Anti glomerular basement membrane disease (AGBM) is an autoinmune disorder characterised by the presence of anti-glomerular basement membrane (Anti-GBM) antibodies, alveolar hemorrhage, necrotizing glomerulonephritis, and linear deposition of immunoglobulins through direct inmunofluorescence. Genetic predisposition, among other factors, plays an important role in the development of the disease. Previous studies have shown that HLA-DR15 and HLA-DR4 increase the risk of presenting it, while HLA-DR1 and HLA-DR7 protect against its development.
We describe the first case of two non-twin siblings with AGBM and identical HLA, with HLA-DR4 as risk factor and HLA-DR7 as protection factor. We propose the importance of analysing HLA in siblings of patients with AGBM, to determine the degree of genetic susceptibility and to carry out a close follow-up on them, with the aim of achieving an early diagnosis and treatment in case of presenting the disease.
La enfermedad antimembrana basal glomerular (EMBG) es un trastorno autoinmune caracterizado por la presencia de anticuerpos anti-membrana basal glomerular (AMBG), hemorragia pulmonar, glomerulonefritis necrotizante y depósito lineal de inmunoglobulinas en inmunofluorescencia directa. La predisposición genética, entre otros factores, posee un papel importante en el desarrollo de la enfermedad. Estudios previos han demostrado que el HLA-DR15 y HLA- DR4 se asocian con mayor riesgo de presentarla, mientras que el HLA-DR1 y HLA-DR7 han demostrado ser factor de protección frente a su desarrollo.
Describimos el primer caso de dos hermanos no gemelos con EMBG con tipaje HLA idéntico, con factor de riesgo HLA-DR4 y factor de protección HLA-DR7. Planteamos la importancia de analizar el tipaje de histocompatibilidad en hermanos de pacientes con EMBG, para determinar el grado de susceptibilidad genética y plantear en ellos un seguimiento estrecho, con el objetivo de lograr un diagnóstico y tratamiento precoces en caso de presentar la enfermedad.
Anti-glomerular basement membrane disease (anti-GBM disease) is a rare autoimmune disorder, with an incidence of 0.5–1 cases per million population and a slight predominance in males. It is characterised by the presence of anti-glomerular basement membrane (anti-GBM) antibodies, pulmonary haemorrhage, necrotising glomerulonephritis and linear immunoglobulin deposition in direct immunofluorescence in renal biopsy1. Anti-GBM antibodies are directed against the NC1 domain of the collagen type IV alpha 3 chain, which is expressed in the glomerular basement membrane (GBM) of the kidney and the basement membrane of the pulmonary alveoli, although antibodies against other different antigenic domains have also been described2. Although the pathogenesis is not fully understood, environmental exposure, genetic predisposition and the presence of autoreactive T cells play an important role in the development of this disease3. There have been isolated reports of familial cases without any specific genetic alteration. However, it has been found that certain histocompatibility antigens such as HLA-DR15 and HLA-DR4 increase susceptibility to the disease, and that others such as HLA-DR7 and HLA-DR1 are protective factors against it4,5.
We report the first case of two non-twin siblings with anti-GBM disease, with identical HLA and HLA-DR7 as protection factor. Both were smokers and shared HLA-DR4. This suggests that it is likely that the coexistence of environmental factors, such as smoking, and the HLA-DR4 risk factor, does not prevent that the presence of HLA-DR7 protects against the development of the disease. It is therefore important to perform histocompatibility typing in siblings of patients with anti-GBM disease to determine the degree of susceptibility and consider whether or not they require close follow-up aiming of achieve early diagnosis and treatment should the disease develop.
