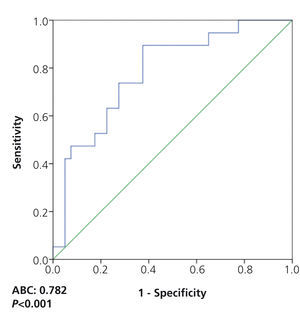
Introducción: Estudios recientes sugieren que los niveles del receptor soluble de la uroquinasa (suPAR) podrían ser útiles para diferenciar la glomeruloesclerosis focal y segmentaria (GFS) idiopática de otras glomerulopatías causantes de síndrome nefrótico, pero estos datos no han sido confirmados en estudios independientes. El objetivo de nuestro estudio es analizar si los niveles circulantes de suPAR son útiles para identificar la enfermedad renal primaria en enfermos afectos de síndrome nefrótico secundario a GFS, enfermedad por cambios mínimos o nefropatía membranosa (NM) idiopática. Métodos: Se realizaron mediciones de niveles de suPAR circulante en el momento del diagnóstico en 60 pacientes con síndrome nefrótico secundario a GFS, enfermedad por cambios mínimos (ECM) y NM. Se analizaron las correlaciones entre niveles de suPAR y variables demográficas, clínicas y bioquímicas. La sensibilidad y la especificidad de suPAR para diferenciar a los enfermos con GFS se analizaron mediante curvas ROC. Resultados: Tras ajustar por edad y función renal, los niveles de suPAR fueron significativamente más elevados en enfermos con GFS que en ECM (p < 0,001), pero no hubo diferencias entre GFS y NM (p = 0,12). Un valor de suPAR ≥ 3452 pg/ml tuvo una sensibilidad del 73,7 % y una especificidad del 72,5 %, con un área bajo la curva (ABC) de 0,782 ± 0,124, p = 0,001, para identificar a los enfermos con GFS. Tras excluir a los enfermos con NM, un valor ≥ 3531 pg/ml tuvo una especificidad del 99,93 % para diferenciar entre ECM y GFS. Conclusiones: Los valores de suPAR por sí solos no diferencian entre los tres tipos de glomerulopatía. Sin embargo, tras excluir el diagnóstico de NM, un nivel de suPAR > 3531 pg/ml podría tener una elevada especificidad (pero baja sensibilidad) para el diagnóstico de GFS.
Introduction: Recent studies suggest that soluble urokinase-type plasminogen activator receptor (suPAR) levels could be useful for distinguishing idiopathic focal segmental glomerulosclerosis (FSGS) from other glomerulopathies that cause nephrotic syndrome, but these data have not been confirmed in independent studies. The objective of our study is to analyse whether circulating levels of suPAR are useful for identifying primary kidney disease in patients with nephrotic syndrome secondary to FSGS, minimal change disease or idiopathic membranous nephropathy (MN). Methods: We measured circulating suPAR at diagnosis in 60 patients with nephrotic syndrome secondary to FSGS, minimal change disease (MCD) and membranous nephropathy (MN). The correlations between suPAR levels and demographic, clinical and biochemical variables were analysed. The sensitivity and specificity of suPAR in distinguishing FSGS patients were analysed by ROC curves. Results: After adjusting for age and renal function, suPAR levels were significantly higher in patients with FSGS than in those with MCD (p<.001), but there were no differences between FSGS and MN (P=.12). A suPAR value ≥3452pg/ml had a sensitivity of 73.7% and a specificity of 72.5%, with an area under the curve (AUC) of 0.782±0.124, p=.001, for identifying patients with FSGS. After excluding patients with MN, a value ≥3531pg/ml had a specificity of 99.93% for distinguishing between MCD and FSGS. Conclusions: suPAR values alone do not distinguish between the three types of glomerulopathy. Nevertheless, after excluding the diagnosis of MN, a suPAR level >3531pg/ml could have a high specificity (but a low sensitivity) in the diagnosis of FSGS.
