Primary hyperaldosteronism (PAH) is an important cause of secondary hypertension (HTN). The study of the same requires a high clinical suspicion in addition to a hormonal study that confirms hormonal hypersecretion. It is important to start the appropriate treatment once the diagnosis is confirmed, and for this is necessary to demonstrate whether the hormonal hypersecretion is unilateral (patients who could be candidates for surgical treatment) or bilateral (patients who are candidates for pharmacological treatment only).
At the Hospital del Mar since 2016 there has been a multidisciplinary work team in which Nephrologists, Endocrinologists, Radiologists and Surgeons participate to evaluate cases with suspected hyperaldosteronism and agree on the best diagnostic-therapeutic approach for these patients, including the need for adrenal vein sampling, which is a technique that in recent years has become the gold standard for the study of PAH.
In the present study we collect the experience of our centre in performing AVC and its usefulness for the management of these patients.
El hiperaldosteronismo primario (HAP) es una causa importante de hipertensión arterial (HTA) secundaria. El estudio del mismo precisa de una alta sospecha clínica además de un estudio hormonal que confirme la hipersecreción hormonal. Es importante iniciar el tratamiento adecuado una vez se confirma el diagnóstico y para ello es preciso demostrar si la hipersecreción hormonal es unilateral (pacientes que podrían ser tributarios a tratamiento quirúrgico) o bilateral (pacientes que son tributarios a tratamiento únicamente farmacológico).
En el Hospital del Mar desde el año 2016 existe un equipo de trabajo multidisciplinar en el que participan Nefrólogos, Endocrinólogos, Radiólogos y Cirujanos para evaluar los casos con sospecha de hiperaldosteronismo y consensuar el mejor abordaje diagnóstico-terapéutico de estos pacientes, incluyendo la necesidad de cateterismo venoso adrenal que es una técnica que en los últimos años se ha erigido como gold standard para el estudio de HAP.
En el presente estudio recogemos la experiencia de nuestro centro en la realización de CVA y en la utilidad de este para el manejo de estos pacientes.
Primary hyperaldosteronism (PHA) is one of the main causes of secondary hypertension (HT).1 The prevalence varies greatly from one study to another and partly depends on the populations studied (non-hypertensive vs hypertensive). Studies report a prevalence of up to 10% in hypertensive patients and up to 20% among patients with resistant HT (patients with blood pressure [BP] not controlled despite treatment with three or more drugs at appropriate doses, one of them being a diuretic).1 Early identification and treatment of patients with PHA are important, as their cardiovascular risk has been shown to increase substantially compared to patients with essential hypertension.1,2 In certain cases, moreover, the appropriate diagnosis will enable specific treatment which may resolve the HT, but at least improve the prognosis.
PHA has different forms of presentation. Typically these are patients with poorly controlled HT. The characteristic analytical finding in these patients is hypokalaemia. However, this only occurs in 40% of PHA cases.2,3 The diagnosis of this condition continues to be a challenge today. Initial investigations include determining plasma aldosterone, plasma renin activity and the aldosterone/renin ratio, with elevated plasma aldosterone levels in conjunction with a decrease or even suppression of plasma renin activity being typical of patients with PHA. The aldosterone/renin ratio is the parameter with the greatest sensitivity when starting tests for PHA. However, it is not exempt from false positives and negatives, particularly in association with drugs being taken at the time the test is performed. If the aldosterone/renin ratio is positive, confirmation with a diagnostic test is recommended, such as the saline infusion test, captopril test or stress test with furosemide. There is currently no confirmatory test that is considered the gold standard.4
Once the diagnosis is confirmed, it is important to characterise the PHA subtype (aldosterone-producing adenoma or idiopathic hyperaldosteronism) to assess whether the hormone secretion is unilateral or bilateral, as this will determine the treatment. Clinical practice guidelines propose computed tomography (CT) as an imaging test to assess the adrenal glands.2,4 There are, however, limitations to the information provided by CT. CT is good at identifying adenomas of >1cm in size, but is less effective in the diagnosis of microadenomas. Moreover, as non-functioning adenomas cannot be distinguished by CT, there is the potential risk of adrenalectomy being indicated in a patient who has bilateral secretion.2,5 Adrenal vein sampling (AVS) has the advantage of overcoming the limitations of the CT scan and improving diagnosis of the PHA subtype. It is an invasive procedure in which both adrenal veins are catheterised for the functional study of both glands. Although AVS is a complex technique, particularly the catheterisation of the right adrenal vein due to its anatomical variations, small lumen, and short length, at experienced centres, sensitivity can be as high as 95% and specificity 100%. It has therefore become the gold standard to determine lateralisation of hormone secretion. However, despite all of this, there is currently no international consensus on the protocol to follow and the values for determining lateralisation.5
Our aim with this study was to describe our experience in performing AVS and its outcomes and contribution to the diagnosis and treatment of PHA at Hospital del Mar in Barcelona, Spain.
