Introduction: A number of studies have reported an alarming increase in the incidence of obesity related-glomerulopathy. Focal segmental glomerulosclerosis has recently been shown to be key lesion leading to end-stage renal disease in these cases. Methods: Nineteen patients with obesity-related focal segmental glomerulosclerosis (O-FSGS) and 16 with idiopathic focal segmental glomerulosclerosis (I-FSGS) were examined by percutaneous renal biopsy. Glomerular and interstitial parameters in O-FSGS and I-FSGS were compared morphometrically as well as immunomorphological study was done to evaluate immunohistochemical profile of TGFbeta- 1, monocytes/macrophages, T lymphocytes and a-SMA in these groups. Results: The total glomerular cells, mesangial areas, glomerular a-SMA staining, glomerular monocytes/macrophages and glomerular T-cells were in IFSGS increased in comparison with O-FSGS, most of them significantly, whereas total glomerular area was significantly greater in O-FSGS patients. The tubular immunoexpression of TGF-b-1, interstitial volume and interstitial a-SMA staining were significantly increased in I-FSGS cases in comparison with O-FSGS group, but interstitial CD68+ and CD3+ cells did not differ significantly. Moreover, we found in both O-FSGS and I-FSGS groups significant positive correlations between glomerular immunoexpression of a-SMA and glomerular CD 68+ cells, tubular immunoexpression of TGF-b-1 and interstitial immunoexpression of a-SMA as well as tubular immunoexpression of TGF-b-1 and interstitial volume. Negative correlation between tubular immunoexpression of TGF-b-1 and interstitial CD3+ cells was significant only in IFSGS group. Conclusions: Our morphometric and immunohistochemical study suggests that O-FSGS and I-FSGS are separate morphological entities whereas mechanisms of glomerular and interstitial injury in these cases seem to be rather similar.
Introduction
Focal segmental glomerulosclerosis (FSGS) is the histological lesion found in 10-20% of proteinuria or nephrotic syndrome in children and 25% in white adults1. In addition to proteinuria, microscopic hematuria, hypertension and renal insufficiency are common features at presentation1. Cases of FSGS in which renal function is stable for several years are occasionally observed, but it generally shows a more or less rapid decline toward chronic renal failure2,3. However, exact information about the mechanisms involved in the clinical course of the disease is still lacking and the precise etiology and pathogenesis of FSGS is not known, as well4. Familial forms of FSGS suggested a genetic role in the pathogenesis of FSGS. Recently, studies of familial FSGS have demonstrated mutations in slit diaphragm and podocyte proteins that are critical in forming and maintaining the glomerular filtration barrier5.
Although FSGS is predominantly idiopathic it may be also secondary to certain diseases like heroin-associated nephropathy or reflux nephropathy, HIV infection1 or elevated muscle mass6 with a presentation indistinguishable from idiopathic FSGS (I-FSGS). Moreover, in the past three decades FSGS was commonly regarded as a form of obesity-related glomerulopathy (O-FSGS) 7. The prevalence of obesity all over the world has been steadily rising in consequence to increases in dietary intake and sedentary lifestyle8. Mechanisms of kidney damage in obesity include glomerular hyperfiltration, renal remodeling and extracellular matrix proliferation likely involve neurohumoral factors, local growth factors and cytokines9. Especially, the role of TGF-beta-1, monocytes/macrophages, T lymphocytes and myofibroblasts is stressed10-13. Furthermore, the fundamental study of D’Agati group on obesity-related glomerulopathy14 suggested that this entity differs from I-FSGS in some clinical and histological parameters. Therefore, the present study was undertaken to compare morphometric glomerular and interstitial parameters in O-FSGS and I-FSGS as well as to evaluate immunohistochemical profile of TGF-beta-1, monocytes/macrophages, T lymphocytes and a-SMA in these groups.
Material and methods
Patients
Nineteen patients with O-FSGS and 16 with I-FSGS were examined by percutaneous renal biopsy. All biopsies had been performed solely for diagnostic purposes. All of our patients were adults: the mean age in O-FSGS group was 34±11.8 years (12 males and 7 females) and 36.2±10.6 years (10 males and 6 females) in I-FSGS group. Obesity was defined as BMI> 30 kg/m2. Renal biopsies from patients with secondary FSGS other than O-FSGS and with diabetic nephropathy were carefully excluded.
