Relapsing polychondritis (RP) is a rare autoimmune condition of unknown aetiology which is characterised by recurrent inflammatory flare-ups of cartilage structures mainly affecting the nasal, auricular and laryngotracheal cartilage. Renal involvement is unusual and, when it does present, it is associated with poorer survival.1 Below we describe a case of a patient with RP and associated glomerular disease.
A 39-year-old male, who was admitted to hospital in June 1986 due to fever and symmetric migratory polyarthritis of large and small joints, conductive hearing loss secondary to seromucinous otitis media and conjunctivitis. During the admission, he presented with bilateral auricular chondritis with erythema and pain on palpitation. Biopsy of the auricular cartilage was performed, which confirmed the diagnosis of RP.
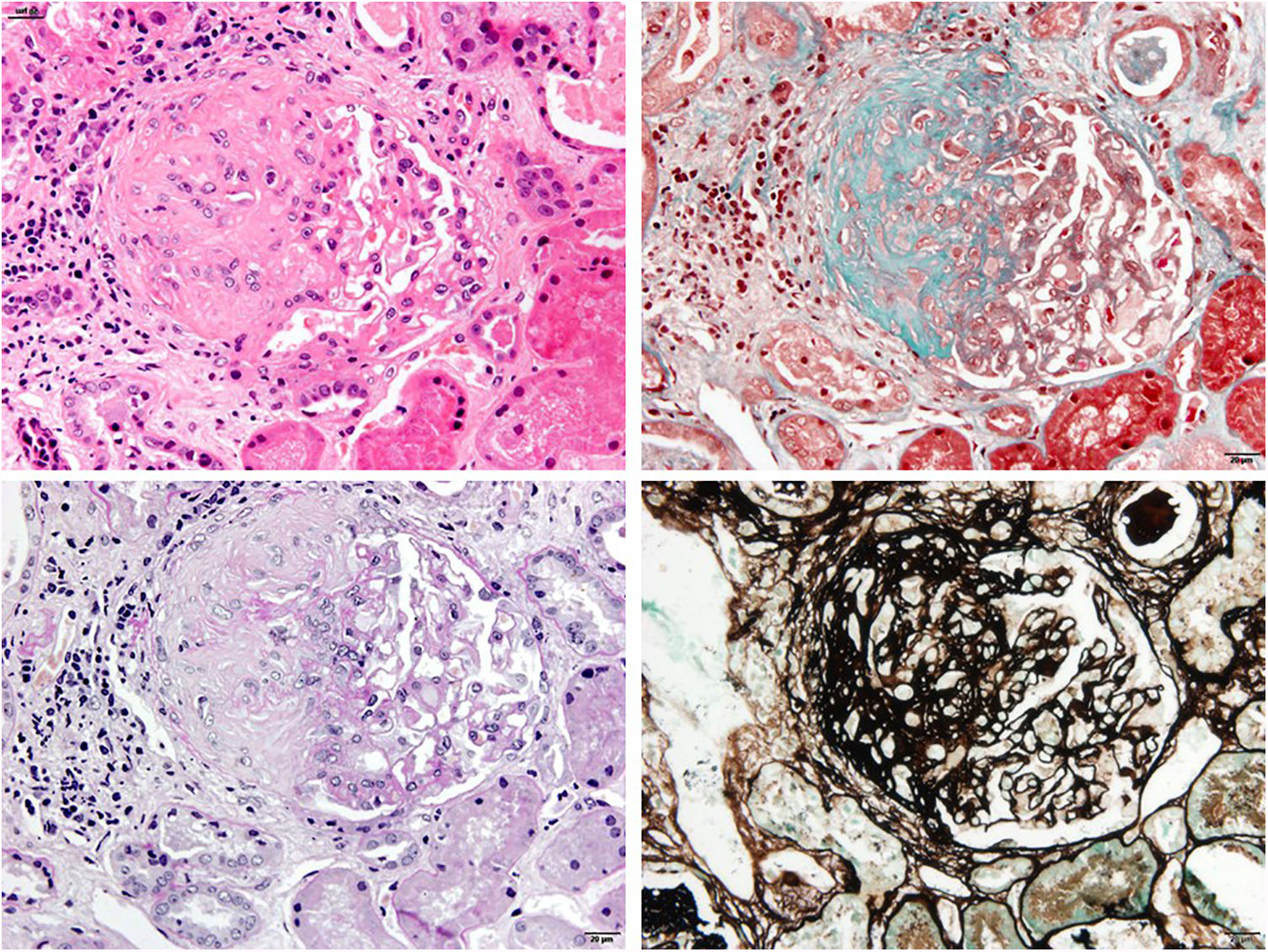
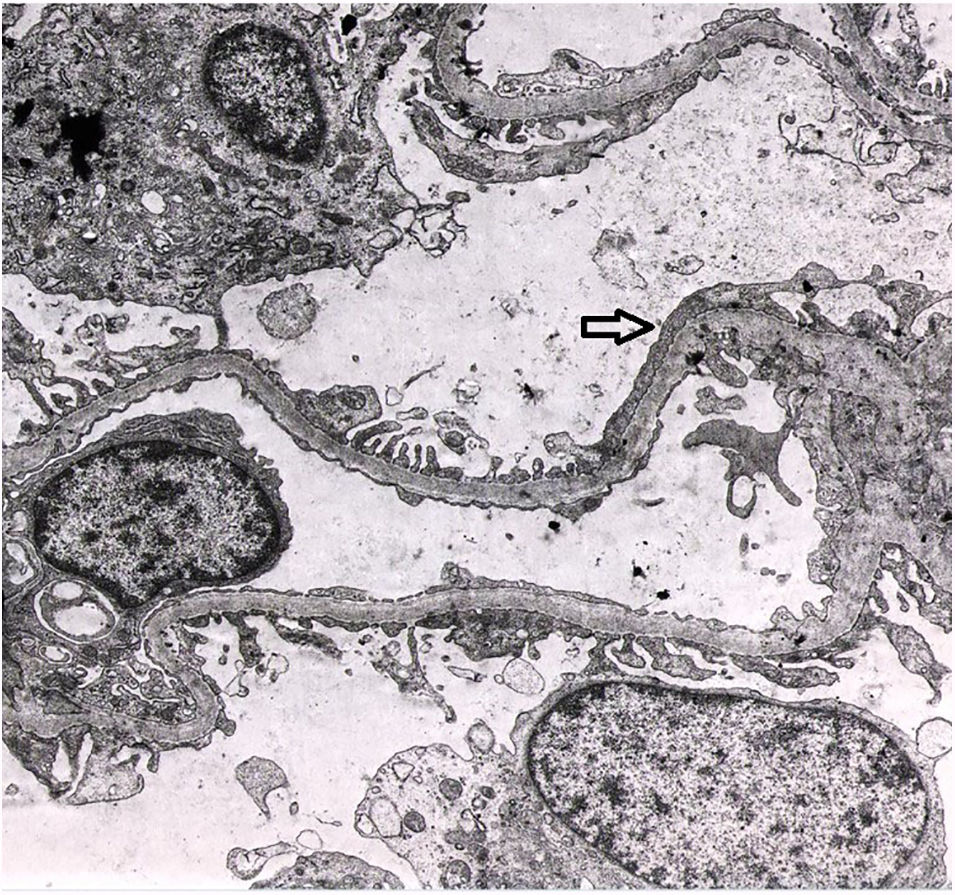
In the laboratory tests, the patient presented mild normocytic anaemia, leucocytosis, thrombocytosis and elevation of acute-phase reactants (C-reactive protein 22mg/dl), with sterile blood cultures and urine culture. Kidney function was normal, with proteinuria of 0.5g/24h and microscopic haematuria. Antinuclear antibodies and anti-DNA and the rheumatoid factor were negative, complement (C3 and C4) values were in the normal range on several occasions. Kidney function was maintained within normal limits, but the patient presented proteinuria of 2.9g/24h and abnormalities in the urinary sediment which included microscopic haematuria and leucocyturia. This is why a kidney biopsy was performed. Optical microscopy showed findings compatible with focal segmental hyalinosis with periglomerular sclerosis (Fig. 1). Segmental IgM deposits were revealed using the immunofluorescence technique, and an irregular podocyte effacement was observed using electron microscopy (Fig. 2). Treatment was started with prednisone at 1mg/kg weight and captopril at maximum doses, with a favourable clinical course of the patient's polychondritis and renal response with reduction