Introducción: Las alteraciones del metabolismo óseo-mineral presentan una alta prevalencia en los pacientes con enfermedad renal crónica (ERC), siendo mayor conforme avanza el estadio de enfermedad. El diagnóstico de dichas alteraciones se basa fundamentalmente en la determinación de niveles de hormona paratiroide (PTH-i). Sin embargo, la determinación de esta hormona no es sencilla y está sometida a gran variabilidad. Los métodos para procesar las muestras de PTH-i no están estandarizados, hecho que podría ser una fuente importante de variabilidad preanalítica. Objetivo: Analizar la variabilidad en los resultados de la determinación de la PTH-i comparando distintas formas de procesar la misma muestra de plasma tratado conácido etilendiaminotetraacético (EDTA) en pacientes con ERC. Material y métodos: Se han analizado 294 muestras, correspondientes a 49 pacientes con ERC, 18 procedentes de la Consulta de Trasplante Renal (36,7%) y 31 del programa de Hemodiálisis Crónica de nuestro Centro (63,3%). Se ha procesado la misma muestra de cada uno de nuestros pacientes de seis maneras distintas, comparando las medias entre el grupo de referencia o gold standard y los otros grupos a estudio. Las muestras se procesaron con diferentes condiciones de temperatura y tiempo antes de ser congeladas, constituyendo seis grupos: centrifugación y congelación inmediata (grupo 1, de referencia); muestra a temperatura ambiente una hora, centrifugación y mantenimiento en nevera (2-8 ºC) durante 0, 8 o 24 horas (grupos 2 A, 2B y 2C, respectivamente); mantenimiento de sangre a temperatura ambiente 3 horas, mantenimiento en nevera (2-8 ºC) durante 0 y 8 horas (grupos 3A y 3B). La PTH-i se ha determinado mediante Inmunoradiometria (IRMA Total Intact Scantibodies assay). Se ha realizado el test de homogeneidad de varianzas y normalidad, y depués comparaciones por pares con el t-test con la corrección de Bonferroni. Resultados: La PTH-intacta media en el grupo de referencia fue 202,5 ± 199,72 pg/ml. Las medias de PTH-intacta en distintos grupos analizados fueron 196 ± 203,23 pg/ml, 202,8 ± 200,2 pg/ml, 200,06 ± 194,87 pg/ml, 204,08 ± 204,073 pg/ml, 197,94 ± 182,31 pg/ml. Los resultados fueron prácticamente superponibles, no encontrando diferencias significativas respecto al grupo de referencia (p = 0,87, p = 0,99, p = 0,95, p = 0,96, p = 0,90 al comparar con grupos 2A, 2B, 2C, 3A y 3B, respectivamente). Conclusiones: La utilización de EDTA como conservante en el procesamiento de las muestras analíticas para la determinación sanguínea de PTH-i permite un mayor tiempo de procesamiento de la misma, sin la exigencia de su congelación inmediata, mostrando una mínima variabilidad en los resultados obtenidos según diferentes formas de procesamiento. Estos resultados pueden ayudar a establecer estrategias logísticas para el procesamiento de muestras sanguíneas en los pacientes con ERC.