Minimal change disease (MCD), focal segmental glomerulosclerosis (FSGS) and membranous nephropathy (MN) are the three primary glomerulopathies responsible for most pure nephrotic syndromes in adults.1-3 The three diseases are similar in presentation, and as such, it is not usually possible to distinguish between them using clinical or biochemical criteria and, in order to obtain an accurate diagnosis, it is necessary to perform a renal biopsy. The presence of a soluble glomerular permeability factor is currently considered the main aetiological factor of primary FSGS.4-10 In a recent study, Wei et al.11 identified the soluble urokinase-type plasminogen activator receptor (suPAR) as one of the potential factors responsible for podocyte injury in FSGS and proposed that a serum level higher than 3000pg/ml could be a sensitive and specific biomarker for distinguishing idiopathic FSGS from other nephrotic syndrome forms. However, although the number of patients with MN included in this study was low, some patients with MN displayed suPAR values that were higher than the selected cut-off point, which suggests that suPAR does not allow for adequate differentiation between both diseases. Furthermore, the data of Wei et al. were not reproduced in an independent study that was carried out on a small number of patients, in which no differences were observed in suPAR levels between patients with primary and secondary FSGS and between both forms and patients with minimal change disease.12,13
In a subsequent study, Wei et al.14 measured suPAR levels in young patients with corticosteroid-resistant FSGS and in patients younger than 18 years of age recruited by the European consortium PodoNet for the corticosteroid-resistant nephrotic syndrome study and they confirmed that, in both groups of patients with FSGS, suPAR values were significantly higher than those of healthy controls, but with a very heterogeneous distribution and a considerable overlapping of values between patients and healthy controls. In the American cohort of the aforementioned study, baseline suPAR levels were independently associated with the glomerular filtration rate, which is similar to the findings of Maas et al.12, and with black patients, which suggests that the suPAR value may be different depending on the level of renal function or the ethnic group studied.
In a study carried out very recently in China15 that included both children and adults with idiopathic MN, MCD and secondary FSGS, higher suPAR levels were found in patients with idiopathic FSGS than in the other groups, but suPAR levels did not distinguish between idiopathic FSGS and other glomerulopathies due to the considerable overlapping of values between groups. Furthermore, mean suPAR levels of patients with idiopathic FSGS reported in the aforementioned study were clearly lower than those observed in previous studies,11,12 which, along with the evidence of racial influence reported in the North American cohort,12 suggests that there could be ethnic differences in the distribution of suPAR levels or in the pathogenic relationship between suPAR and FSGS. These findings suggest the clinical usefulness of serum suPAR levels with regard to inconclusive FSGS in clinical practice.16 However, regardless of any potential differences there may be between studies in terms of ethnic/geographic variables, sample size or idiopathic FSGS form classification criteria, the studies carried out to date agree on identifying a patient group with FSGS that present high serum suPAR levels, but the data provided do not allow us to know whether these levels are associated or not with certain clinical characteristics of the disease.
In this study, we measured circulating suPAR levels at the time of diagnosis in a cohort of patients with idiopathic nephrotic syndrome caused by MCD, FSGS or MN with the following objectives: 1) To analyse the clinical, biochemical and histopathological variables associated with circulating suPAR levels. 2) To analyse whether serum suPAR level is useful for distinguishing between patients with patients with FSGS and those with MCD and MN. 3) To analyse whether, in patients with idiopathic FSGS, there are differences in the baseline clinical or biochemical characteristics of the disease depending on the circulating suPAR level.
PATIENTS AND METHOD
We included 60 patients >18 years of age with pure nephrotic syndrome, defined by proteinuria >3.5g/day, serum albumin <3.5 g/dl, hyperlipidaemia and oedema, in the absence of haematuria or high blood pressure, secondary to idiopathic FSGS, MCD or MN, diagnosed by renal biopsy. Primary FSGS was diagnosed when there was evidence of typical lesions in the optical microscope (all biopsies were analysed by the same pathologists) and diffuse podocyte effacement in the electron microscope, and after ruling out secondary aetiologies, including: renal mass reduction, morbid obesity, nephropathy associated with human immunodeficiency virus, heroin or cocaine use, infection by parvovirus B19, analgesic, bisphosphonate or interferon use, vesicoureteral reflux or obstructive sleep apnoea. In 4 patients under the age of 30, we carried out a genetic study that ruled out the presence of nephrin or podocin mutations. All patients with FSGS displayed the classic variant (NOS). In the immunofluorescence study, we detected IgM deposits in 12 patients, C3 deposits in 3, IgM and C3 deposits in 2, and no deposits in 3 patients. Idiopathic MCD was diagnosed when there was an absence of apparent lesions in the optical microscope and the presence of diffuse podocyte fusion in the electron microscope, after ruling out a history of medication use or associated lymphoproliferative processes. MN was diagnosed when there was evidence of characteristic data in the optical microscope and subepithelial IgG and C3 deposits in the immunofluorescence study. In all patients with membranous nephropathy included in the study, we confirmed that antibodies were positive against the phospholipase A2 receptor (anti-PLA2R) and we ruled out potential secondary causes, as per the protocol. When the blood samples were obtained, no patient was receiving treatment with corticosteroids, immunosuppressants, angiotensin II receptor antagonists, aldosterone receptor antagonists or statins.