of proteinuria to 0.4g/24h and disappearance of microscopic haematuria. After discharge from Nephrology, steroids were maintained for six months due to his RP and steroid treatment was subsequently received on request for short periods, according to the joint inflammation symptoms. During his clinical course, at points the patient tested positive for cytoplasmic antineutrophil cytoplasmic antibodies (c-ANCA) against serine proteinase-3 (PR3) at low titres, but without presenting clinical data of vasculitis or other extrarenal manifestations. Anti-myeloperoxidase (MPO) antibodies and cryoglobulins were negative. After 32 years of evolution of his condition, the patient has presented acceptable progress of his kidney function (serum creatinine of 1.4mg/dl and proteinuria of 0.6g/24h) on treatment with renin–angiotensin axis blocking drugs.
RP is a rare autoimmune disease which, in addition to compromising cartilage structures, also affects non-cartilage structures with a high content of glycosaminoglycans, such as the lungs, heart, eyes and blood vessels. Renal involvement in the clinical course of RP is very rare and it was Neil et al., in 1978, who reported the first cases of glomerular disease associated with RP.2 In the literature review, the prevalence of renal involvement in RP is highly variable. It is possible to find historical series such as that of the Mayo Clinic, with a prevalence of 22% renal involvement according to clinical criteria (presence of haematuria or proteinuria) or histological criteria (with the most prevalent lesions being mesangial nephropathy and segmental necrotising and crescentic glomerulonephritis).1 However, more recent studies report a significantly lower percentage than that reported previously. In a German series, which included 62 patients with RP, only 6.5% had associated renal damage,3 and in another large Asian study which evaluated the clinical and prognostic characteristics of 158 patients with RP, the prevalence of renal involvement was only 3%.4 In a recent review, Dion et al. analysed 142 patients with RP and no patient presented renal involvement.5
