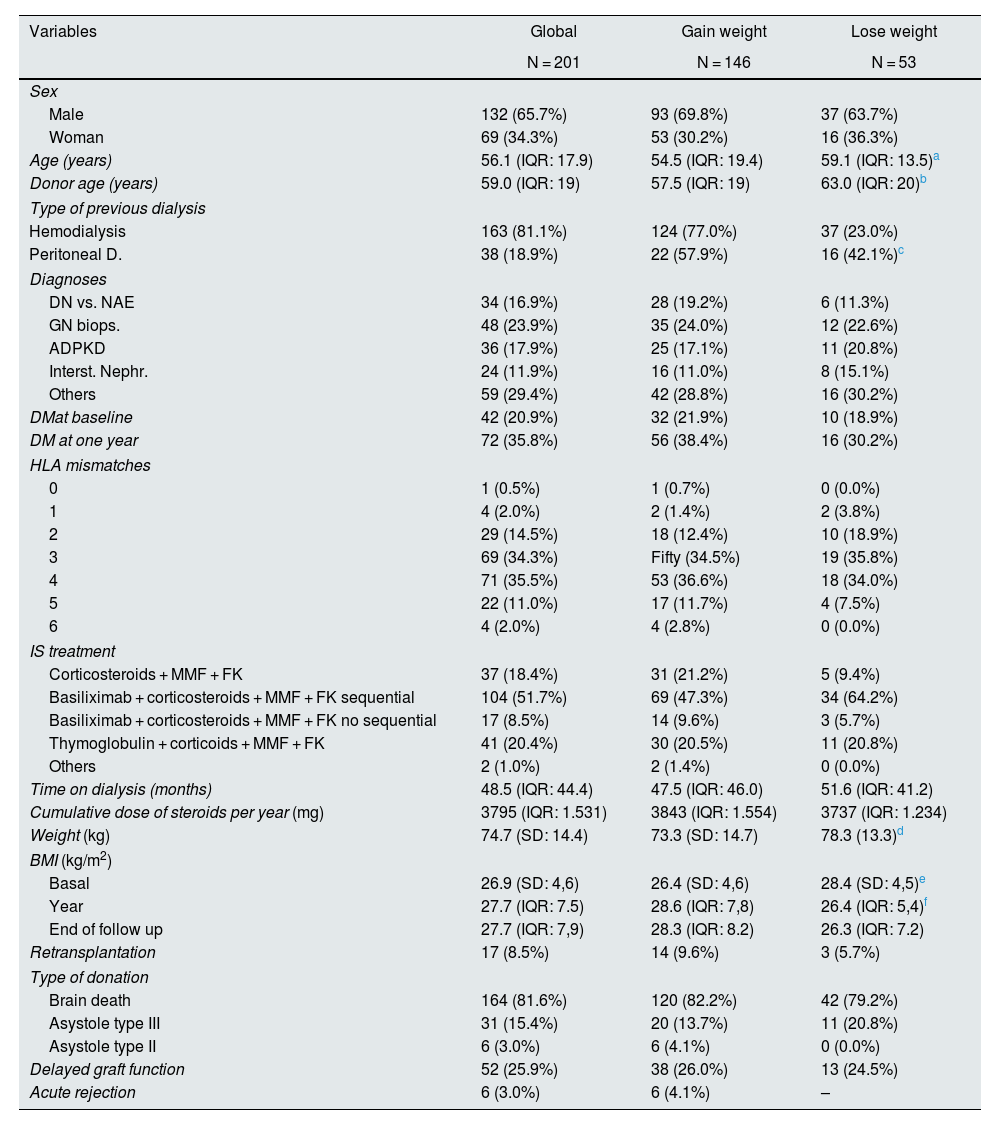
Baseline characteristics separated according to weight gain or loss per year.
| Variables | Global | Gain weight | Lose weight |
|---|---|---|---|
| N = 201 | N = 146 | N = 53 | |
| Sex | |||
| Male | 132 (65.7%) | 93 (69.8%) | 37 (63.7%) |
| Woman | 69 (34.3%) | 53 (30.2%) | 16 (36.3%) |
| Age (years) | 56.1 (IQR: 17.9) | 54.5 (IQR: 19.4) | 59.1 (IQR: 13.5)a |
| Donor age (years) | 59.0 (IQR: 19) | 57.5 (IQR: 19) | 63.0 (IQR: 20)b |
| Type of previous dialysis | |||
| Hemodialysis | 163 (81.1%) | 124 (77.0%) | 37 (23.0%) |
| Peritoneal D. | 38 (18.9%) | 22 (57.9%) | 16 (42.1%)c |
| Diagnoses | |||
| DN vs. NAE | 34 (16.9%) | 28 (19.2%) | 6 (11.3%) |
| GN biops. | 48 (23.9%) | 35 (24.0%) | 12 (22.6%) |
| ADPKD | 36 (17.9%) | 25 (17.1%) | 11 (20.8%) |
| Interst. Nephr. | 24 (11.9%) | 16 (11.0%) | 8 (15.1%) |
| Others | 59 (29.4%) | 42 (28.8%) | 16 (30.2%) |
| DMat baseline | 42 (20.9%) | 32 (21.9%) | 10 (18.9%) |
| DM at one year | 72 (35.8%) | 56 (38.4%) | 16 (30.2%) |
| HLA mismatches | |||
| 0 | 1 (0.5%) | 1 (0.7%) | 0 (0.0%) |
| 1 | 4 (2.0%) | 2 (1.4%) | 2 (3.8%) |
| 2 | 29 (14.5%) | 18 (12.4%) | 10 (18.9%) |
| 3 | 69 (34.3%) | Fifty (34.5%) | 19 (35.8%) |
| 4 | 71 (35.5%) | 53 (36.6%) | 18 (34.0%) |
| 5 | 22 (11.0%) | 17 (11.7%) | 4 (7.5%) |
| 6 | 4 (2.0%) | 4 (2.8%) | 0 (0.0%) |
| IS treatment | |||
| Corticosteroids + MMF + FK | 37 (18.4%) | 31 (21.2%) | 5 (9.4%) |
| Basiliximab + corticosteroids + MMF + FK sequential | 104 (51.7%) | 69 (47.3%) | 34 (64.2%) |