Methods
Serum creatinine was measured by the IDMS-traceable compensated method (Hitachi Modular P-800 Roche Diagnostics, Germany). The estimated glomerular filtration rate (eGFR) was calculated using the CKD-EPI formula.17 SuPAR levels were measured from serum samples using a commercial ELISA kit (Human uPAR Quantikine® ELISA kit; R&D Systems, Minneapolis, MN, USA; intra-assay variability: 4.1%-7.5%; inter-assay variability: 5.1%-5.9%).11 To analyse reproducibility of measurements, three or more suPAR measurements were taken during the nephrotic phase before starting treatment in 11 patients and coefficients of variation below 10% were observed. Anti-PLA2R antibodies were measured by a commercial immunofluorescence assay (Anti-Phospholipase A2 receptor IIFT; Euroimmun AG, Lübeck, Germany).18,19
Pathological analysis of renal biopsies
The biopsies were stained with haematoxylin and eosin, PAS (Periodic acid–Schiff)- methenamine and Masson's trichrome for morphological analysis and immunofluorescence studies were carried out with antibodies against IgA, IgG, IgM, C3, fibrinogen and light chains and were processed for an electron microscope study. In biopsies with a MN pattern, we carried out immunohistochemical staining with anti-PLA2R antibodies (HPA012657, Sigma-Aldrich Co.LLC. St Louis, USA).
This study adhered to the parameters established by the Declaration of Helsinki. All patients gave their informed consent in writing and the bioethics committee of the corresponding centre approved the study.