Although most of the wide range of histological lesions associated with RP which have been reported up to now correspond with segmental necrotising and crescentic glomerulonephritis,6–8 other types of lesions have also been reported, such as mesangial IgA nephropathy, minimal change disease and membranous glomerulonephritis.6,9 There is considerable lack of knowledge with regard to the aetiopathogenesis of renal damage associated with RP, with a possible autoimmune mechanism gaining increasing importance. For this reason, treatment is empirical and is based on the use of corticosteroids or other immunosuppressants, according to the severity of the disease. In cases of greater renal aggressiveness (necrotising and crescentic glomerulonephritis), the use of cyclophosphamide and plasmapheresis, in addition to corticosteroids, has been reported.7,10
The presence of ANCA has been reported in a percentage of cases of RP; however, there is diverging information with regard to the aetiopathogenic role of these in the association with vasculitis. Papo et al., in a series of 23 patients with RP, reported ANCA positivity by immunofluorescence in 30% of the cases (three cases were c-ANCA and five were p-ANCA); however, when analysing these same patients with another more specific technique (ELISA), none of those who had c-ANCA were PR3-positive and four of the five p-ANCA patients were MPO-positive.11 Therefore, although ANCA can be detected in RP, its relevance requires an additional analysis in the clinical context of each patient.
Unlike other reported cases, the clinical course of the kidney disease in our patient has been very favourable, with the administration of corticosteroids and renin–angiotensin axis blocking drugs also coinciding with long periods of remission of his RP.
Please cite this article as: Canllavi E, Alonso M, Fernández M, Gutiérreza E, Morales E. Policondritis recidivante y glomeruloesclerosis segmentaria y focal: coincidencia o causalidad. Nefrologia. 2020;40:360–362.