| Basiliximab + corticosteroids + MMF + FK no sequential | 17 (8.5%) | 14 (9.6%) | 3 (5.7%) |
| Thymoglobulin + corticoids + MMF + FK | 41 (20.4%) | 30 (20.5%) | 11 (20.8%) |
| Others | 2 (1.0%) | 2 (1.4%) | 0 (0.0%) |
| Time on dialysis (months) | 48.5 (IQR: 44.4) | 47.5 (IQR: 46.0) | 51.6 (IQR: 41.2) |
| Cumulative dose of steroids per year (mg) | 3795 (IQR: 1.531) | 3843 (IQR: 1.554) | 3737 (IQR: 1.234) |
| Weight (kg) | 74.7 (SD: 14.4) | 73.3 (SD: 14.7) | 78.3 (13.3)d |
| BMI (kg/m2) | |||
| Basal | 26.9 (SD: 4,6) | 26.4 (SD: 4,6) | 28.4 (SD: 4,5)e |
| Year | 27.7 (IQR: 7.5) | 28.6 (IQR: 7,8) | 26.4 (IQR: 5,4)f |
| End of follow up | 27.7 (IQR: 7,9) | 28.3 (IQR: 8.2) | 26.3 (IQR: 7.2) |
| Retransplantation | 17 (8.5%) | 14 (9.6%) | 3 (5.7%) |
| Type of donation | |||
| Brain death | 164 (81.6%) | 120 (82.2%) | 42 (79.2%) |
| Asystole type III | 31 (15.4%) | 20 (13.7%) | 11 (20.8%) |
| Asystole type II | 6 (3.0%) | 6 (4.1%) | 0 (0.0%) |
| Delayed graft function | 52 (25.9%) | 38 (26.0%) | 13 (24.5%) |
| Acute rejection | 6 (3.0%) | 6 (4.1%) | – |
DM: diabetes mellitus; SD: standard deviation; eGFR: estimated glomerular filtration rate (CKD-EPI) DN vs. NAE: diabetic nephropathy vs. nephroangiosclerosis; BMI: body mass index; ADPKD: autosomal dominant polycystic kidney disease; IQR: interquartile range.
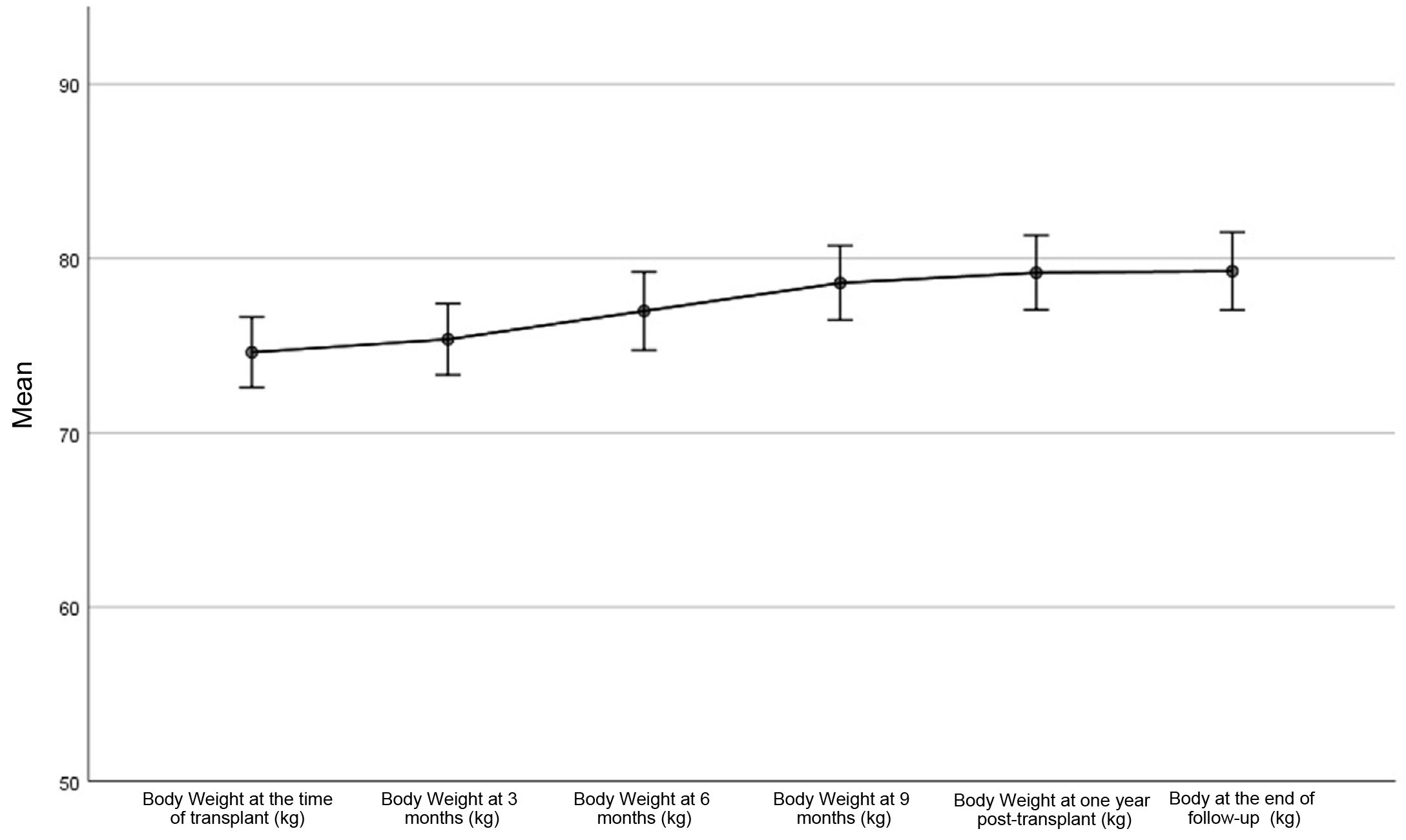
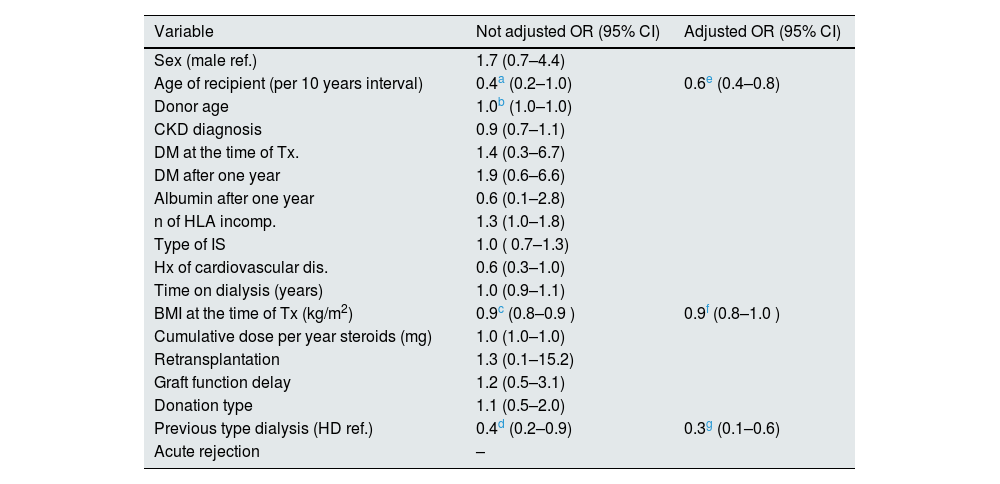
