It is recommended that IgA nephropathy (IgAN) is treated with steroids when the glomerular filtration rate (GFR) is >50ml/min and proteinuria >1g/day. Few studies have been performed comparing the two accepted steroid regimens (1g/day methylprednisolone pulses for 3 consecutive days at the beginning of months 1, 3 and 5, followed by 0.5mg/kg prednisolone on alternate days vs. 1mg/kg/day oral prednisolone). The aim of this study was to compare these two steroid regimens in IgAN treatment.
MethodsWe selected 39 patients with biopsy-proven IgAN treated with steroids. Mean age at diagnosis was 37.5 years, 23 males (59%), baseline proteinuria (Uprot) was 2.1 g/day and median serum creatinine (SCr) was 1.5mg/dl. The mean follow-up period was 56 months. Twenty-five patients (64%) were treated with methylprednisolone pulses and 14 (36%) with oral steroids.
ResultsPatients treated with steroid pulses presented lower relapse risk, defined as the reappearance of Uprot >1g/day and an Uprot increase of more than 50% (incidence rate ratio of 0.18, 95% CI 0.02–0.5). The Kaplan–Meier analysis showed longer relapse-free period (p=0.019). This result was confirmed in a multivariate analysis (p=0.026). However, we did not find other differences between the two steroid regimens.
ConclusionsIn comparison to oral steroids, the intravenous pulse regimen was associated with a lower risk of relapse in IgAN, a known independent negative predictor of renal survival. No differences were found regarding the other renal outcomes.
Se recomienda el tratamiento de la nefropatía por IgA (NIgA) con esteroides cuando el índice de filtración glomerular (IFG)>50ml/min y proteinuria >1g/día. Pocos han sido los estudios realizados comparando los 2 esquemas de esteroides aceptados (1g/día de metilprednisolona en pulsos durante 3 días consecutivos en el principio de los meses 1, 3 y 5 seguido de 0,5mg/kg en días alternos de prednisolona vs. 1mg/kg/día de prednisolona oral). El objetivo de este estudio fue comparar estos 2 esquemas de esteroides en el tratamiento de la NIgA.
MétodosFueron seleccionados 39 pacientes con NIgA demostrada por biopsia y tratados con esteroides. La edad media al diagnóstico fue de 37,5 años, 23 varones (59%), proteinuria basal (Uprot) 2,1g/día y la creatinina sérica mediana (SCR) 1,5mg/dl. El periodo medio de seguimiento fue de 56 meses. Veinticinco de los pacientes (64%) fueron tratados con pulsos de metilprednisolona y 14 (36%) con esteroides orales.
ResultadosLos pacientes tratados con pulsos de esteroides presentan menor riesgo de recaída, definido como la reaparición de una Uprot>1g/día y aumento de más del 50% de la Uprot (razón de tasa de incidencia: 0,18; IC 95%: 0,02-0,5) y el Kaplan-Meier mostró período más largo libre de recaída (p=0,019). Este resultado se confirmó en un análisis multivariante (p=0,026). Sin embargo, no se encontraron otras diferencias entre los esquemas de esteroides.
ConclusionesEn comparación con los esteroides orales, el esquema en pulsos intravenosos se relacionó con un menor riesgo de recaída en la NIgA, un conocido predictor negativo independiente de la supervivencia renal. No se encontraron diferencias en cuanto a los otros outcomes renales.