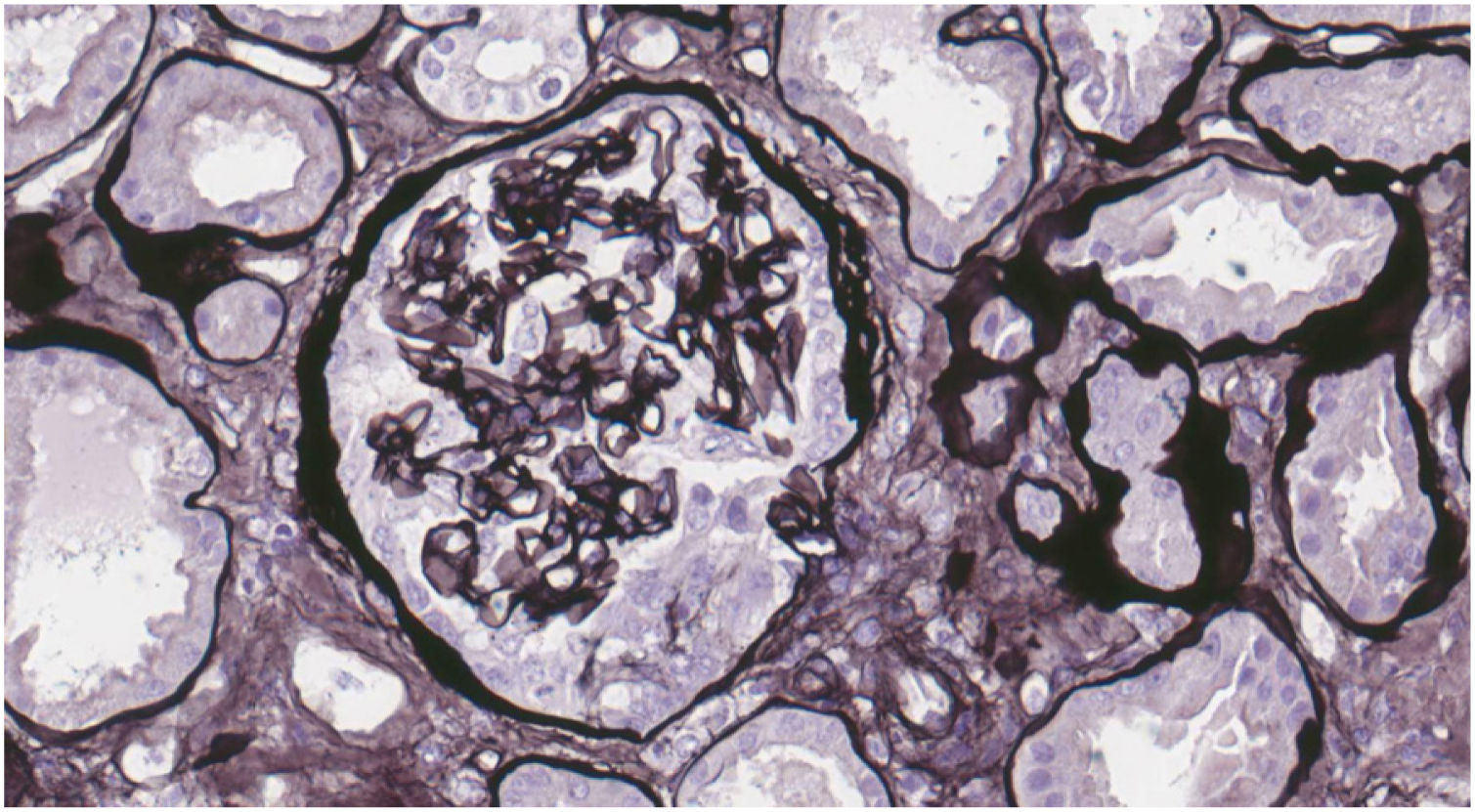
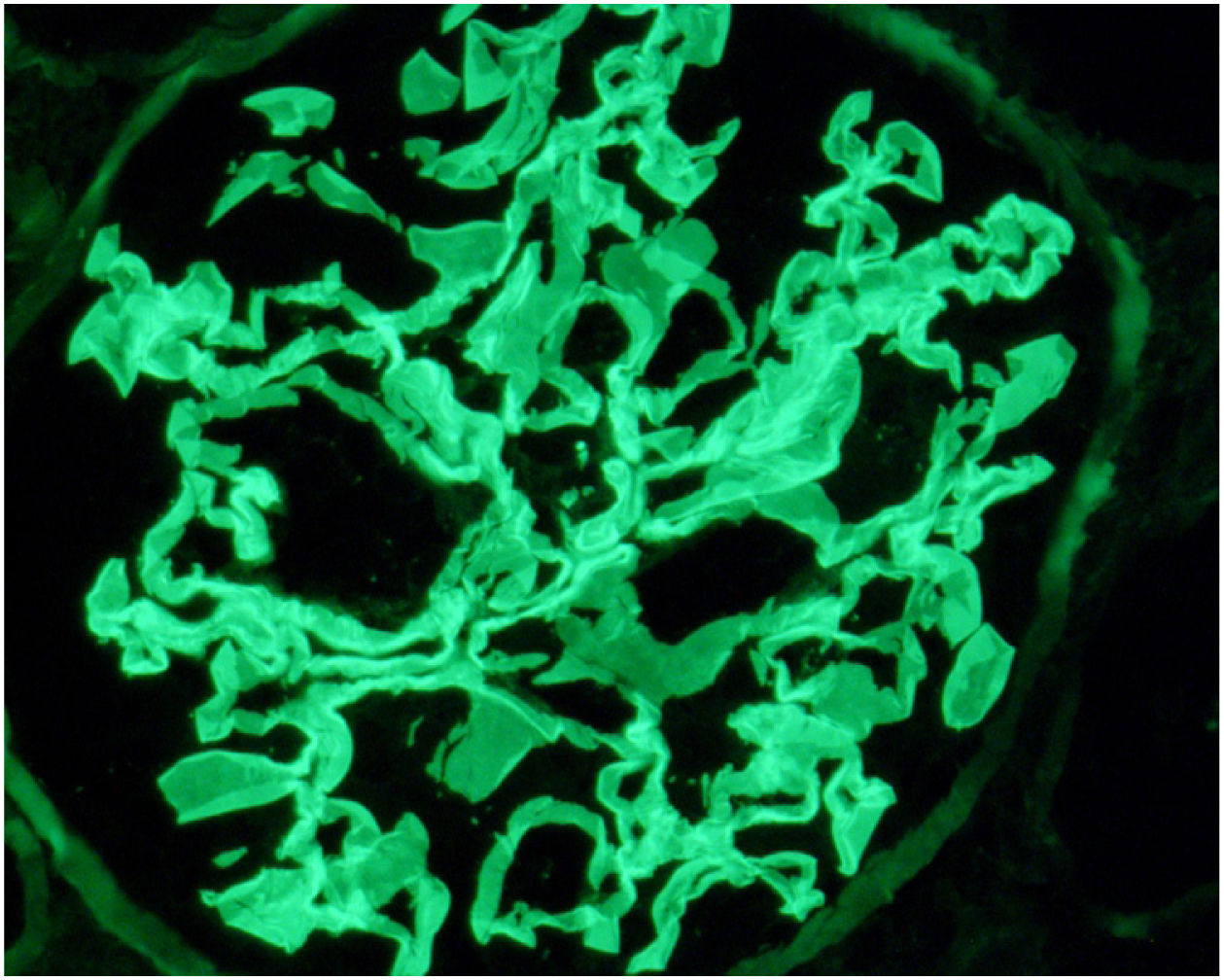
Case reportWe present the case of a 19-year-old male, an active smoker with a history of attention deficit hyperactivity disorder. He consulted the Hospital Emergency Service in August 2017 with a one-week history of coughing and dyspnoea. On physical examination he present crackles in both lungs and chest X-ray showed bilateral interstitial infiltrates. He was admitted with suspected bilateral pneumonia. At 48h, he had a progressive decrease in diuresis and haemoptysis. Arterial blood gases showed respiratory failure, for which he required orotracheal intubation with invasive mechanical ventilation and he was transfered to the Intensive Care Unit (ICU). Blood tests showed haemoglobin 8.6mg/dl, creatinine 4.35mg/dl and urea 250mg/dl. Urinalysis showed macrohaematuria and protein/creatinine ratio 3.5g/g. We requested a glomerular study which was negative for anti-GBM antibodies, with all other findings being normal or negative (complement, immunoglobulins, proteins, antinuclear antibodies [ANA], antineutrophil cytoplasmic antibodies [ANCA] and serology for human immunodeficiency virus [HIV], hepatitis B virus [HBV] and hepatitis C virus [HCV]). Fibre-optic bronchoscopy was performed and the findings were consistent with alveolar haemorrhage, diagnosing pulmonary renal syndrome. We started immunosuppressive therapy with intravenous boluses of methylprednisolone 1g a day for three consecutive days, and oral prednisone 1mg/kg/day, intravenous cyclophosphamide every three weeks, dose adjusted to glomerular filtration rate (GFR), plasma exchange (16 sessions in total), and renal replacement therapy with haemodialysis was required. A renal biopsy was performed after discharge from ICU once the patient was stabilised. Optical microscopy showed 28 glomeruli, none of them sclerosed. A circumferential epithelial crescent was detected in one of them (Fig. 1). Direct immunofluorescence showed