Clinical and laboratory findings at the time of biopsy in cases with O-FSGS and I-FSGS are summarized in Table I. At the time of renal biopsy, a high percentage of patients in both groups showed nephrotic syndrome or heavy proteinuria. Clinical renal impairment (serum creatinine greater than 1.5 mg/dl) was noted in 4 O-FSGS patients and in 6 I-FSGS patients. Elevated blood pressure was observed in 10 O-FSGS and 5 I-FSGS cases. Hematuria accompanied proteinuria in 3 O-FSGS and 6 I-FSGS patients.
In all cases, diagnosis of FSGS was based on characteristic findings by light microscopy (sections stained with hematoxylin and eosin, Masson-Trichrome, Jones' silver impregnation and periodic acid-Schiff followed by Alcian Blue) as well as electron-microscopy and immunofluorescence using standard protocols. Thickness of each section was controlled according to the method described by Weibel15.
Immunohistochemistry
Paraffin sections were mounted onto superfrost slides, deparaffinized, then (for TGF-β-1, α-SMA and CD68 only) treated in a microwave oven in a solution of citrate buffer, pH 6.0 for 20 min and transferred to distilled water. Endogenous peroxidase activity was blocked by 3% hydrogen peroxide in distilled water for 5 min, and then sections were rinsed with Tris-buffered saline (TBS, DakoCytomation, Denmark) and incubated with: polyclonal goat-anti-human TGF-β-1 antibody (Santa Cruz Lab., dilution 1:200), α-SMA (clone P1b5, DakoCytomation, Denmark, dilution 1:50), monoclonal mouse anti-human CD3 T cell antibody (Clone PC3/188A, DakoCytomation, Denmark, dilution 1:50) and monoclonal mouse anti-human CD68 antibody (DakoCytomation, Denmark, dilution 1:100). Afterwards LSAB+/HRP Universal kit (DakoCytomation, Denmark) prepared according to the instruction of the manufacturer was used. Visualisation was performed by incubating the sections in a solution of 0.5 mg/ml 3,3'-diaminobenzidine (DakoCytomation, Denmark), in Tris-HCl buffer, pH 7.6, containing 0.02% hydrogen peroxide, for 10 min. After washing, the sections were counter-stained with hematoxylin and coverslipped. For each antibody and for each sample a negative control was processed in parallel by incubation in the absence of the primary antibody and always yielded negative results. In each specimen staining intensity of TGF-β-1 in renal tubules was recorded semiquantitatively by two independent observers in 10 adjacent high power fields and graded from 0 (staining not detectable), 1 (weak immunostaining), 2 (moderate immunostaining intensity) and 3 (strong staining). The mean grade was calculated by averaging grades assigned by the two observers and approximating the arithmetical mean to the nearest unity.
Morphometry
Only non-sclerotic glomeruli were examined. Morphometry was performed by means of image analysis system consisting of a PC computer equipped with a Pentagram graphic tablet, Indeo Fast card (frame grabber, true-color, real-time), produced by Indeo (Taiwan), and color TV camera Panasonic (Japan) coupled with Carl Zeiss microscope (Germany). This system was programmed (MultiScan 8.08 software, produced by Computer Scanning Systems, Poland) to calculate:
-the surface area of a structure whose perimeter was traced
-the number of objects (automatic function with manual correction)
-the surface area of a structure using stereological net.
All glomeruli in PAS-alcian blue stained sections, except those that were sclerotic or evidently tangentially cut were measured. As a tangentially section was defined one in which the apparent diameter was < 50% of the maximum diameter. The exclusion of tangentially cut glomeruli reduces the yield for analysis by < 15%.16 The coloured microscopic images were saved serially in the memory of a computer, and then quantitative examinations had been carried out. The quantitative examination included the following glomerular parameters:
- Total glomerular area (the inner limit of Bowman’s capsule was traced-semiautomatic function.)
- Total glomerular nuclei per total glomerular area: mesangial, endothelial and visceral epithelial nuclei (these objects were automatically counted and followed out with manual correction, as needed.) The same method was used for counting glomerular CD3+ and CD68+ cells per glomerular cross-section.
- Mesangial area per cent of total glomerular area (in PASalcian blue staining) and a-SMAstaining per cent of total glomerular area. These parameters were measured using point counting method which is an adaptation of the principles of Weibel.15 The point spacing being 16μm. Total number of the points of a net was 169, and total area was 36864 sq. μm. The percentage of a-SMA staining and mesangial area was an expression of the number of points overlying these structures as a percentage of the total points counted.