IgA nephropathy (IgAN) is the most frequent primary glomerulonephritis in the developed countries. Regardless of the treatment used, more than 50% of the patients with IgAN have a progressive disease.1 The VALIGA cohort estimated a rate of glomerular filtration ratio (GFR) decline of 1.8±7.5ml/min/year.2 Therefore, it is predictable that 20–40% of these patients develop end-stage renal disease (ESRD) within 20 years.3
Currently there is not a specific treatment for the pathogenic process beyond IgAN. The KDIGO guidelines4 recommend blood pressure control with the use of one renin–angiotensin system inhibitor/blocker in all patients. This approach has the stronger evidence in slowing the disease progression and reducing the proteinuria and the slope of GFR.4,5
Besides these measures, with a significantly lower evidence, this guidelines also suggest the use of corticosteroids for 6 months in those that have proteinuria higher than 1g/day and preserved GFR (GFR>50ml/min) after supportive therapy.4 The treatment with steroids was associated with lower risk of progression to ESRD and lower urinary protein excretion.6
There are two steroid regimens accepted to treat IgAN: the steroid pulse regimen (also known as “Pozzi scheme”, i.e. 1g/day methylprednisolone pulses for 3 consecutive days at the beginning of months 1, 3 and 5 followed by 0.5mg/kg of oral prednisolone in alternate days) and the oral steroid regimen (oral prednisolone 1mg/kg/day for 2 months and then a reduced dose for the next 4 months).4 According to the available studies any of these therapeutic schemes can be used, because both showed to be better than supportive therapy alone.7–9
Currently, we do not have any reason to choose one steroid scheme to treat IgAN instead of the other, as no RCT compared both regimens, so this choice is only based on the beliefs of the physicians and it is not an evidence-based decision. Since the use of pulses scheme is more laborious and expensive it is important to clarify whether its use is associated with better renal outcomes relatively to the simpler and cheaper oral scheme.
The aim of this study was to investigate if there is any advantage of a steroid pulse regimen relatively to oral steroid scheme in the outcomes of the IgAN treatment.
Study designThis is a single center, 9-year retrospective cohort study of patients with biopsy-proven IgAN treated with steroids.
Subjects and methodsWe selected 62 patients with primary IgAN, who had been biopsied between January 2006 and December 2014, from these only 39 were eligible for the study. The inclusion criteria were: having proteinuria greater than 1g/day after at least 6 months of treatment with angiotensin-converting enzyme inhibitors (ACE-I) and/or angiotensin receptor blockers (ARB) before steroids, having received at least 6 months of steroid therapy (pulse or oral regimen), age>18 years at diagnosis and follow-up duration>12 months. All the patients included in the study were under RAAS blockage for at least 6 months. The doses were titrated upward as far as tolerated by each patient.
The exclusion criteria were: pregnancy, chronic advanced liver disease, presence of Henoch–Schonlein purpura or atypical forms of IgAN (acute kidney injury or crescentic IgAN).
Clinical information concerning demographic, clinical and laboratory data were collected from the clinical process. For the purpose of this study all the kidney biopsies were reviewed and classified according to Oxford classification by the same kidney pathologist.
Complete remission was defined as a proteinuria<0.3g/day without worsening of renal function at the end of steroid therapy (6 months). Partial remission was defined as at least a 50% reduction in proteinuria compared with baseline after 6 months of therapy.
Relapse was defined as the recurrence of proteinuria>1g/day and at least a 50% increase in proteinuria compared to the lowest proteinuria achieved after treatment.
Our endpoints were a combined endpoint of doubling serum creatinine (SCr) or ESRD, rate of complete or partial remission, GFR decline rate and relapse rate.
According to the type of steroid regimen used the patients were divided in two groups: pulse steroid group (n=25, 64%) and oral steroid group (n=14, 36%).
The cumulative prednisolone-equivalent steroids dose (in grams) was obtained by multiplying the dose per day (pill or injection) by the prednisolone conversion factors, then multiplying it by the number of days of treatment.
Statistical analysisVariables are expressed as frequencies for categorical variables, mean values with SD for continuous variables and median values with interquartile ranges for ordinal variables.
Comparison between groups was performed using T-test for normally distributed variables, Wilcoxon test for non-normally distributed variables and χ2 test for categorical variables. Survival curves were estimated by Kaplan–Meier analysis and compared by the log-rank test. Cox regression analysis was used for multivariable analysis.
Statistical analysis was performed with SPSS system 21.0 (SPSS Inc., Chicago, IL). For all comparisons, a p<0.05 was considered statistically significant.
ResultsA total of 39 biopsy-proven IgAN patients were included in the study. Their baseline demographic, clinical and laboratory characteristics are summarized in Table 1. Of the studied population 59% were male, mean age was 37.5 years, 94.9% were caucasian and had a median follow-up time of 56 months after treatment.