an intense positive linear pattern for IgG, Kappa and Lambda, and mesangial and parietal granular pattern for C3 (Fig. 2). Occasional reduplications of the basement membrane of nonspecific appearance were observed on electron microscopy. With these findings, anti-GBM disease was diagnosed. After several weeks of hospitalisation, the patient developed a number of complications, including secondary gastrointestinal infection due to cytomegalovirus, and severe leucopenia, for which cyclophosphamide was switched to rituximab. He had two doses of 375mg/m2/week, but after another episode of alveolar haemorrhage, induction treatment with cyclophosphamide was completed. He received a total of six cycles of cyclophosphamide (total dose 6200mg), with remission of the signs and symptoms and laboratory abnormalities, and he recovered normal renal function which is maintained.
Over a year later, in October 2018, the sister of the above case went to the Emergency Service. She is a 23-year-old woman, an active smoker with no other relevant previous medical history. She complained of a week-long history of right-side abdominal pain, vomiting and asthenia. On physical examination, she was found to have crackles in both lung bases and oedema in her lower limbs. Blood tests showed haemoglobin 8.4mg/dl, creatinine 6.27mg/dl and urea 143mg/dl. Urinalysis showed severe haematuria, moderate pyuria and protein/creatinine ratio 0.90g/g. Chest X-ray showed bilateral interstitial infiltrates. We completed investigations with the glomerular function protocol (complement, immunoglobulins, proteins, ANA, ANCA, anti-GBM antibodies and HIV, HBV, HCV serology) with normal or negative results. We were unable to perform fibre-optic bronchoscopy due to the patient's refusal and intolerance, but considering the family history and symptoms suggestive of pulmonary renal syndrome, she was started on early treatment with plasmapheresis (she had a total of nine sessions), intravenous boluses of