The interstitial expression of a-SMA was measured as a surface fraction using point counting method, as well. Under the net described above 10 randomly selected adjacent fields of the renal cortex were investigated. Glomeruli and large blood vessels were neglected. As most of the α-SMA immunostaining was within cytoplasmic processes, these structures were included in calculation. The α-SMA-positive staining was expressed as the percentage of points overlying α-SMA-positive areas. The same method was used to estimate interstitial volume in sections stained with Masson trichrome: it was expressed as the percentage of points overlying renal cortical interstitium.
Interstitial T lymphocytes and monocytes/macrophages were determined by counting CD3+ as well as CD68+ cells (semiautomatic function) in a sequence of ten consecutive computer images of 400 x high power fields -0.0047mm2 each. The only adjustments of the field were made to avoid glomeruli and large vessels. The results were expressed as the mean number of CD3 and CD68 immunopositive cells per mm2.
STATISTICAL METHODS
Differences between groups were tested using unpaired Student’s t-test preceded by evaluation of normality and Levene’s test. The Mann-Whitney U test was used where appropriate. Correlation coefficients were calculated using Spearman’s method. Results were deemed statistically significant if p < 0.05
RESULTS
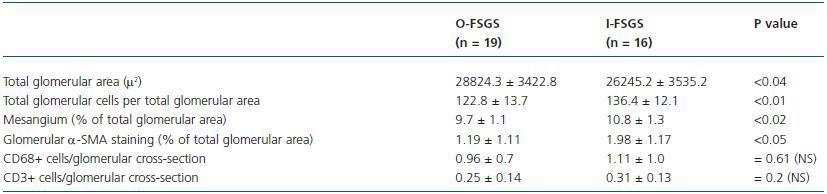
The morphometric data of the glomerular parameters appear from table 2. The mean values of total glomerular cells per total glomerular area, mesangium (% of total glomerular area), glomerular a-SMA staining, glomerular monocytes/macrophages and glomerular T-cells were in IFSGS increased in comparison with O-FSGS, most of them significantly, whereas mean value of total glomerular area was significantly greater in O-FSGS patients.
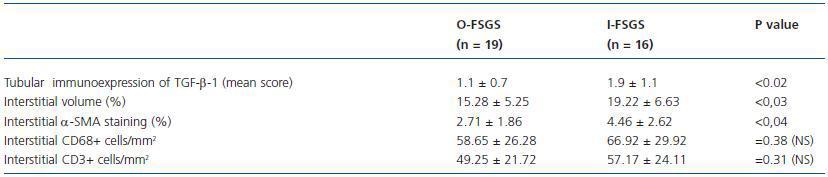
The semiquantitative data concerning the immunoexpression of TGF-β-1 in renal tubules and morphometric data on the interstitial volume, interstitial α-SMA staining and interstitial CD68+ cells as well as CD3+ cells are presented in table 3.
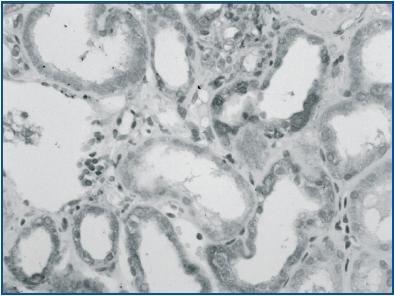
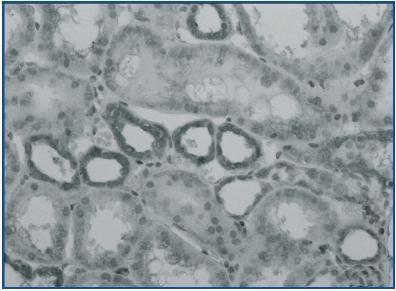
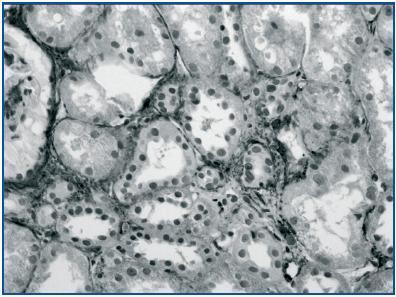
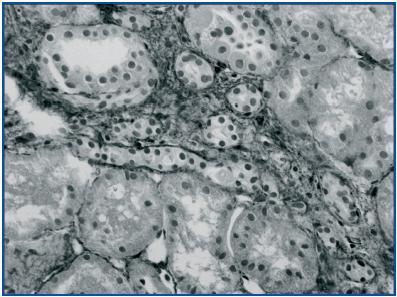
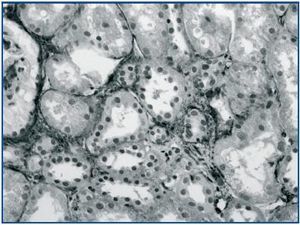
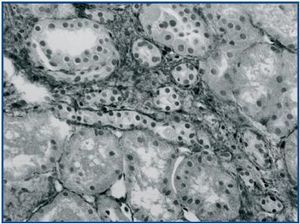
In renal biopsy specimens obtained from patients with OFSGS and I-FSGS TGF-β-1 was detected in the renal tubular epithelial cells (figures 1 and 2.) In some sections, weak immunoexpression of TGF-β-1 was detected in isolated cells in the interstitial inflammatory infiltrates. These cells were excluded from analysis. In both O-FSGS and I-FSGS groups TGF-β-1 expression was absent from glomerular areas.