Baseline characteristics of the studied population.
| Variables | Patients (n=39) | Oral steroids (n=14) | Steroid pulses (n=25) | p |
|---|---|---|---|---|
| Age, years | 37.5±1.8 | 36.7±9.1 | 37.9±12.3 | 0.332 |
| Gender, male | 23 (59.0) | 10 (71.4) | 13 (56.5) | 0.410 |
| Race, black | 2 (5.1) | 1 (7.1) | 1 (4.0) | 0.811 |
| Cumulative prednisolone-equivalent steroids dose, g | 14.1 (6.1–14.4) | 6 (5.4–7.2) | 14.3 (14.1–14.6) | <0.001 |
| Hypertension (HTN) | 18 (46.2) | 5 (35.7) | 13 (52.0) | 0.121 |
| Smoke | 7 (17.9) | 5 (55.6) | 2 (16.7) | 0.102 |
| Proteinuria, g/day | 2.1 (1.5–3.8) | 3.1 (1.1–5.5) | 2.0 (1.6–3.6) | 0.139 |
| GFR, ml/min | 53 (31–74) | 41 (14.3–78.8) | 54 (39.5–71.5) | 0.160 |
| History of macroscopic hematuria | 8 (20.5) | 3 (21.4) | 5 (20) | 0.901 |
| Microscopic hematuria | 32 (82.1) | 10 (71.4) | 22 (88) | 0.408 |
| Serum albumin, g/dl | 3.5±0.6 | 3.4±0.7 | 3.6±0.5 | 0.792 |
| Serum total cholesterol, mg/dl | 220.7±56.0 | 233.4±82.0 | 214.1±36.9 | 0.430 |
| Follow-up, months | 56 (24–104) | 51(12–90.5) | 56 (31–104) | 0.761 |
| Person-time, months | 2578 | 808 | 1770 | <0.001 |
| Histological grade – Oxford classification | ||||
| Mesangial cellularity (M1) | 29 (85.3) | 11 (91.7) | 18 (81.8) | 0.419 |
| Segmental glomerulosclerosis (S1) | 13 (38.2) | 3 (25) | 10 (45.5) | 0.321 |
| Endocapillary hypercellularity (E1) | 19 (55.9) | 9 (75) | 10 (45.5) | 0.157 |
| Tubular atrophy/interstitial fibrosis (T1/T2) | 27 (79.4) | 8 (66.6) | 19 (86.3) | 0.693 |
Values are: mean±SD, median (interquartile range) or frequencies [n (%)].
The baseline characteristics in both groups were similar concerning to age, gender, hypertension prevalence, GFR, urinary protein excretion, micro and macroscopic hematuria and serum albumin (Table 2). Regarding the renal biopsies, we found that the four parameters of the Oxford classification were also similar between groups. The cumulative prednisolone-equivalent steroids dose was significantly higher in the group of pulses regimen (14.3 vs. 6g, p<0.001).
Univariate analysis for the development of the main outcomes and side effects.
| Oral steroids (n=14) | Steroid pulses (n=25) | IRR (confidence interval) | p | |
|---|---|---|---|---|
| Doubled SCr or ESRD | 3 (21.4%) | 8 (32%) | 1.22 | 0.507 |
| Incidence ratea | 3.7 (1.2–11.5) | 4.5 (2.2–9.0) | (0.29–7.12) | |
| GFR decline rate, ml/min/year | −4.0±6.3 | −4.5±8.0 | 0.729 | |
| GFR at the end of the follow-up, ml/min | 54.2 (34.5–97.6) | 45.7 (26.9–66.3) | 0.430 | |
| Complete or parcial remission | 9 (75%) | 20 (80%) | 0.810 | |
| Relapse | 5 (35.7%) | 2 (8.0%) | 0.18 | 0.044 |
| Incidence ratea | 6.2 (2.6–14.9) | 1.1 (0.3–4.5) | (0.018–0.85) | |
| Microscopic hematuria at the end of the follow-up | 5 (35.7) | 5 (20) | 0.46 | 0.231 |
| 2.8 (1.25–7.1) | 6.1 (3.01–13.2) | (0.11–1.98) | ||
| Median proteinuria, g/day | ||||
| At 6 months (end of treatment) | 0.3 (0.1–1.2) | 0.4 (0.2–1.2) | 0.903 | |
| At 12 months | 0.2 (0.1–0.3) | 0.4 (0.1–0-9) | 0.894 | |
| At 36 months | 0.2 (0.1–0.4) | 0.5 (0.1–1.1) | 0.920 | |
| At 60 months | 0.3 (0.2–0.6) | 0.3 (0.1–1.4) | 0.997 | |
| Proteinuria<0.3g/day | ||||
| At 6 months (end of treatment) | 7 (50%) | 9 (36%) | 0.410 | |
| At 12 months | 8 (80%) | 10 (47.6%) | 0.120 | |
| At 36 months | 5 (71.4%) | 7 (38.9%) | 0.328 | |
| At 60 months | 4 (66.7%) | 5 (45.5%) | 0.548 | |