Background: The measurement of i-PTH circulating is not easy due to its analytical variablity. Variability that appears in the process that goes from the sample collection to the final result determination. There are several important aspects that can influence within the pre-test variability: type of sample (serum o plasma), temperature, time elapses from blood extraction to freezing and from freezing to i-PTH quantification. Blood coming from centres far from our laboratory do not always meet the required processing conditions. Our aim was to study the stability of i-PTH with varying conditions of temperature and time until freezing in patients with chronic kidney disease (CKD). Method: We have analyzed 294 blood samples of 49 patients with chronic kidney disease (18 transplantated patients (36.7%) and 31 patients in haemodyalisis (63.3%)). The blood samples were collected using tubes treated with ethylenediaminotetraacetic acid (EDTA); these samples were subjected to different conditions of temperature and time before they were frozen, constituting 6 groups: blood centrifuged and plasma immediately frozen (group A or reference group); blood maintained 1 hour at room temperature and plasma stored at 2-8 ºC during 0, 8 and 24 hours (groups B,C,D); blood maintained 3 hours at room temperature and plasma stored at 2-8 ºC during 0 and 8 hours (groups E,F). The intact PTH (i-PTH) was measured using the immunoradiometric assay (IRMA Total Intact Scantibodies assay). We have analyzed the differences between the PTH-i mean values in the referenced groud and the others. We have applied the tests of homogeneity variance and normality and we have perform a comparation by pairs with the t-test including the Bonferroni correction. Results: The mean value of intact- PTH in the referente Group was 202.5±199.72 pg/ml. The means values of intact-PTH in the other groups were 196 ± 203.23 pg/ml, 202.8 ± 200.2 pg/ml, 200.06 ± 194.87 pg/ml, 204.08 ± 204.073 pg/ml, 197.94 ± 182.31 pg/ml. The results were practically identical for each group. We did not find important differences with respect to the reference group (p = 0.87, p = 0,99, p = 0,95, p = 0,96, p = 0,90 when comparing with groups 2a, 2b, 2c, 3a y 3b). Conclusions: The use of EDTA maintain the PTH stability during a longer period without the necessity of freezing the samples immediately. These results can help to state strategies to management the samples in patients with ERC.
INTRODUCTION
Alterations in the calcium-phosphorus metabolism are a prevalent problem and of great importance to patients with CKD (chronic kidney disease).1-8 Appropriate and early evaluation, as well as treatment is vital for optimum control of our patients.
The ideal diagnostic method for the grade and type of renal osteodystrophy is still iliac crest biopsy following tetracycline double labelling,9-25 but, due to its invasive and toilsome nature, it is of difficult application in daily clinical practice.26 At present, diagnosis and treatment of these alterations are based on the determination of plasmatic levels of intact parathyroid hormone (iPTH).10,17,27-31 However, the determination of this hormone is not simple and is subjected to great variability.32-34 In addition to this, methods used to process the iPTH samples are not standardised, a fact which could be an important source of pre-test variability.
AIM OF THE STUDY
To analyse variability in iPTH determination results comparing different methods of processing the same sample of plasma treated with ethylenediaminetetraacetic acid (EDTA) in patients with CKD at different stages of development.
MATERIAL AND METHOD
In our study, we analysed 294 blood samples from 49 patients with chronic kidney disease (CKD) at different stages of development, 18 were from transplanted patients at the Renal Transplant Unit (36.7%) and 31 from the Chronic Haemodialysis Unit at our hospital (63.3%).
All the blood samples were collected in the morning (between 8 and 9 am) after an eight hour fast and predialysis for the patients undergoing substitutive kidney treatment. The blood was drawn at the Haemodialysis Service of our hospital. Three tubes of EDTA were extracted from each patient: two 5ml tubes and one 10ml tube. Immediately after the extraction, they were sent to the refrigerator at the Nuclear Medicine Department, where each was processed differently, as will be described herewith.
Reference group or group 1 (EDTA 5ml tube):
- Blood sample collected (and quickly sent refrigerated to the Nuclear Medicine Department).
- Refrigerated centrifugation.
- Plasma immediately frozen.
Group 2 (EDTA 10ml tube):
- Blood sample collected.
- Maintained one hour at room temperature.
- Normal centrifugation.
- Separated into 3 tubes:
- One of the tubes is immediately frozen. Group 2A
- One tube is refrigerated for eight hours and then frozen. Group 2B
- Another tube is refrigerated for > 24 hours and frozen. Group 2C
Group 3 (EDTA 5ml tube):
- Blood sample collected.
- Maintained > 3 hours at room temperature.
- Normal centrifugation.
- Separated into two tubes:
- Immediately frozen. Group 3A
- One tube is refrigerated for > 8 hours and frozen. Group 3B
The processing methods used for the blood samples in our study were those usually applied in this domain. The iPTH was measured using the immunoradiometric assay (IRMA Total Intact Scantibodies assay, 2nd generation). The withinrun variability obtained was less than 2.5%. Group 1 was considered the gold standard, since these are the generally recommended conditions for this type of samples, independently of the method or preservative used (EDTA or citrate).