Overall, there was a statistically significant weight gain during the first year after transplantation. The mean weight gain was 4.5 kg (95% CI: 3.5–5.6; p < 0.001). This trend was maintained until the end of the follow-up, although the weight gain after the first year until the end of the study was small and not statistically significant: mean 0.09 kg, 95% CI: −0.9 to 1.0; p = 0.858 (Fig. 1). One year after the transplant, 146 patients (73.4%) had gained weight (median 7.0 kg [IQR: 7.6]). The rest of patients, a 26.6% of the total (53 patients) presented weight loss: −3.9 kg (IQR: 5.3). At the end of follow-up, 142 patients (71.4%) had gained weight since the time of transplantation (7.2 kg, [IQR: 9.2]) and 28.6% of patients lost weight [−4.4 kg, (IQR: 5.0]). A 54.3% of patients had a weight gain greater than 5%, while 11.6% lost more than 5% of their weight since transplantation. There were no significant differences in weight gain between men and women (p = 0.637 and p = 0.256 for one year and end of follow-up, respectively), and no relationship was found between weight gain and the cumulative dose of steroids received (p = 0.358 and p = 0.455 for the year and end of follow-up, respectively). By multivariable logistic regression, the patient’s age (OR per 10 years: 0.6; 95% CI: 0.4–0.8; p = 0.002), previous dialysis modality (OR: 0. 3; 95% CI: 0.1–0.6; p = 0.003), and BMI at the time of transplantation (OR: 0.9; 95% CI: 0.8–1.0; p = 0.003), were the determinants of weight gain or loss, with a tendency towards weight loss in patients with a higher BMI at the time of transplantation, with more age and those coming from peritoneal dialysis (Table 2).