Statistical analysis
Quantitative variables were expressed as the mean ± 1 standard deviation and the qualitative variables were expressed as a proportion. Group means were compared using the Student’s t-test for independent data in the case of two means, or variance analysis with the Bonferroni correction for group comparison in the case of more than two means. Categorical variables were compared using the χ2 test. In each separate kidney disease, we analysed the relationship between suPAR levels and demographic and biochemical variables and we considered suPAR concentrations as continuous variables and tertiles. We carried out a variance analysis, ordering the variables studied in tertiles with the aim of identifying the variables that were independently associated with the circulating suPAR level. After analysing the differences between groups, we analysed the sensitivity and specificity of suPAR levels in order to identify patients with FSGS, by ROC curves. We considered P values <.05 to be significant. The statistical calculations were carried out using the SPSS 20.0 software.
RESULTS
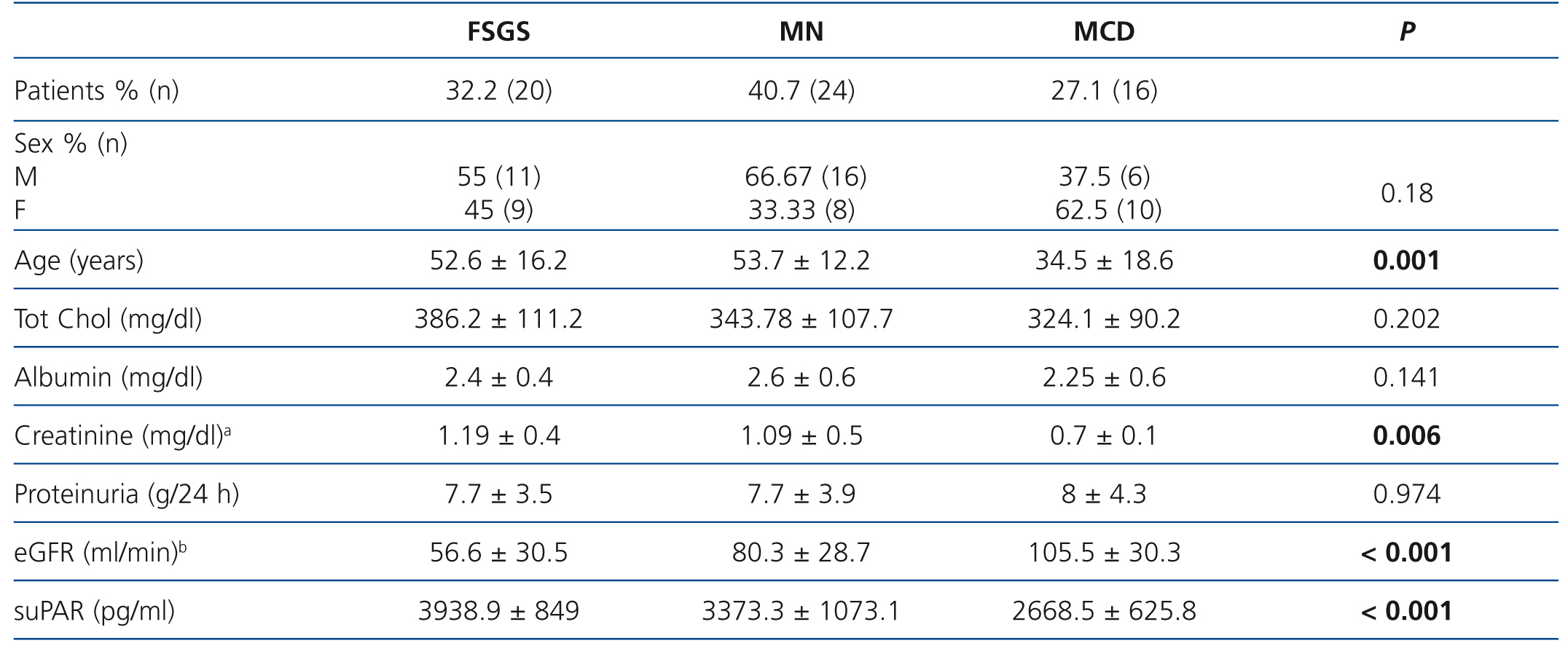
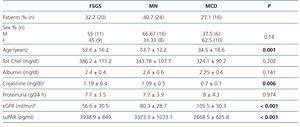
Table 1 summarises the clinical and demographic characteristics of the patients included in the study. We did not observe significant differences in terms of distribution by sex between study groups. Patients with MCD were younger and had lower serum albumin levels than patients with FSGS and MN.