Population and methodsIn 2016, a multidisciplinary working group was set up at Hospital del Mar in Barcelona for joint assessment and decision-making in patients with suspected PHA. AVS had already been carried out on a non-systematic basis at Hospital del Mar before the creation of the working group. Still, it was through the group in 2016 that coordinated AVS performed in candidate patients became established. For this study, data was collected from 51 patients from 2016 to 2020.
The main criteria for performing AVS is clinical suspicion with a compatible baseline hormone study, mostly with confirmatory tests for PHA.
For the initial investigation of the patients, our centre's specific test protocol was followed. The likely candidate population includes: patients with grade 2 and/or grade 3 HT with resistant HT; patients with HT and hypokalaemia (spontaneous or diuretic-induced); patients with HT and adrenal incidentaloma in an imaging test; patients with HT and a family history of early-onset HT (HT and/or stroke aged <40); HT with a first-degree family history of PHA; and anyone in whom a diagnosis of secondary hypertension is being considered.
For this analysis, BP was considered controlled when, from three clinical measurements taken as recommended in the European Society of Cardiology and Hypertension guidelines6, the average systolic (SBP) and diastolic (DBP) values were less than 140 and 90mmHg, respectively.
Laboratory testsInitially, baseline plasma aldosterone (ng/dl) and plasma renin (ng/mL/h) and the plasma aldosterone/plasma renin activity (PA/PRA) ratio were determined. The blood sample for the hormone tests is taken in the morning two hours after waking and with the patient seated after resting for about 30min. The sample is kept at room temperature before centrifuging and then the plasma is frozen. Baseline plasma aldosterone ≥15ng/dl and/or a PA/PRA ratio >30 was considered as positive. To prepare the patient, serum potassium was corrected (>3.5mmol/l), a normal sodium diet was recommended for the six days before the analysis, and drugs which could directly affect the renin-angiotensin-aldosterone axis were withdrawn, allowing only treatment with verapamil, diltiazem, doxazosin and hydralazine; in the case of mineralocorticoid antagonists, these were discontinued for the previous 6–8 weeks.
In patients in whom an PA/PRA ratio >20 was obtained, a confirmatory PHA test was performed. At our centre we carry out a saline overload or a captopril suppression test. The saline overload test involved administering two litres of 0.9% saline for four hours at 500mL/h, determining baseline and post-overload potassium, PA and PRA levels. No complications were reported in the seven patients who had the saline overload test. We do not routinely perform the saline overload test in patients with severe hypertension, advanced-stage chronic kidney disease, a history of heart failure, arrhythmias or severe hypokalaemia. If the PA was >8ng/dl after the test, the patient was diagnosed with PHA. The captopril suppression test involved measurement of PA and PRA levels at baseline and after administering 25mg of captopril orally (or 50mg if body weight >85kg). If PA was >15ng/dl or the PA/PRA ratio was >50, the test was considered as positive.7
After the diagnosis of PHA, a non-contrast thin-slice CT (3mm) of the adrenal glands was performed.
Adrenal vein samplingThe patients candidates for AVS, and patients in whom there was uncertainty, were discussed at a multidisciplinary session with nephrologists, endocrinologists, urologists, surgeons and radiologists, in order to reach a consensus on the approach to take in terms of continuing diagnostic procedures. AVS was indicated in patients who met any of the following criteria: unilateral adenoma (>1cm) in patients aged over 40 (patients <40 with unilateral adenoma and confirmatory PHA tests were considered for elective surgical treatment, as the prevalence of non-secreting adrenal incidentalomas in this age group is very low); PHA confirmed but with negative imaging test; unilateral micronodules (<1cm); bilateral micronodules; unilateral adrenal hyperplasia.
AVS was performed in the vascular intervention suite using ultrasound-guided access to the femoral vein (generally double right access) and simultaneous extraction of 4mL of blood from each adrenal vein, as well as a peripheral venous sample through the same femoral access, to calculate the ratios between the adrenal and peripheral samples. ACTH infusion (Cosyntropin) was not used routinely. The procedure was considered a technical success when the selectivity index was significant on both sides (adrenal to peripheral cortisol ratio >3:1). An aldosterone/cortisol ratio of the dominant gland with respect to the non-dominant gland greater than 4 was considered as a lateralisation criterion.