Univariate and multivariate logistic regression. Determinants of change in weight one year after transplantation.
| Variable | Not adjusted OR (95% CI) | Adjusted OR (95% CI) |
|---|---|---|
| Sex (male ref.) | 1.7 (0.7–4.4) | |
| Age of recipient (per 10 years interval) | 0.4a (0.2–1.0) | 0.6e (0.4–0.8) |
| Donor age | 1.0b (1.0–1.0) | |
| CKD diagnosis | 0.9 (0.7–1.1) | |
| DM at the time of Tx. | 1.4 (0.3–6.7) | |
| DM after one year | 1.9 (0.6–6.6) | |
| Albumin after one year | 0.6 (0.1–2.8) | |
| n of HLA incomp. | 1.3 (1.0–1.8) | |
| Type of IS | 1.0 ( 0.7–1.3) | |
| Hx of cardiovascular dis. | 0.6 (0.3–1.0) | |
| Time on dialysis (years) | 1.0 (0.9–1.1) | |
| BMI at the time of Tx (kg/m2) | 0.9c (0.8–0.9 ) | 0.9f (0.8–1.0 ) |
| Cumulative dose per year steroids (mg) | 1.0 (1.0–1.0) | |
| Retransplantation | 1.3 (0.1–15.2) | |
| Graft function delay | 1.2 (0.5–3.1) | |
| Donation type | 1.1 (0.5–2.0) | |
| Previous type dialysis (HD ref.) | 0.4d (0.2–0.9) | 0.3g (0.1–0.6) |
| Acute rejection | – |
DM: diabetes mellitus; CKD: chronic kidney disease; HD: hemodialysis; HLA: Human Leukocyte Antigen; 95% CI: 95% confidence interval; BMI: body mass index; IS: immunosuppression; OR: odds ratio; Tx: kidney transplant.
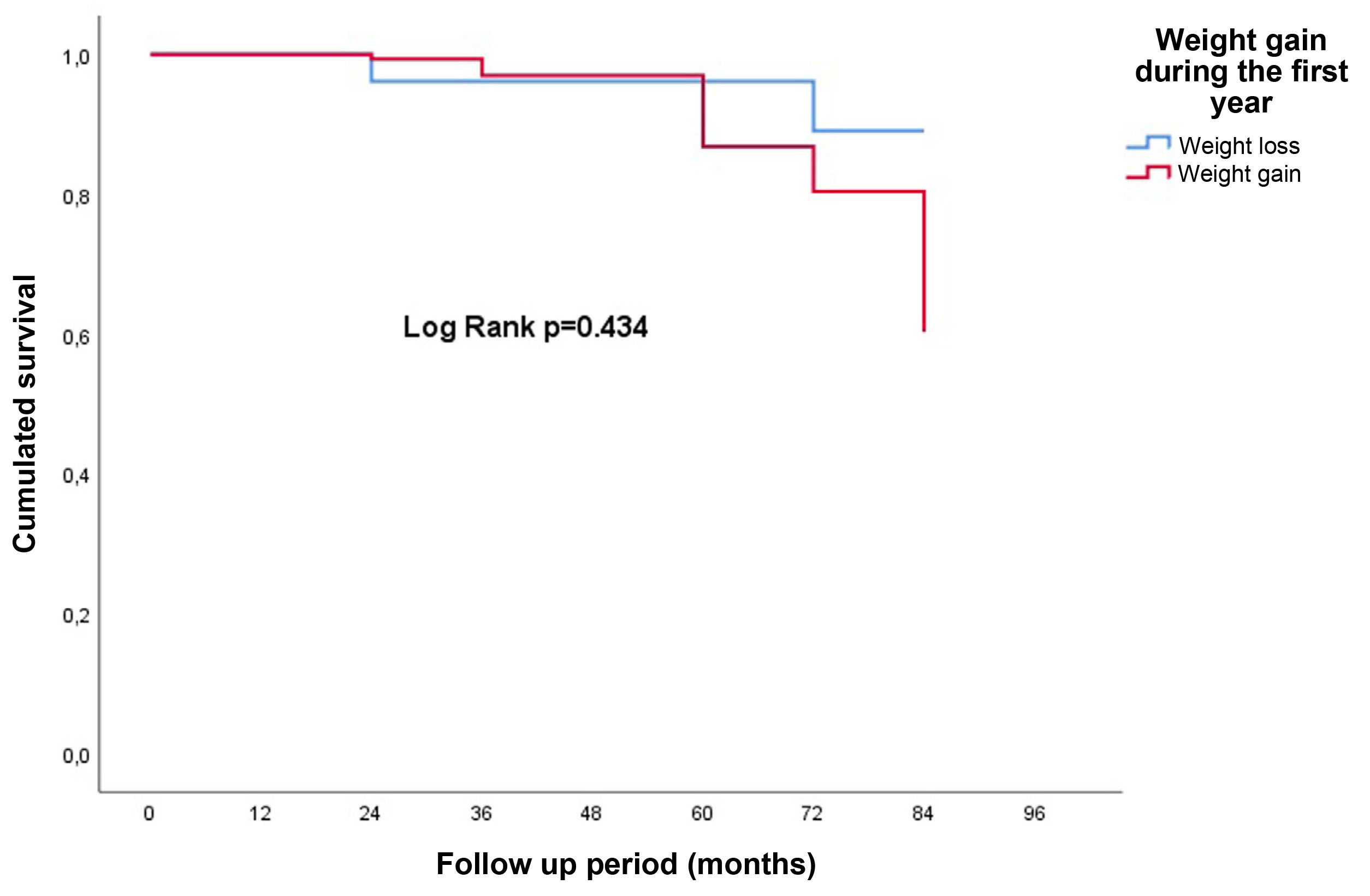
Using the multivariable Cox regression model adjusted for potential confounding variables, weight gain or loss after one year “per se” did not influence graft loss: HR: 1.1; 95% CI: 0.3–4.8; p = 0.895 (Fig. 2). Neither did it matter whether the weight gain or loss was greater than 5% (p = 0.5).