Patients with FSGS displayed significantly lower eGFR values than in the other two groups and patients with MCD displayed higher eGFR values (FSGS vs. MN, P=.040; FSGS vs. MCD, p<.001; MN vs. MCD, p=.045)
No significant differences were observed in suPAR values between sexes in any of the three glomerular disease groups.
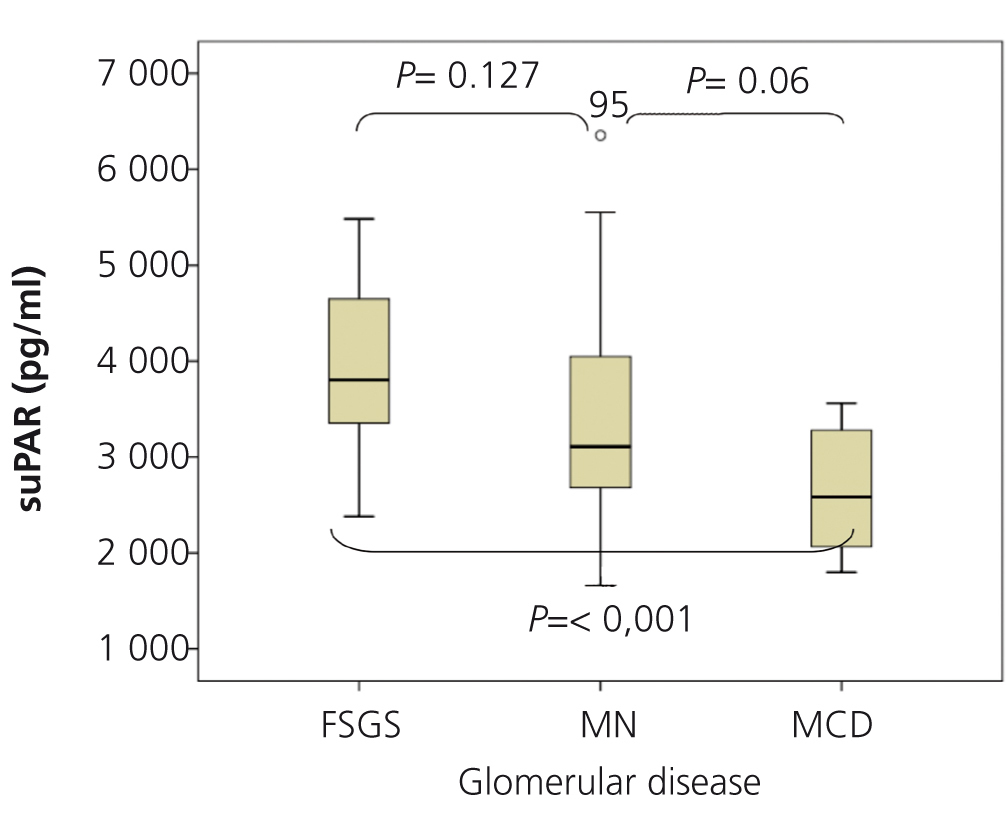
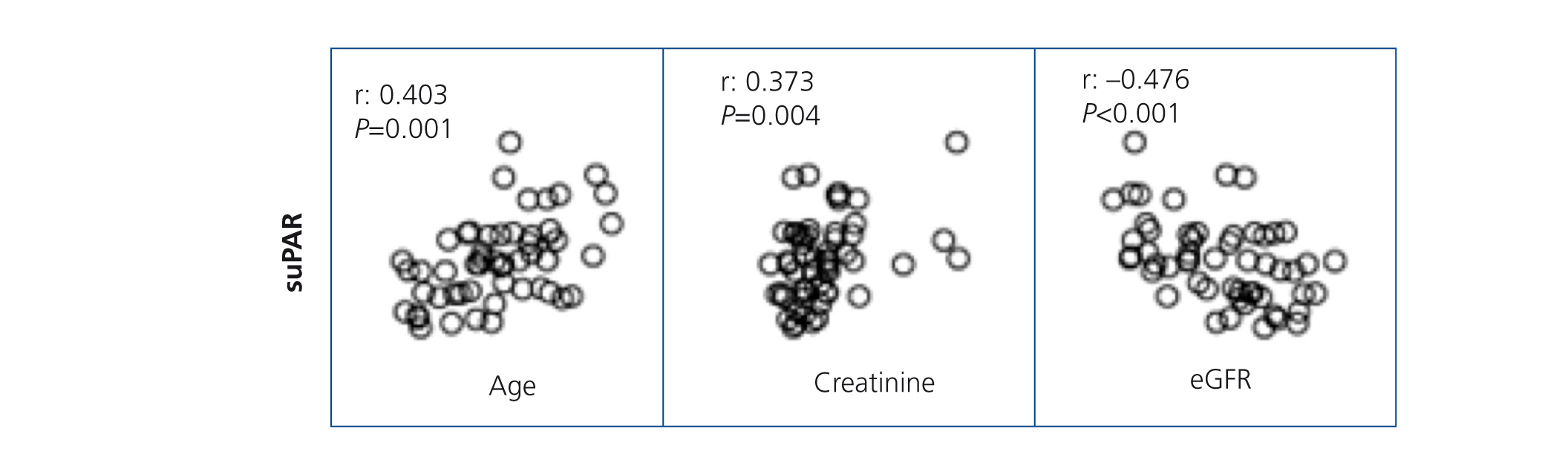
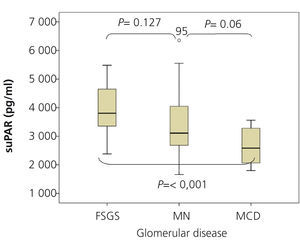
SuPAR levels of patients with MCD were significantly lower than in patients with FSGS (2668.5±625.8 vs. 3938.9±849pg/ml, p<.001), but we did not observe significant differences between patients with MN and FSGS (3373.3±1073.1 vs. 3938.9±849pg/ml, p=.127) or between MCD and MN (2668±625.8 vs. 3373.3±1073.1 p=.055) (Figure 1). In the overall patient sample, suPAR values were correlated positively with age and serum creatinine and negatively with eGFR (Figure 2). We did not observe statistically significant correlations between suPAR levels and proteinuria.
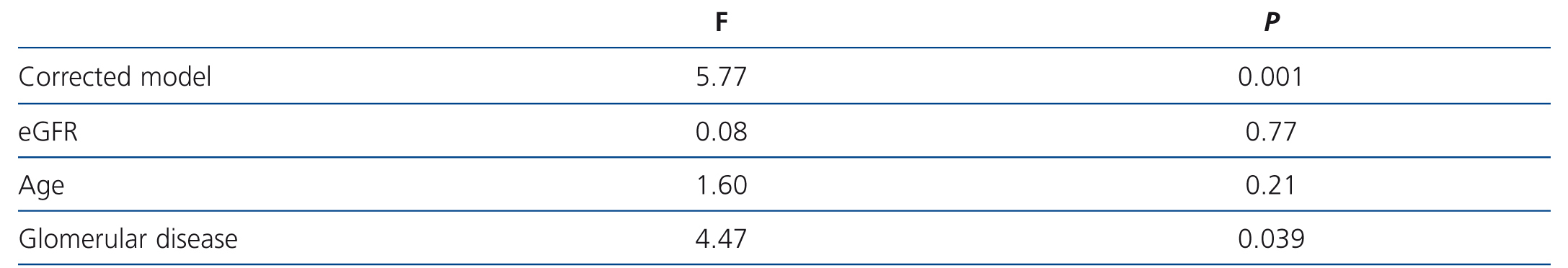
In the variance analysis, we observed that, after adjusting for age and glomerular filtration rate, the only variable significantly associated with suPAR level was the type of glomerular disease and this association was due to patients with MCD having significantly lower suPAR levels than patients with FSGS and MN. Overall, the model explained 26.9% of the variability observed in suPAR levels (F: 4.47, p=.039) (Table 2), which were not explained by the differences in age and glomerular filtration rate between the groups.