In this retrospective observational study, we analysed the utility of AVS in a cohort of 51 patients with primary hyperaldosteronism, evaluating the results, decision-making, treatment and outcome in these patients. Demographic, analytical and additional test data were collected for all patients included. The SPSS version 25.0 program (Cary, NC, USA) was used to carry out the statistical analysis.
ResultsWe collected data from 51 patients diagnosed with PHA; 76% were male and mean age at diagnosis was 53.1±9.2 years. The majority (85.5%) were overweight or obese. and 13% had type 2 diabetes.
All the patients included in the study were hypertensive and met at least one of the criteria for the diagnosis of PHA. Table 1 shows the average SBP and DBP figures, the number of drugs taken before the diagnosis of PHA, and the biochemical laboratory data, including the albumin/creatinine ratio determined in a fresh urine sample obtained in the morning.
Baseline characteristics in the sample.
| Variable | |
|---|---|
| Baseline SBP (mmHg) | 146.8±19.5 |
| Baseline DBP (mmHg) | 87.2±13.2 |
| Number of drugs/patient | 3.3±1.38 |
| Patients with aldosterone antagonists, n (%) | 18 (35.3) |
| eGFR (ml/min/1.73 m2) CKD-EPI | 85.4±18.7 |
| Plasma potassium (mmol/l) | 3.4±0.5 |
| Plasma sodium (mmol/l) | 142±2.2 |
| Urine albumin/creatinine ratio (mg/g)a | 10.7 [5.1−58.1] |
eGFR: estimated glomerular filtration rate; DBP: diastolic blood pressure; SBP: systolic blood pressure.
The albumin/creatinine ratio (ACR) at diagnosis of PHA was 10.7 [5.1−58.1] mg/g. The mean estimated glomerular filtration rate was 85.4±18.7mL/min/1.73m2, while plasma potassium was 3.4±0.5mmol/l.
In the study of the renin-angiotensin-aldosterone axis, 82.3% of the patients had an PA/PRA ratio greater than 30, while 50% of them had serum potassium levels from 2 to 3mmol/l.
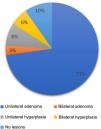
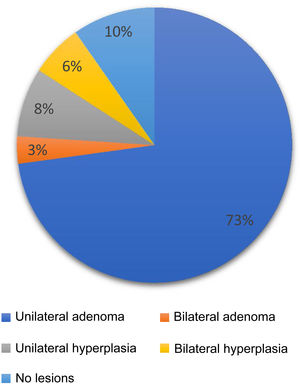
Additional investigationsThirty-one captopril suppression tests were performed (24 were positive and seven negative), and seven saline infusion tests were performed, all positive and with no associated complications. Confirmatory tests were not performed on the rest of the patients due to medical contraindications or high suspicion of PHA. All patients had an imaging test with a thin-slice CT; 72.6% of the scans were compatible with unilateral adenoma, 3.2% had bilateral adenoma, 8.1% unilateral hyperplasia, 6.1% had bilateral hyperplasia, and 9.7% had no lesions detected in the adrenal glands (Fig. 1).
Adrenal vein sampling and treatmentOf the 51 patients diagnosed with PHA, AVS was performed in 47, with the procedure being a technical success in 91.5% of cases. Correct catheterisation of the right adrenal vein was not achieved in four patients. No complications associated with the endovascular procedure were reported. Table 2 shows the relationship between the tests performed and the final treatment decision.
Relationship between the results of additional investigations and adrenal vein sampling (AVS).
| List of tests | Number | % | Adrenalectomy | |
|---|---|---|---|---|
| Yes (%) | No (%) | |||
| Positive hormonal study and positive CT and AVS lateralises | 24 | 47 | 16 (66.7) | 8 (33.3)a |
| Positive hormonal study and negative CT and AVS lateralises | 3 | 5.8 | 3 (100) | 0 |
| Positive hormonal study and positive CT and AVS does not lateralise or is not diagnostic | 16b | 31.4 | 0 | 16 (100) |
| Positive hormonal study and negative CT and AVS does not lateralise | 4 | 7.8 | 0 | 4 (100) |
| Decision without AVS | 4 | 7.8 | 4 (100) | 0 |
Of the 51 patients with PHA, the hormone study coincided with the CT and AVS findings in 47%. However, in another 31% of the patients, the hormone study was diagnostic and the CT showed findings suggestive of unilateral secretion, but the AVS did not show lateralisation in them, so surgery was ruled out. In 6% of the patients, the hormone study confirmed the diagnosis of PHA, and despite the imaging test showing the adrenal glands to be normal, when performing the AVS, lateralisation was found and surgery indicated. In another four patients, surgical treatment was decided on without prior AVS. Three of these patients met surgical criteria in size (>40mm), and one had an image of a three-lobed nodule, which on its own was considered as an indication for surgical excision.