Influence of body mass index on the evolution of the graftAt the time of transplantation there were 49 patients (24.6%) with a BMI greater than 30 kg/m2. This percentage of obese patients increased to 37.5% (75 patients) after one year and to 39.5% (79 patients) at the end of follow-up; these differences were statistically significant for both comparisons (p < 0.001). The mean age of the obese patients was 59.1 (IQR: 13.7) years, being higher than that of the non-obese: 55.7 (IQR: 19.0) years (p = 0.023).
Of the 73 patients who underwent transplantation with a normal BMI, 30 (41.1%) evolved to be overweight, and one (1.4%) to grade 1 obesity one year after transplant; none lost weight. Seventy-five patients were transplanted with overweight, 30 (40.0%) evolved to grade 1 obesity, and one patient (1.3%) to grade 2 obesity; 8 patients (10.7%) went from overweight to normal BMI. Among the transplanted patients, 40 had grade 1 obesity; of them, 9 (22.5%) evolved to grade 2 obesity, and 2 (5.0%) to morbid obesity; 6 patients (15.0%) became overweight; none evolved to normal BMI. One patient (12.5%) of the 8 with grade 2 obesity changed their stage to morbid obesity; a 37.5% (3 patients) went on to grade 1 obesity; none changed to normal BMI. Two patients underwent a transplant with a BMI of less than 18.5 kg/m2, both evolved to normal BMI after the transplant. One patient underwent a transplant with morbid obesity, remaining in the same situation after one year.