After grouping the patients of each glomerular disease group by the respective suPAR tertiles, no significant differences were observed between groups in any of the clinical or biochemical characteristics of the disease at diagnosis (data not displayed).
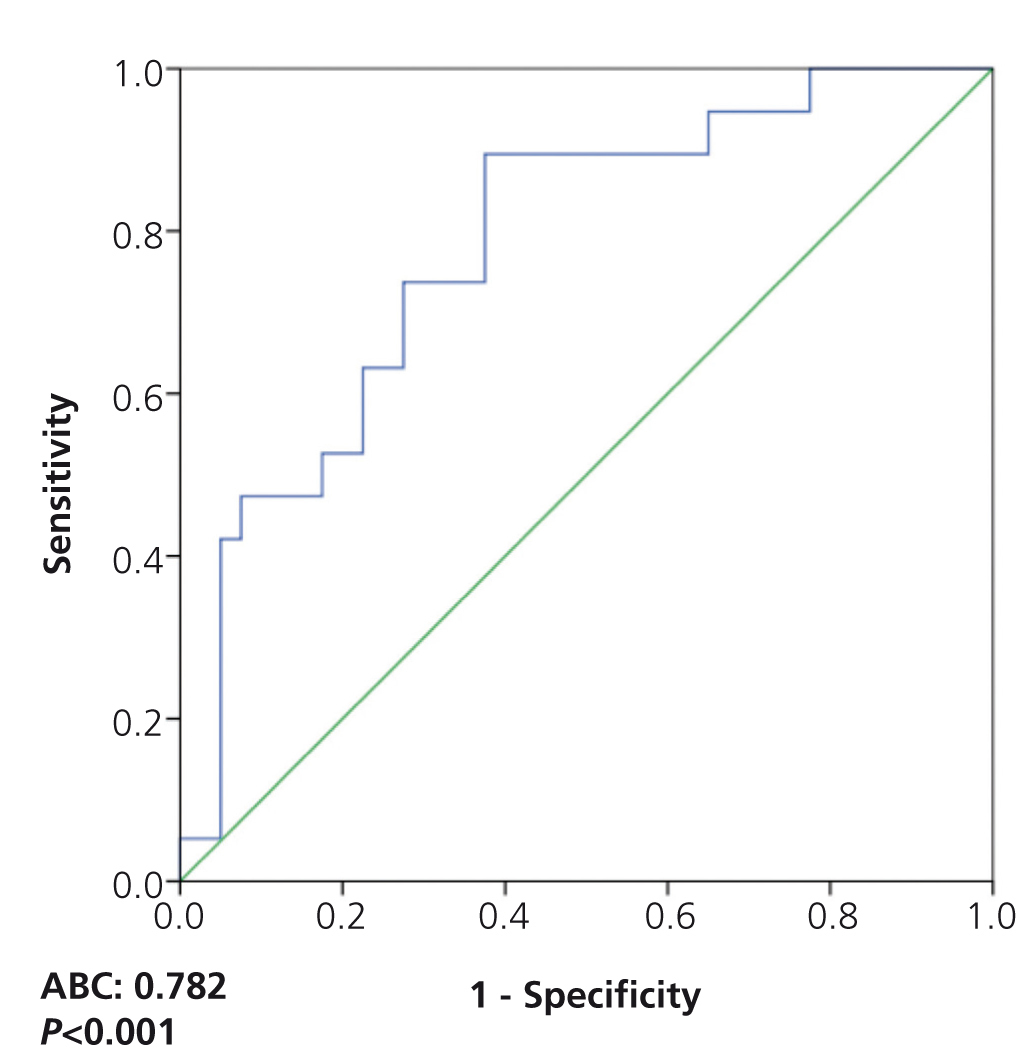
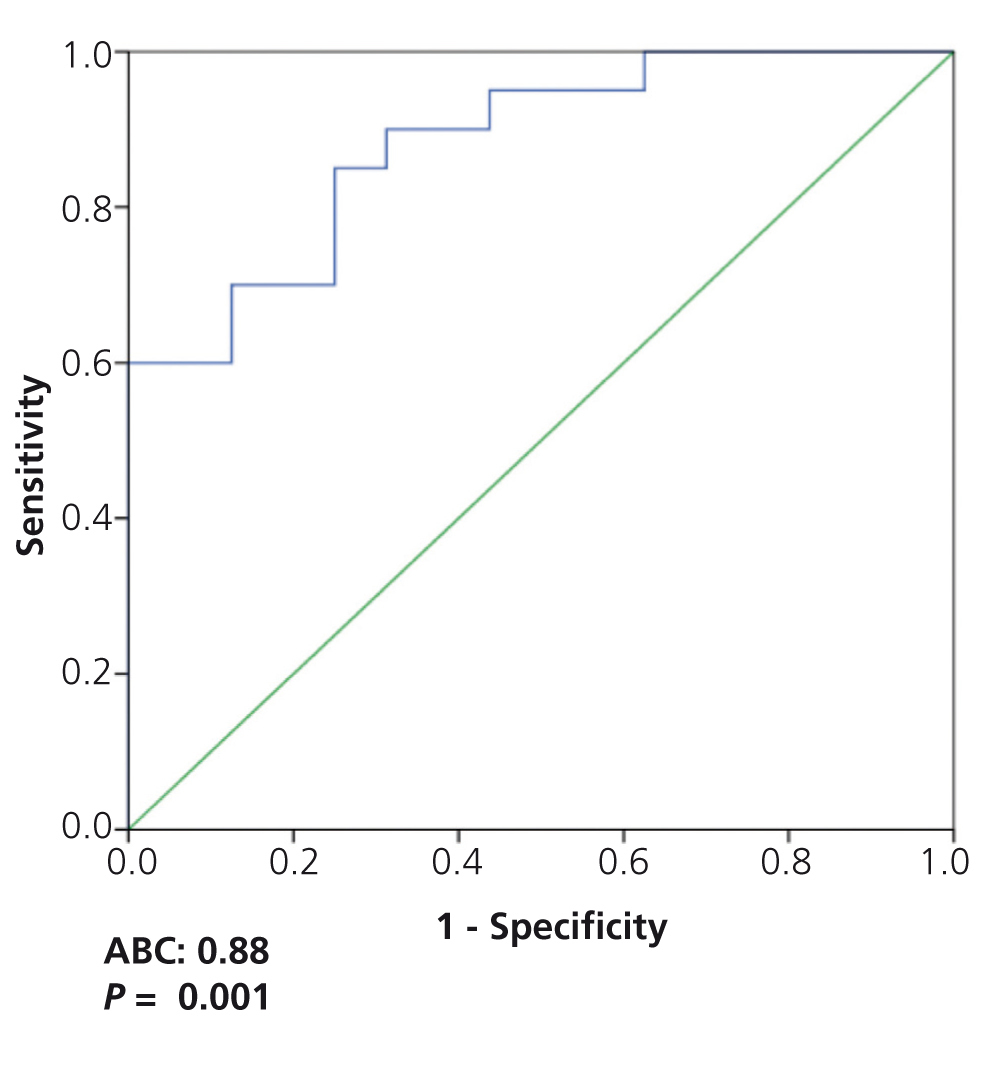
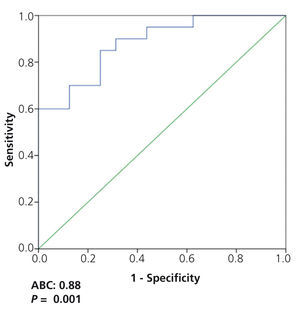
On analysing the potential diagnostic value of the serum suPAR level for the identification of patients with FSGS in the overall patient group, we observed that a value of 3452pg/ml had a sensitivity of 73.7% and a specificity of 72.5%, with an area under the curve (AUC) of 0.782±0.124, p=.001 (Figure 3). On analysing the capacity of suPAR to distinguish between patients with FSGS and MCD, after excluding patients with MN from the analysis, we observed that a value ≥3442.4pg/ml had a sensitivity of 70% and a specificity of 87.5%, with an AUC of 0.881 ± 95% confidence interval (CI) 0.107, p<.001. Values ≥3530.9pg/ml had a specificity of 99.93% for distinguishing between the two diseases (Figure 4) at the expense of sensitivity.