methylprednisolone 1g for three consecutive days, followed by oral prednisone 1mg/kg/day and intravenous cyclophosphamide every three weeks, dose adjusted to GFR. A renal biopsy was performed once the patient had stabilised. Optical microscopy showed 35glomeruli, none of them sclerosed. In five glomeruli, rupture of the glomerular basement membrane with circumferential epithelial crescents was detected. The interstitium showed no fibrosis or inflammation. Direct immunofluorescence showed an intense positive linear pattern for IgG Kappa and Lambda, and discontinuous granular and linear morphology of C3. Electron microscopy produced no relevant findings. With these findings, anti-GBM disease was diagnosed. The patient received six cycles of cyclophosphamide (total dose 6700mg) with an adequate response to immunosuppressive therapy, remission of her signs and symptoms and laboratory abnormalities, and return to normal renal function, which has remained stable to date.
Both cases fully recovered renal function, probably due to the rapidity to establish treatment and the fact that the limited number of crescents found in the renal biopsy. As both patients had the same disease and shared similar risk factors, it was decided to obtain HLA typing, which showed the same degree of histocompatibility between the two non-twin siblings (mismatch 0 of 6): A*02, A*23, B*35, B*44, DRB1*04, DRB1*07.
DiscussionAnti-GBM disease occurs in a genetically predisposed individual after an environmental factor triggers the disease. There is a lack of communication between the innate and adaptive immune systems and an aberrant immune response5. Moreover, the mechanisms for anti-GBM antibody formation in anti-GBM disease are not fully understood; it is suggested that autoreactive T cells may play an important role in the pathogenesis. Therefore, genetic susceptibility together with environmental factors and loss of immune regulation contribute to the development of anti-GBM disease1,3.