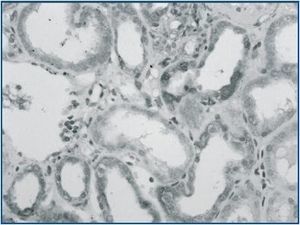
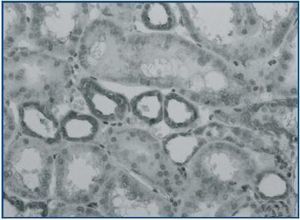
The mean values of the immunoexpression of TGF-β-1, interstitial volume and α-SMA staining (figures 3 and 4) were significantly increased in I-FSGS patients in comparison with O-FSGS group. The mean values of the interstitial CD68+ cells and CD 3+ cells were also increased in I-FSGS patients, however these differences were not significant.
The correlations between selected glomerular and interstitial parameters in patients with O-FSGS and I-FSGS are shown in table 4. In both O-FSGS and I-FSGS groups significant positive correlation existed between glomerular immunoexpression of α-SMA and glomerular CD68+ cells. Moreover, tubular immunoexpression of TGF-β-1 and interstitial immunoexpression of α-SMA as well as tubular immunoexpression of TGF-β-1 and interstitial volume were also in these groups positively and significantly correlated whereas negative correlation between tubular immunoexpression of TGF-β-1 and interstitial CD3+ cells was significant only in I-FSGS group. The correlations between glomerular immunoexpression of α-SMA and glomerular CD 3+ cells as well as between tubular immunoexpression of TGF-β-1 and interstitial CD68+ cells were week and not significant.
DISCUSSION
In 1974 an association between massive obesity and severe proteinuria was reported for the first time.17 Since then, a number of studies have reported an alarming increase in the incidence of obesity related-glomerulopathy and pointed that obesity is a significant risk factor for the appearance of endstage renal disease.18-22 Focal segmental glomerulosclerosis has recently been shown to be key lesion leading to end-stage renal disease in these cases.7 Although some clinical and morphological differences between O-FSGS and I-FSGS were recently reported,14 the present study is to our knowledge the first morphometric and immunohistochemical comparison of these glomerulopathies.
As might be expected from definition, our morphometric study on glomerular parameters confirmed earlier findings of Praga et al.7 that glomerular area in O-FSGS group was significantly increased in comparison with I-FSGS patients. Similarly, in the paper of Kambham et al.14 the incidence of glomerulomegaly was significantly higher in obesity-related glomerulopathy versus I-FSGS. It must be noted, however, that in this study obesity-related group included also patients with glomerulomegaly alone. The pathophisiology of obesity-related glomerulomegaly is up to now not completely understood.14 Probably both an increased renal plasma flow and elevated glomerular filtration rate play a role in these cases.23 Moreover, in the present study in I-FSGS group total glomerular cells, mesangial areas and a-SMA staining were significantly increased as compared to O-FSGS. Therefore, these findings are in concordance with observations of Adelman et al.24 and Kambham et al.14 who found glomerular changes to be less prominent in O-FSGS. Our results are also consistent with prior suggestions, that a-SMA synthesis in mesangial cells is frequently associated with increased cell proliferation. These phenotypic changes may be an indicator of mesangial cells activation after injury and may have important pathophysiologic consequences.10,25,26 Although the number of glomerular CD68+ and CD3+ cells in both groups investigated did not differ significantly, we found in these groups significant positive correlation between glomerular α-SMA staining and CD68+ but not CD3+ cells. This observation raises the possibility that monocytes/macrophages play a role in phenotypic changes of the mesangial cells, however we are aware that a morphometric analysis does not lend itself to establish such casual associations. It is noteworthy that in our study glomerular staining for TGF-β-1 was completely negative, whereas Wolf et al.27 found TGF-β-1 in glomerular endothelial cells of the rat, but these results were received in vitro on cultured cells and could not be transferred directly into human pathology. Glomerular immunoexpression of TGF-β-1 was also noted in some patients with various glomerulopathies, but not in control cases.10
As regard renal interstitial volume, we found it in I-FSGS to be significantly increased as compared with O-FSGS.