After one year, 74 patients (37.2%) increased their BMI, while 9.5% decreased it. The rest maintained the same BMI as at the time of transplantation. A 14.4% of patients went from normal BMI to overweight, and 1.0% from normal BMI to obese, while 4.0% went from overweight to normal BMI. No obese patient went to normal BMI. A 25.0% of the patients had a normal BMI one year after the transplant, with 112 patients (61.0%) without obesity. This percentage of normal BMI rose to 27.5% at the end of follow-up.
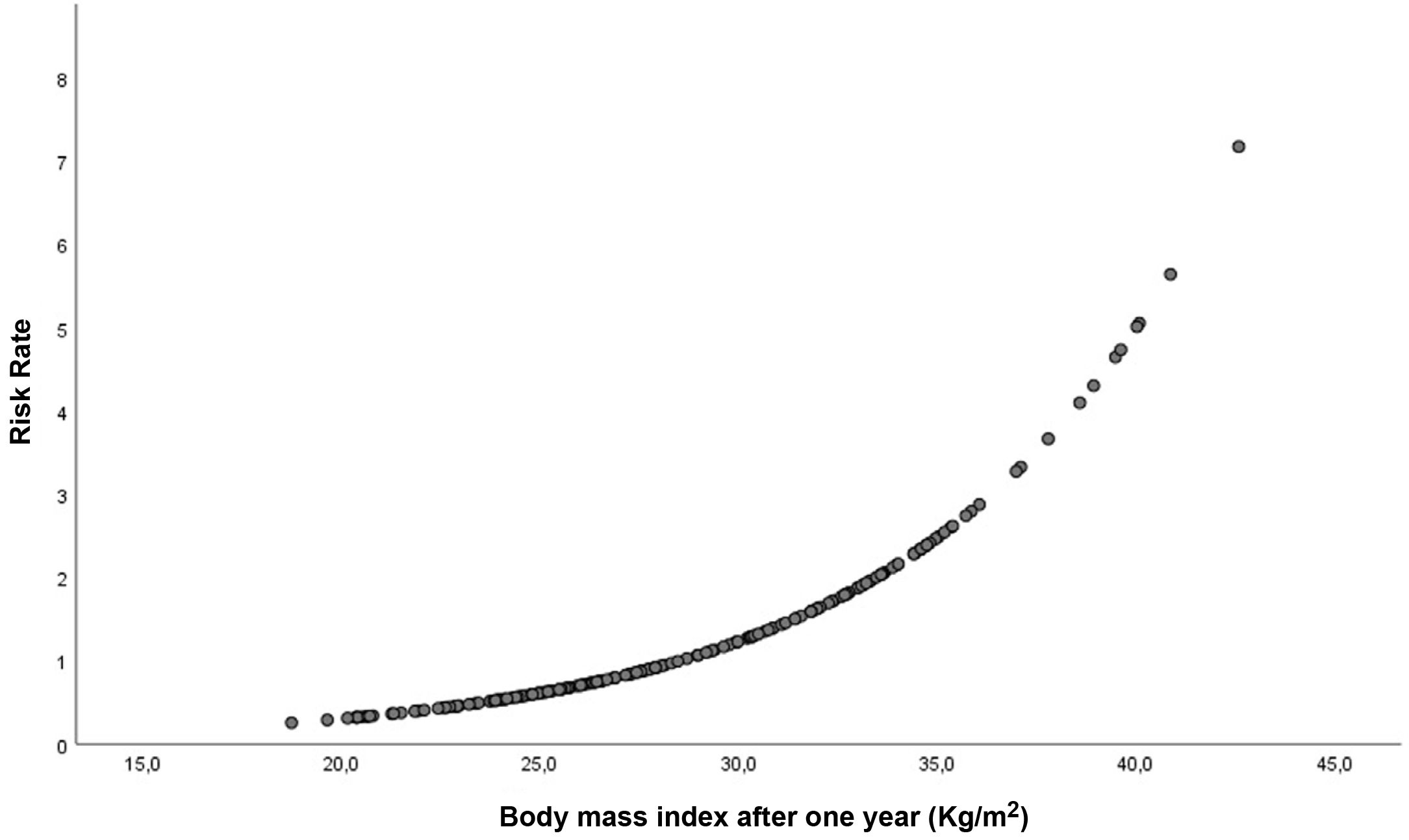
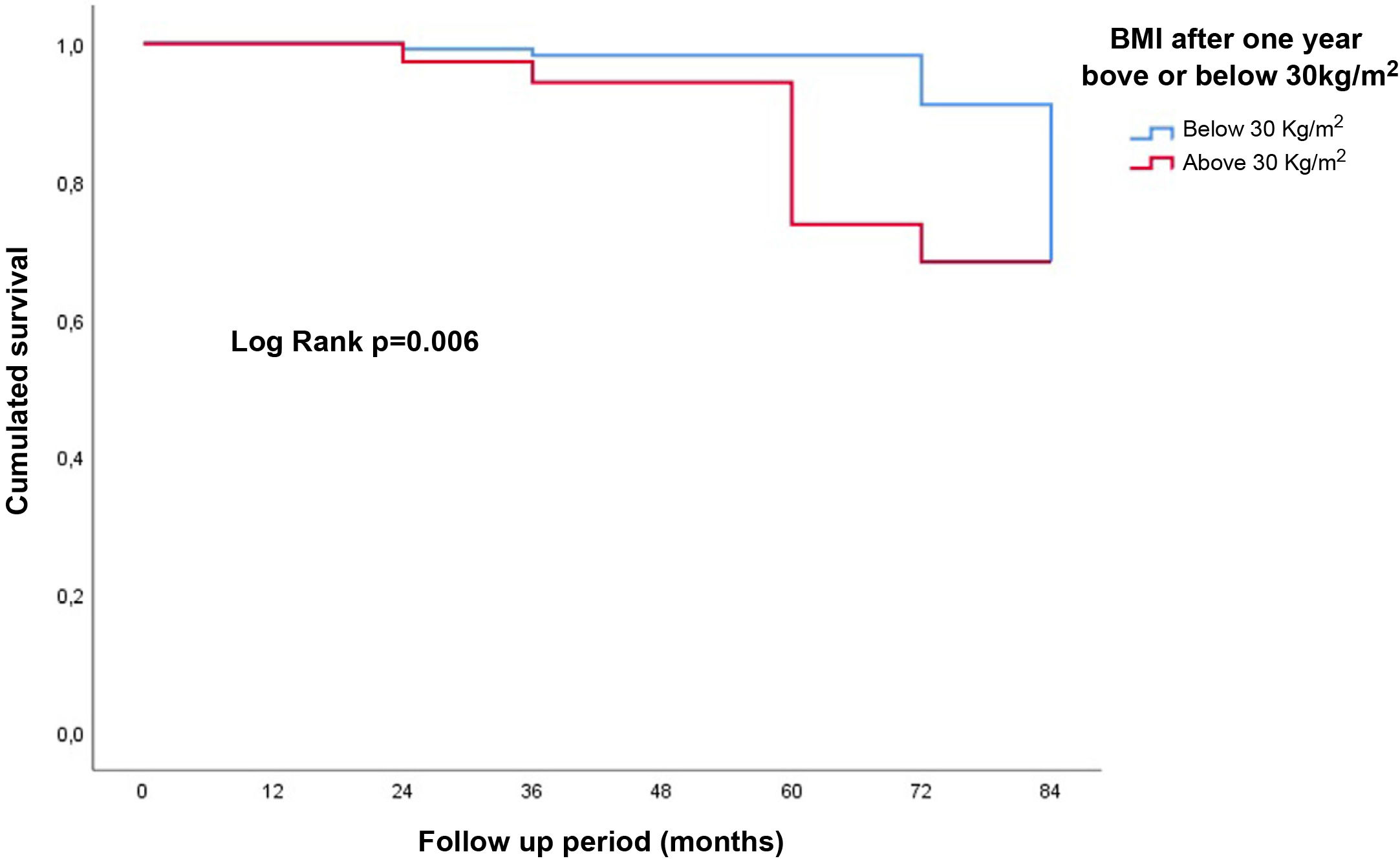
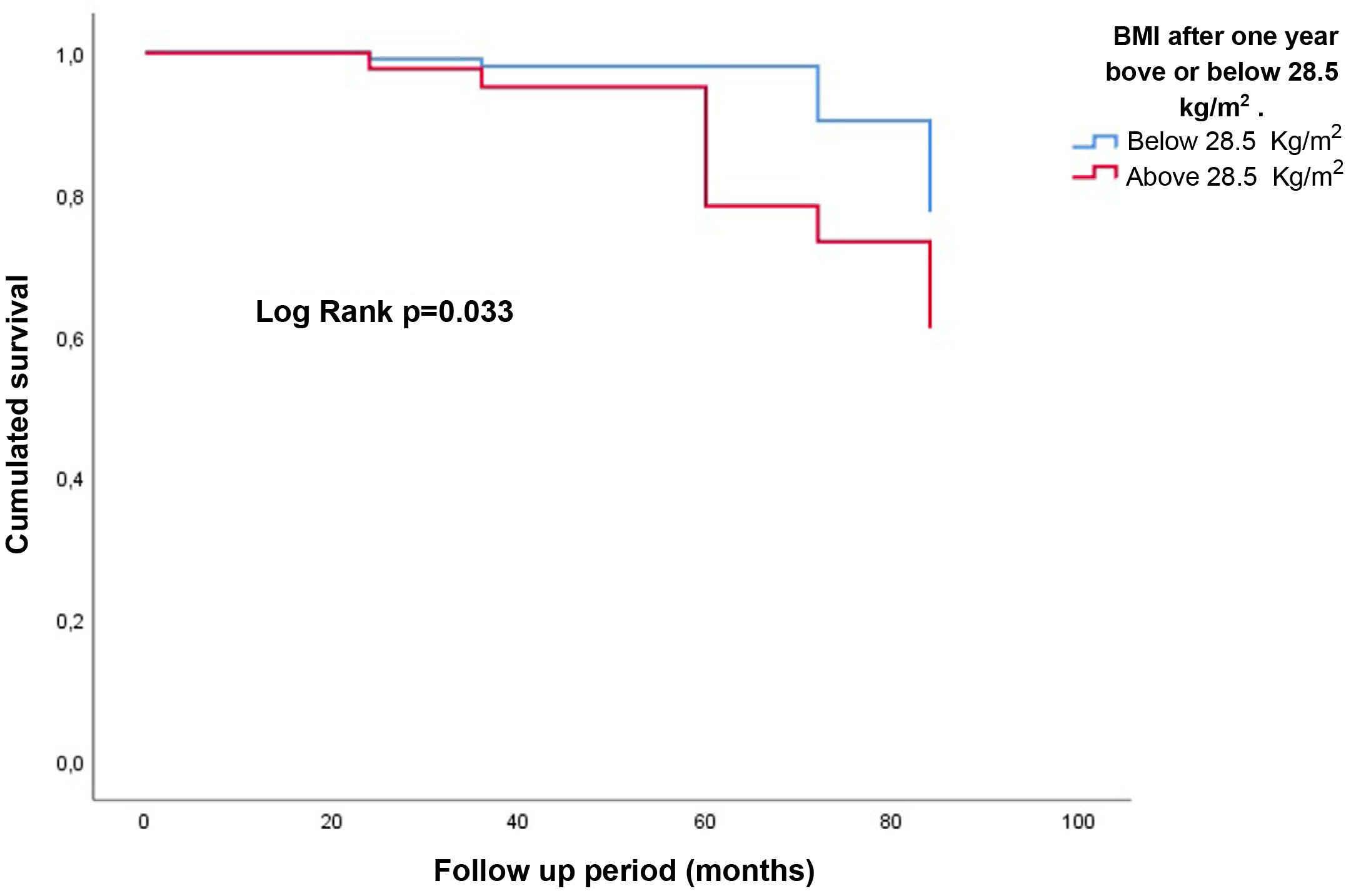
In adjusted multivariate Cox regression analysis, BMI as a continuous variable was associated with poorer graft survival: HR 1.3; 95% CI: 1.1–1.6; p = 0.003 (Fig. 3). Obesity at one year (BMI ≥ 30 kg/m2) negatively influenced the evolution of the graft at the end of follow-up: HR: 7.0 (95% CI: 1.3–38.0); p = 0.025 (Fig. 4). No increased risk was found by using, as the cut-off point, a BMI above normal (25 kg/m2). In our sample of patients, the value of BMI at one year of transplant from which the risk of graft loss increases was 28.5 kg/m2. Patients with a BMI above this value had a higher risk of graft loss: HR: 3.4 (95% CI: 1.1–32.1; p = 0.039) (Fig. 5).