| Side effects of steroids | ||||
| De novo or worsening diabetes | 2 (14.3%) | 2 (8%) | ||
| Incidence ratea | 0.003 (0.0006–0.009) | 0.001 (0.003–0.005) | 0.46 (0.03–6.3) | 0.790 |
| Cushingoid state | 1 (7.1%) | 2 (8%) | ||
| Incidence ratea | 0.006 (0.0008–0.004) | 0.0025 (0.0006–0.009) | 0.23 (0.004–4.4) | 0.510 |
HR, hazard ratio; IRR, incidence rate ratio.
Values are: mean±SD, median (interquartile range) or frequencies [n (%)].
During the follow-up period, 8 patients in the group of steroid pulse regimen and 3 in the oral prednisolone regimen reached the combined outcome of doubling SCr or ESRD, however this difference was not statistically significant (IRR 1.22, 95% CI 0.29–7.12). The mean GFR decline rate was also not different between groups (steroid pulse – 4.5ml/min/year vs. steroid oral scheme – 4.0ml/min/year).
In both groups, 75% or more of the total patients achieved partial or complete remission. On the other hand, the patients treated with steroid pulses showed lower relapse rate (incidence rate 1.1 vs. 6.2 relapses/person-months and an incidence rate ratio of 0.18, 95% CI 0.02–0.85).
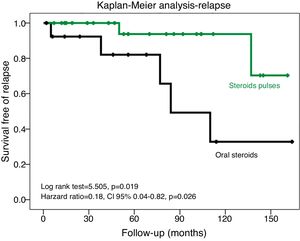
Kaplan–Meier analysis showed that the patients treated with steroid pulse regimen had a longer period of time free to relapse (Log Rank test=5.505, p=0.019) (Fig. 1). Cox regression analysis confirmed this result in a model adjusted to age and baseline proteinuria (HR=0.18, 95% CI 0.04–0.82, p=0.026) (Table 3).
The median urinary protein excretion decreased in both groups soon after the treatment (at 6 months of follow-up), and both groups maintained a similar urinary protein excretion. Similarly, the proportion of patients that achieved proteinuria lower than 0.3g/day were comparable between the two groups during the follow-up.
The survival analysis did not find steroid pulses as a significant predictor for the other analyzed endpoints such as achieving complete or partial remission.
Regardless of the higher cumulative prednisolone-equivalent steroids dose, the pulses regimen did not have higher incidence of steroids side effects. There was not an increased incidence of diabetes, cushingoid state or infections severe enough to require special medical care, such as antibiotic prescription (no cases reported), during the follow-up (Table 2).
DiscussionIn our cohort of patients, the IgAN treatment with steroid pulse regimen was associated with a lower risk of relapses. However, no significant differences were found regarding the main renal outcomes, such as development of ESRD, eGFR decline or remission rate.
It is well studied that the relapse rate was an independent negative predictor of renal survival in patients with IgAN.10 In our study, the lower rate of relapses in the pulses steroid group was not associated with a slower progression of the disease, evaluated by eGFR decline rate or incidence of ESRD. IgA nephropathy is a glomerular disease with a slow progression to ESRD. Different studies predicted that less than 50% of the patients with IgAN will progress to ESRD in 20–25 years.3 We believe that in order to evaluate long-term outcomes, such as evolution to ESRD in IgAN, our cohort with a median follow-up period of 56 months (4.6 years) is short. This can, at least in part, explain why we did not found differences between groups regarding ESRD development.