In study of Kambham et al.14 on obesity related glomerulopathy and I-FSGS the severity of tubular atrophy and interstitial fibrosis was not statistically different. Probably this difference depends on fact that, as was mentioned above, the obesity-related group included in this study also patients with glomerulomegaly alone. Moreover, interstitial fibrosis in cited paper was assessed only semiquantitatively. Interestingly, in the present study, also the tubular immunoexpression of TGF-β-1 in I-FSGS group was significantly greater than in O-FSGS patients. Furthermore, in both O-FSGS and I-FSGS groups there were significant positive correlations between the immunoexpression of TGF-β-1 and interstitial volume. These observations may suggest that TGF-β-1 is actively involved in the pathogenesis of renal scarring in these nephropathies. Similarly, the study of Goumenos et al.10 which included 9 cases of FSGS showed that tubulointerstitial immunoexpression of TGF-β-1 was related to the degree of interstitial fibrosis and renal function impairment. Our results support also observations of these authors that this pathway is probably common to various renal diseases as the severity of TGF-β-1 immunoexpression is related rather to the degree of renal damage than to the type of renal injury10. Moreover, recently the role of leptin, a small peptide hormone in activation of TGF-β-1 system in obese patients is also taken into consideration.12,19,27
Consequently, our morphometric study showed that interstitial expression of α-SMA was in I-FSGS patients significantly increased as compared with O-FSGS group. We observed interstitial staining for α-SMA in a distribution comparable to that of connective interstitial tissue. In addition, in both O-FSGS and I-FSGS groups strong positive correlations existed between interstitial immunoexpression of TGF-β-1 and a-SMA. It has been demonstrated that cytokines such as TGF-β-1 released by tubular cells and macrophages,28 which plays a key role in the induction of fibrosis, may induce the myofibroblast phenotype in resting fibroblasts or trans-differentiation of tubular epithelial cells.11,29 However, we did not find significant positive correlation between immunoexpression of TGF-β-1 and interstitial CD68+ cells. This observation supports point of view of Nishida et al.30 who suggest that the role of monocytes/macrophages in this process may be very complex. These authors presented evidence that infiltrating monocytes/macrophages in renal tissue may play a beneficial antifibrotic role that surprisingly requires the action of angiotensin.
Although the interstitial CD3+ cells did not differ significantly in O-FSGS and I-FSGS cases, in both groups investigated negative correlations existed between TGF-β-1 immunostaining and CD 3+ cells. It is noteworthy however, that only in I-FSGS patients this correlation was statistically significant. This is in concordance with findings that TGF-β-1 inhibits T-cell proliferation, and this biological effect may be of relevance in limiting the acute inflammatory response.31 However, TGF-β-1 has probably variable effects on the immune system both inhibiting cellular proliferation and promoting T-cell memory and cytotoxic function.32 Therefore, the relationship between TGF-β-1 and T lymphocytes in renal pathology is up to now not fully elucidated.
In conclusion, our morphometric and immunohistochemical study suggests that O-FSGS and I-FSGS are separate morphological entities and points out that the latter is more aggressive and destructive glomerulopathy. On the other hand the mechanisms of glomerular and interstitial injury in these cases seem to be rather similar.
Financial support: Medical University of Lodz grant No 503-6038-1.
Table 1. Clinical and laboratory findings at the time of biopsy in cases with O-FSGS and I-FSGS
Table 2. A morphometric comparison of glomerular parameters in cases with O-FSGS and I-FSGS
Table 3. Tubular immunoexpression of TGF-β-1, and analysis of interstitial volume, α-SMA, CD3+ and CD68+cells in O-FSGS and I-FSGS groups
Table 4. Spearman rank order correlations between selected glomerular and interstitial parameters in patients with O-FSGS and I-FSGS
Figure 1.
Figure 2.
Figure 3.
Figure 4.