DISCUSSION
The results of our study indicate that circulating suPAR levels are related to the type of kidney disease that causes the nephrotic syndrome, but also to age and renal function. The relationship observed between suPAR levels, age and the glomerular filtration rate coincides with that reported in previous studies.11-14 In the multivariate analysis, we observed that after making the corresponding adjustments, the only variable with significant association was the type of glomerular disease and the association was due to MCD patients displaying significantly lower suPAR levels than patients with FSGS and MN, without significant differences being observed between the two latter groups. The differences found between FSGS and MCD coincide with those reported in two previous studies.11-14 Both mean levels and the distribution of suPAR values observed in our FSGS patient group are similar to those reported by Wei et al.11, but unlike the latter, our data coincide with those described by Huang15 in which suPAR levels higher than 3000ng/ml do not allow adequate differentiation between patients with FSGS and patients with the other two glomerular diseases that cause nephrotic syndrome. Patients with MCD displayed a more homogeneous distribution and lower variability in suPAR levels than patients with FSGS and MN. Given the wide dispersion of suPAR values observed in the FSGS patient group, we considered it to be of interest to analyse whether there were differences in the clinical profile of the disease depending on the suPAR level, but we did not find an association between the latter and any clinical or biochemical characteristic studied.
Through analysis of ROC curves, we observed that suPAR levels had a low sensitivity and specificity for adequately identifying patients with FSGS mainly due to the high overlapping of values between patients with FSGS and MN on one hand, and between those with MN and MCD on the other.
Furthermore, taking into account the existing differences between FSGS and MCD with regard to the prognosis and/or response to treatment and their differences in suPAR levels, we considered it to be of particular interest to analyse the usefulness of suPAR for distinguishing between the two glomerular diseases. Our data indicate that the sensitivity of suPAR in distinguishing between them was low due to the considerable number of patients with FSGS and low suPAR levels. Nevertheless, it was possible to identify a suPAR level with a high specificity (99%) to rule out the diagnosis of MCD, due to the low likelihood of finding a suPAR concentration equal to or greater than 3531ng/ml in MCD. This data, if confirmed in independent studies, could have practical implications in cases in which the renal biopsy is inconclusive or in cases classified as MCD that do not respond to treatment as expected.
The main limitation of this study is its sample size. On analysing the distributions in suPAR levels in each group and the differences between groups, it is possible to consider that the inclusion of a higher number of patients, with a reduction in the CI range, could result in a higher sensitivity for distinguishing between FSGS and MCD. However, given the wide dispersion of suPAR levels observed both in FSGS and in MN, although an increase in the sample size would allow detection of significant differences between both, it seems indisputable that a low suPAR value would not allow us to rule out the diagnosis of FSGS, nor would a high suPAR value allow us to rule out MN. Another limitation to bear in mind, which is common to all studies that include patients with both diseases, is the potential classification error between MCD and FSGS. The diagnostic criteria used in this study exclude the possibility of patients with MCD having been classified as FSGS patients. However, due to the focal nature of the lesions, it is not possible to rule out the possibility that some patients with FSGS may have been classified as MCD patients when no segmental sclerosis lesions have been detected in the optical or electron microscopes in the kidney tissue sample available for analysis. Although we are aware of this limitation, we consider it unlikely that this would lead to a systematic error with an important influence on the results.
In summary, our data indicate that in patients with nephrotic syndrome caused by MCD, FSGS or MN, after adjusting for age and renal function as the main confounding variables, MCD patients had significantly lower serum suPAR levels than FSGS or MN patients. Despite these differences, the high overlapping of values between groups means that suPAR values alone do not provide useful information in clinical practice for distinguishing between the three types of glomerulopathy. However, once MN has been definitively ruled out as the cause of nephrotic syndrome, in patients for whom there are doubts about whether to diagnose MCD or FSGS due to the presence of inconclusive lesion patterns and when it is not possible to carry out another biopsy, a suPAR level higher than 3531pg/ml could be a useful indicator of a high degree of suspicion of FSGS.
Conflicts of interest
The authors declare that they have no conflicts of interest related to the contents of this article.
Table 1. Clinical-demographic variables in the patient sample studied
Table 2. Variables associated with circulating levels of soluble urokinase-type plasminogen activator receptor. ANOVA
Figure 1. Levels of soluble urokinase-type plasminogen activator receptor by glomerular disease type
Figure 2. Correlations between serum levels of soluble urokinase-type plasminogen activator receptor, clinical and biochemical variables in the sample
Figure 3. Soluble urokinase-type plasminogen activator receptor values for identifying patients with idiopathic focal segmental glomerulosclerosis
Figure 4. ROC curve of soluble urokinase-type plasminogen activator receptor values to distinguish between focal segmental glomerulosclerosis and minimal change disease