We should point out that of the 24 patients who had a positive hormone study with a diagnostic CT and an AVS showing lateralisation, eight were still awaiting the decision on surgical intervention at the time of writing this review.
Blood pressure and drug changesChanges in BP and the number of drugs were analysed six months after adrenalectomy in patients in the surgical group and six months after the AVS in patients given exclusively medical treatment.
As shown in Table 1, the mean BP before being tested for PHA was 146.8 (±19.5)/87.2 (±13.2) mmHg, with BP being poorly controlled in 78% of the patients. Most of the patients were treated with at least three antihypertensive drugs and 36% were taking aldosterone antagonists before being investigated.
After conducting the PHA testing and deciding on the course of action in terms of treatment, 78% of the patients in the overall sample had their BP under control and 44% were able to reduce the number of antihypertensive drugs they were taking.
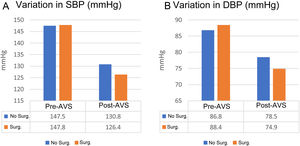
Among the patients who underwent adrenalectomy, 73.9% had improved BP control six months after surgery, and the same percentage were able to reduce the number of drugs. Analysing overall improvement, defined as improvement in BP and/or reduction in the number of drugs, 91% of the patients who had an adrenalectomy obtained benefit in the management of their HT. The mean decrease in BP in this group was 21.4mmHg for SBP and 13.5mmHg for diastolic BP (p=0.005 for both). Fig. 2 shows the relationship between pre-AVS and post-AVS SBP and DBP in the patients who had surgery and those given purely drug treatment, respectively.
Variation in systolic (A) and diastolic (B) blood pressure between the groups treated with adrenalectomy (Surg.) and those who remained on medical treatment (No Surg.) after adrenal vein sampling.
AVS: adrenal venous sampling; Surg.: surgical intervention; DBP: diastolic blood pressure; SBP: systolic blood pressure.
Among the non-surgical patients who had an AVS and who were on drug therapy for their hypertension, in 69.2% control of their BP improved after the AVS and 15.4% were able to reduce the number of drugs they were taking. There was also a 49.6% increase in patients on treatment with aldosterone receptor antagonists. In this group, overall improvement was found in 61.5% of the sample and the mean changes in BP were 16.7mmHg for SBP and 8.3mmHg for DBP (p<0.05 in both).
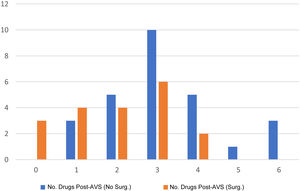
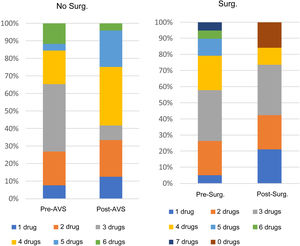
Fig. 3 shows the relationship between the number of drugs/patient in the two groups after optimisation of the respective treatments. Between the two groups, the number of drugs/patient after intervention was lower in the adrenalectomy group, in which 13% of patients were able to discontinue all antihypertensive drugs.
Three patients in whom the CT did not show a unilateral adenoma benefited from adrenalectomy after having the AVS, which did show lateralisation. These three patients did not have adequate BP control prior to adrenalectomy and were receiving aldosterone antagonist therapy. In one of them, BP control had not improved six months after surgery, but overall, the other two patients did obtain improvements in their HT. Six months after adrenalectomy, one of them reduced the number of drugs from seven to three, achieving adequate BP control. The third patient was able to reduce from five drugs to three, also with adequate BP control.