Relationship of body mass index and weight gain or loss with the development of diabetes mellitus and cardiovascular eventsOne year after the transplant there were 14.9% of new diabetic patients, increasing from 20.9% at the time of the transplant to 35.8% after one year of follow-up. Weight gain during this first year was not related to the development of new diabetics (p = 0.360). The BMI was not related to the appearance of diabetes, both as a continuous variable (p = 0.954) and by BMI stages (p = 0.665). The determinants of the appearance of new diabetics one year after the transplant using multivariable logistic regression were age: OR 2.4 for periods of 10 years of age (95% CI: 1.4–4.0), p < 0.001; time on dialysis prior to transplant: OR 1.1 per year (95% CI: 1.0–1.2), p = 0.022, and the type of donor: asystole type III (OR: 3.4; 95% CI: 1.2–9.6; p = 0.022) and asystole type II (OR: 7.9 ; 95% CI: 1.1–55.6; p = 0.037) taking cadaveric donation as reference.
During the follow-up period, 21 patients (10.4%) had a major CVD; the most frequent event was acute coronary syndrome (42.9%), followed by peripheral arterial disease in 33.3% of cases and ischemic stroke in 19.0% of cases. Weight gain or loss per se, or the BMI did not influence the development of CVD. The previous history of cardiovascular disease and being diabetic at one year were associated with a higher risk of new cardiovascular events: HR: 1.9; 95% CI: 1.3–2.9; p = 0.001 and HR: 5.6; 95% CI: 1.9–16.9; p = 0.002, respectively.