The environmental stimuli contributing to the development of anti-GBM disease include smoking and hydrocarbon fumes, and these may be the environmental triggers responsible for activating the pathogenesis in genetically predisposed patients5. In our case, smoking was a common environmental factor in both siblings. It is speculated that inhaled toxins promote anti-GBM antibody production by exposing collagen epitopes in damaged alveoli5. Another hypothesis suggests that smokers have high levels of thiocyanate in their blood, which would inhibit the activity of peroxidasin, an enzyme that catalyses crosslinking in type IV collagen hexamers of GBM, which would then promote the access of anti-GBM immunoglobulins to the epitopes in the NC1 domain of the α3 chain6.
The genes of the major histocompatibility complex are the most closely related to the genetic susceptibility of this disease. HLA class II alleles play a central role in the regulation of the immune response. However, the underlying mechanism is still not fully understood. HLA association in anti-GBM disease is thought to reflect the ability of certain class II molecules to bind and present antigens to T-cell receptors, contributing to the production of specific antibodies7. Previous studies have shown that the HLA-DRB1*15:01 haplotype (HLA-DR15 serotype), HLA-DRB1*04 (HLA-DR4 serotype) are associated with an increased risk of developing anti-GBM disease. By contrast, HLA-DRB1*01 haplotype (HLA DR1 serotype) and HLA DRB1*07 (HLA-DR7 serotype) have been shown to protect against developing anti-GBM disease1,4,5. In our case, both siblings developed the disease despite having a protective factor, HLA DR7. It is hypothesised that HLA DR15 increases the susceptibility of having the disease as both patients had peptides of the collagen type IV alpha 3 chain in a configuration that activates proinflammatory T cells8. Furthermore, Phelps et al. suggest that the mechanism by which HLA DR1 and DR7 confer a protective effect is the result of capturing the peptides of the collagen type IV alpha 3 chain, preventing their presentation linked to HLA DR159.
A number of cases of familial anti-GBM disease syndromes with similar HLA have been reported in the literature. Due to the low incidence of anti-GBM disease, however, it has not been possible to determine the exact genetic inheritance that may predispose to the disease in affected families10–14.
As in our case, some of the cases reported affect non-twin siblings. Durcan et al.12 report the case of a 71-year-old woman and her 60-year-old brother with anti-GBM disease. Both were former smokers and in HLA typing both had two risk factors (DRB1*15:01 and DRB1*04:03) and no protective factors. Li et al.13 reported the case of a 49-year-old woman with anti-GBM disease, homozygous for DRB1*15:01, and her sister who, at the age of 29, developed the same disease and died during admission; this meant they were unable to either obtain HLA typing or subsequently determine whether or not she had risk or protective HLA for suffering from anti-GBM disease. Angioi et al.14 report the case of a 65-year-old woman with anti-GBM disease, with two brothers who had the disease in adolescence. One of them died, while the other developed end-stage chronic kidney disease and received a kidney transplant. All three were smokers and had been exposed to kerosene. HLA typing of the index case and the brother was performed; they had identical HLA (A1, A3, B7, DQ6, DR15, DR51), both were homozygous for the risk factor HLA-DR15, with no protective factor.
The rest of the cases reported are, for the most part, prior to 1990. They include two members of each family, including twins, siblings and first cousins. Some share environmental exposure factors10,14, but given their publication date, they do not have HLA typing or immunofluorescence diagnosis14.
Our case as well as all those described in the literature were found to have HLA risk for developing the disease. However, none was found to have protective HLA. To the best of our knowledge we reporting the first case of non-twin siblings with anti-GBM disease with identical HLA and risk factor HLA-DR4, despite having the protective factor HLA-DR7. In other words, although HLA-DR7 is considered protective against anti-GBM disease, in our case it was not enough to prevent the development of the disease, probably due to the presence of risk HLA, the same environmental exposure, smoking, and the coexistence of identical HLA.
We advocate the importance of analysing histocompatibility typing in siblings of patients with anti-GBM disease, with the aim of detecting HLA matches or antigens associated with a higher risk of developing the disease. This will facilitate close follow-up and ensure early diagnosis and treatment, thereby improving the prognosis of the disease.
FundingNo funding was received for this study.
Conflicts of interestThe authors declare that they have no conflicts of interest.