DiscussionPHA is the most common cause of secondary hypertension and leads to poor BP control. Treatment is dependent on reaching the right differential diagnosis. For that reason, AVS has been used for the classification of the PHA subtype for 50 years8 and has gained importance in recent years3. In one study, the comparison between examination of the adrenal glands by imaging techniques and AVS demonstrated the superiority of AVS with a sensitivity of 92% versus 67% with CT.9 CT is established as one of the first tests to identify the PHA subtype. Still, it loses diagnostic power with age due to the prevalence of incidentalomas over the age of 35.10 Approximately 95% of patients with PHA are over 35,11 although it cannot be ruled out that this is due to them being investigated late. It is common for CT to show bilateral adrenal hyperplasia, adrenal glands with normal morphology or bilateral adenomas. In these cases, it is necessary to use a more specific examination technique than CT to demonstrate whether or not there is unilateral hypersecretion with suppression of secretion in the contralateral gland.10
Several studies have analysed the utility of AVS in PHA typing compared to imaging tests.11,12 Young et al.,12 analysing 203 patients assessed with both techniques, found that CT was accurate in only 53% of cases. In 22% of the patients, adrenalectomy would have been wrongly ruled out if treatment had been guided by the imaging test results. Similarly, 25% of patients could have had an unnecessary adrenalectomy. In our study, in 31.4% of the patients with a diagnostic hormone study suggesting PHA and an imaging test compatible with unilateral involvement, the AVS ruled out surgery (Table 2). In a review that included 950 patients, the imaging test was inconsistent with the AVS results in 37.8% of the patients, similar to the 40.4% we report here.13
AVS is a complex technique, and the success rate depends on several factors. In our experience, 91.5% of the AVS procedures performed were successful, a rate similar to that described in the literature, where the reported rate is as high as 96%.12,14,15 Improving the study of the PHA subtype and determining whether aldosterone hypersecretion is unilateral or bilateral is important for selecting patients who are candidates for adrenalectomy. The AVIS-2 study has already demonstrated the superiority of adrenalectomy over medical therapy in terms of BP control in these cases.16 Other studies have shown that patients who, despite optimal treatment, are left with a certain degree of hyperaldosteronism, have a greater long-term cardiovascular risk.17 It is important to consider that patients with PHA have echocardiographic changes, such as ventricular hypertrophy, which improve after introducing adequate treatment.18,19
A recent meta-analysis including 31 studies compared long-term cardiovascular outcomes in 3000 patients with PHA and more than 9000 patients with essential hypertension, and found that patients with PHA had increased cardiovascular risk compared to the essential HT group.20 This suggests that early diagnosis of PHA and starting specific treatment improves these patients' prognosis. Moreover, the latest studies suggest that there would be a benefit from surgical treatment in patients with indication for adrenalectomy. In our study, 6% of the patients were able to benefit from surgical treatment after having the AVS, despite the CT scan not being diagnostic. With the fact that PHA is the leading cause of secondary hypertension, this figure supports the need to consider AVS in the majority of patients with a clinical and biochemical diagnosis of PHA, as long as they agree to surgery if so indicated by the results.
As shown Fig. 2, the decrease in both systolic and diastolic BP was significantly greater in the group that underwent adrenalectomy. Previous studies show a similar decrease in BP between patients who receive medical treatment and those who are candidates for adrenalectomy. However, candidates for adrenalectomy have also been found to have a greater reduction in the number of drugs.18,19 In our cohort, as shown in Fig. 4, most of the patients were on fewer than three drugs six months after surgery. Considering that only 50% of patients adhere to their long-term drug treatments,21,22 it would be helpful to identify the patients, such as those with PHA secondary to unilateral secretion, who might most benefit from a reduction in their antihypertensive drugs.
Performing AVS requires expert healthcare professionals with a well-established learning curve, as it involves certain technical difficulties which can only be overcome with dedication and experience. Being able to provide these factors, along with the consensus reached on each patient by the multidisciplinary working group, both on whether or not to perform the AVS and for the subsequent decision concerning treatment, has consolidated Hospital del Mar as one of the main centres of reference in this disorder.
ConclusionsThe data we have provided here confirm that AVS is an essential technique in the study and treatment of PHA. Although the hormone study and imaging tests are fundamental in identifying the patients most likely to benefit from surgery, with AVS we can be more precise and make the most appropriate treatment decision in each case. As we have shown, AVS helps rule out surgery in some patients who have bilateral hypersecretion and would not, therefore, benefit from surgery while also identifying patients who are candidates for surgical treatment despite a negative or inconclusive imaging test but with clear hyperaldosteronism secondary to unilateral secretion. We have also shown that with the help of AVS, selection of patients who then underwent adrenalectomy reduced the number of antihypertensive drugs required and improved control of their hypertension, with the likely consequence of improving their cardiovascular profile.
Conflicts of interestThe authors declare that they have no conflicts of interest.