DiscussionOur results show that, although in general there is weight gain after kidney transplantation, there is also a significant percentage of patients who lose weight. In our patients the determinants of this weight gain or loss is the age with less weight gain or loss at older ages. Similar data are reported by other studies where they find an inverse relationship between age and weight gain after transplantation, the greater incresae in weight observed in younger patients is perhaps related to more food intake when the dietary restrictions associated to chronic renal failure are discontinued.13–15 In the study by Workeneh et al., an increase in daily fat intake was the only determinant of weight gain after kidney transplantation, mainly at the expense of adipose tissue in the trunk region.12 Therefore, dietary advice could prevent this weight gain; however, an intensive nutritional intervention failed to demonstrate a reduction on weight gain as compared to standard nutritional recommendations.16
Another determinant of the evolution of weight after transplantation in our study was the type of renal replacement therapy prior to transplantation, with a tendency to less weight gain in patients coming from peritoneal dialysis. As already described in other studies, it is believed that a less weight gain may be due to a state of overhydration prior to transplantation in this group of patients; the correction of blood volume after transplantation as renal function improves, as well as due to a different plasma concentration of different hormones related to appetite among patients from both techniques.17,18 Although we observed a trend towards a greater risk of weight gain in female sex, we did not find significant differences in the evolution of weight associated with sex, which is contrary to what has been reported in other studies. Racial differences in the study populations could explain these differences.13,15 Nor did we find any differences associated with the dose of steroids received during the first year. The current tendency to quickly reduce or suspend the dose of steroids derived from an improvement in the current immunosuppressive therapy would be the cause of the loss of influence of steroids on the evolution of weight after transplantation found in old works.6,18,19
The influence of the BMI on the evolution of the graft is not sufficiently clarified. In our study, we found a trend of a higher risk of graft loss in obese patients. We also found a relationship between graft loss and BMI as a continuous variable, or if the patients are separated by cut-off point of BMI value from which the risk of graft loss start to increase, a BMI value that is included in the range of overweight. Studies with a large number of patients find no relationship between obesity and graft loss, or find such a relationship only in old studies without demonstrating this worse evolution in more recent studies, alluding to changes in current immunosuppressive therapy as an explanation for this difference.14,19 But the large majority of studies find a worse evolution of the graft as the BMI increases.10,11,20,21 An increase in BMI is associated with a greater risk of developing CKD, and a more rapid evolution towards terminal CKD.22 This is due not only to the association of obesity with other comorbid factors that also cause CKD, such as DM and hypertension, but also due to the development of histological damage derived from adaptive glomerulomegaly in these patients, which in turn it can be influenced by other factors such as a low nephron load at birth. This glomerular hypertrophy results in the development of hyperfiltration and proteinuria that in these patients has been associated to glomerulosclerosis.23,24 After kidney transplantation hyperfiltration occurs since patients receive only one kidney, a situation of which can be aggravated in recipients with a higher BMI than the donor. Therefore, the BMI must be taken into account in the correct donor-recipient match.
In general, after kidney transplantationthere is a global weight gain, although there are patients that lose weight. This weight gain or loss, with an associated increase or decrease in BMI, would result in a greater or lesser risk of graft loss.14,21 However, in our study weight gain or loss by itself was not related to graft loss, not even considering a gain or loss greater than 5% as reported in some studies,17 although other authors have observed a worse evolution of the graft con greater porcentages of wieight change.14 A patient with weight gain an increase in BMI after the transplant would have a worse evolution of the graft than a patient who loses weight and decreases his BMI. We consider that it is not the weight gain or loss itself that influences graft survival, but the BMI reached one year after the transplant. Thus, a patient who gains weight and evolves from overweight to grade 1 obesity after transplantation has the same risk of losing the graft as another patient who loses weight and changes from grade 2 obesity to grade 1 obesity.
Obesity and DM, comorbid factors of the metabolic syndrome, are associated with the development of cardiovascular disease, both in the general population and in transplant recipients.25–27 Obesity has been associated in various studies, although not in all, with a higher incidence of DM after kidney transplantation and, therefore, with an added risk of impaired kidney function.28 Weight gain after kidney transplantation, mainly due to an increase in adipose tissue, especially abdominal visceral fat, could predispose to the development of insulin resistance, the onset of diabetes, chronic inflammation, and cardiovascular events.12 In our work there were almost 15% of new diabetics per year, but we did not find any relationship with weight gain or loss, nor with BMI. As reported in other studies,29,30 we also found no association between weight gain or loss, or BMI with cardiovascular events after transplantation despite the high percentage of obesity at the end of follow-up, probably due to the different characteristics of the population studied.
There are several limitations of our study. First, it is a single-center study, with a limited sample size and without racial diversity. Therefore, the results may not be extrapolated to another population. Overall weight gain after transplantation is lower than that reported in other studies.6,13 Thus, patients who gain or lose weight may remain in the same range of BMI that they had at the time of transplantation, and therefore the change in weight does not reflect the effect on the evolution of the graft if the BMI is taken as a categorical variable. However, if is the BMI is considered as a continuous variable, the effect is evident. Furthermore, we did not have any patient with a BMI lower than 18 kg/m2 therefore the effect of a BMI lower that such a value on the evolution of the graft or on the development of DM or cardiovascular disease, could not be evaluated.
ConclusionIn our study, although the overall trend after kidney transplantation is towards weight gain, there is a significant proportion of patients who lose it. We did not find lower graft survival according to this weight gain or loss; however we did find and effect of BMI on graft survival. A weight gain, with a consequent increase in BMI, will result in a worse evolution. Despite the frequent association between obesity, DM and the development of cardiovascular disease, in our study the weight gain or loss or the BMI did not significantly influence the development of new diabetes or the development of cardiovascular disease.
FinancingThis research has not received specific support from public agencies, commercial sector or non-profit entities.
Conflict of interestsNone by all authors.