We found no differences between the two regimens regarding to proteinuria reduction, remission achievement after treatment or eGFR decline rate.
Only one study11 with 16 patients compared these two regimens for the treatment of IgAN and found no significant benefits of one scheme relatively to the other. However that study has important limitations such as the fact that baseline proteinuria was significantly different between groups and the criteria used to treat IgAN patients with steroids (proteinuria<500mg/day and SCr<1.5mg/dl) were different from the current guidelines.4
Two main reasons have been presented to use high-dose intravenous steroids followed by a moderate oral dose instead of oral steroids alone in the treatment of many kidney and non-kidney diseases: firstly, the earlier and more effective response, and secondly, the lower toxicity.12 The administration of steroid pulse therapy achieves quicker and stronger immunosuppressive and anti-inflammatory effects, decreasing the needed dose of oral steroids in the maintenance therapy (which is associated to the most relevant long-term adverse effects of the steroids, such as Cushing syndrome, diabetes, osteoporosis, etc.) and avoiding the morbidity associated with prolonged nephrotic syndrome.13 This paradox, in which the administration of high-dose intravenous steroids is used to reduce the steroid-related side effects, is based on studies where this regimen is as effective as the oral steroids in full-dose, but less toxic, in patients with idiopathic nephrotic syndromes.14,15 On the other hand, some clinical observations suggest that the immunosuppressive effects of high dose of IV glucocorticoids occur too fast to be explained only by the classic mechanism (genomic) of action, suggesting that this high dose administration of steroids has different pharmacologic effects than those produced with lower doses.16 It was mainly observed in children with steroid-resistant nephrotic syndrome, in which the administration of IV pulses of steroids lead to remission in some cases.13,15 Nowadays there is some evidence that the cellular effects of steroids are mediated by genomic and nongenomic mechanisms.5,17
Coppo,18 reviewed the long-term beneficial or legacy effect of IgAN treatment. “Legacy effect” is a term used to describe the use of a treatment in an early phase of a disease that can produce benefits long after the cessation of the intervention. Some experiments suggests that it is possible to expect a legacy effect of early treatments in IgAN, namely in younger patients, with active and reversible pathology lesion, with normal renal function.18 In IgAN, can we expect a superior legacy effect using high intravenous steroid dose, which can produce genomic and non-genomic effects?
In opposition to the Therapeutic Evaluation of Steroids in IgA Nephropathy (TESTING) trial results, we did not have major steroid side effects, even in the group with IV pulses. Patients treated with pulses regimen were exposed to a higher dose of steroids than those treated with oral prednisolone, however we did not find higher incidence of steroids side-effects in these patients, such as diabetes or infections. Likewise, the other study that compared the two regimens did not find differences regarding side-effects.11 In the literature we found studies evaluating similar steroid regimens in other diseases which report equal or even less adverse effects19–21 and other studies reporting more adverse effects22 with high-dose pulse steroids.
The recent results of Supportive versus immunosuppressive therapy of progressive IgA nephropathy (STOP) Trial23 and the preliminary data from the TESTING Study5 forced us to reflect again on the benefits in renal survival and adverse effects of steroids in the treatment of IgAN. However, while waiting for better designed and longer follow-up RCT there is considerable cumulated evidence to support treating these patients with ACE-I or ARB for at least 6 months and only consider 6-month steroids therapy to try to delay the progression of the disease, when proteinuria higher than 1g/day persists.5
Our study has some limitations. It is a retrospective study, so the choice between the regimens was made based on the physician's opinion, and we do not know the reasons why they choose one scheme over another. We also have a small cohort of patients and the follow-up period is too short to evaluate long-term outcomes.
ConclusionsSteroid pulses compared to oral pulse regimen can be associated with a lower risk of relapse in IgAN, a known independent negative predictor of renal outcomes. However, this regimen did not show advantage in other evaluated renal outcomes, such as GFR decline rate, risk of doubling SCr or ESRD, remission rate, proteinuria range or side effects. Randomized controlled studies are needed to confirm this results and analyze if this potential benefit can improve the long-term outcomes.
Conflict of interestThe authors declare no conflict of interest.