A two by two comparison was performed between the iPTH mean values in the reference group and the others. Therefore, the distribution normality was checked first by applying tests of homogeneity variance and normality. Subsequently, we performed a comparison by pairs with the t-test including the Bonferroni correction. The level of significance was established at p < 0.05.
RESULTS
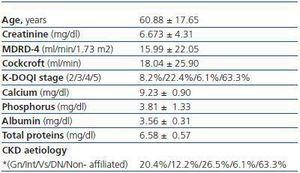
The average age of the population studied was 60.88 ± 17.65 and the age range between 26 and 90 years. Of the 49 patients included in the study, 25 were female and 24 male, which corresponds to 51 and 49%, respectively. The main cause of chronic kidney disease is non-affiliated, followed by vascular and glomerular origin. The remaining descriptive features of our study population are displayed in table 1.
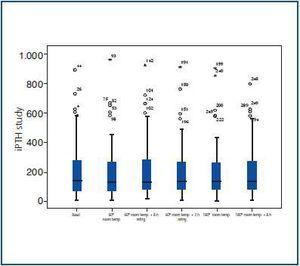
The mean value in the reference group was 202.25 ± 199.72pg/ml. The mean values of intact iPTH in the other groups analysed, displayed in table 2, were 196 ± 203.23pg/ml, 202.8 ± 200.2pg/ml, 200.06 ± 194.87pg/ml, 204.08 ± 204.073pg/ml and 197.94 ± 182.31pg/ml. As can be seen in figure 1, the results were practically identical for each group, with no important differences with respect to the reference group (p = 0.87, p = 0.99, p = 0.95, p = 0.96, p = 0.90), when comparing with groups 2a, 2b, 2c, 3a and 3b, respectively).
DISCUSSION
Parathormone serum determination is difficult to quantify since it is quite unstable both in serum and in the blood43-45 (av. 5 minutes). The reference method is based on refrigerated spinning and freezing the sample immediately after extraction. However, this procedure is very difficult in daily clinical practice, since nephrology activity is not only limited to the hospital domain. In general, samples are drawn at the clinics and then sent to the referral hospitals for analysis. On occasion, these clinics are at quite a distance from the hospitals. Most of the samples are not spun or separated in the place of extraction because they do not have the means required to do so. In addition to this, the samples are spun at room temperature. We can derive from this the importance of knowing and reducing the variability to a maximum in the handling of samples.
At present, there is no standardised method of processing them. There is some controversy on the stability of parathormone in serum or plasma treated with EDTA. Several studies state that the determination of iPTH in serum is less stable than that obtained from plasma and blood sample conserved in tubes treated with EDTA.35-42 However, Omar et al.38 in 2001 observed that the levels of iPTH in EDTA decreased 14.8% after remaining at room temperature for 48 hours. In addition to this, recently, Cavalier et al.42 proved that samples processed with EDTA are not always more stable than in serum. They argue that the stability of the iPTH after 24 hours at -20ºC is greater in serum than for plasma treated with EDTA.
In almost all the studies performed to date, serum and EDTA are compared.35-42 However, one of the limitations which our study presents is that a control group with serum (citrate) was not established. Therefore, we cannot evaluate the determinations of the iPTH in serum and plasma (EDTA) once frozen at -20ºC, nor compare this with the findings of Cavalier et al. In addition to this, we have not analysed what occurs after 48 hours at room temperature in order to compare with the Omar et al. results.
The findings of this study show no significant differences between the reference group and the other groups analysed. It is confirmed that the plasma samples treated with EDTA are stable and its use is recommended with refrigerated centrifugation and immediate freezing is difficult to achieve.
CONCLUSION
The use of EDTA as a preservative in the processing of analytical samples for the blood determination of iPTH allows for greater processing time without the requirement of immediate freezing, displaying a minimal variability in the results obtained according to different methods of processing. These results can help to establish logistical strategies for the processing of blood samples in CKD patients.
Table 1. Descriptive features of the study population
Table 2. Mean iPTH value (pg/ml) for each group studied
Figure 1.