Heparin is the most commonly used anticoagulant to maintain filter and extracorporeal circuit patency during hemodialysis therapy. Heparin-induced thrombocytopenia (HIT), which results in thromboembolism, is a potentially critical complication of heparin therapy. Although the incidence of HIT varies depending on the type of heparin and patient population, no studies evaluated the incidence of HIT among incident hemodialysis patients anticoagulated with low molecular weight heparin (LMWH).
In this single-center, retrospective, observational study, we enrolled consecutive incident hemodialysis patients from January 2013 to December 2016. LMWH (Parnaparin Sodium) was the primary anticoagulant for hemodialysis and all temporary or chronic hemodialysis was started in the inpatient setting at our institute. Incident hemodialysis patients were defined as those who had started hemodialysis during hospitalization and who received regular hemodialysis after discharge. We excluded the following: (1) temporary hemodialysis; (2) administration of unfractionated heparin (UFH); and (3) initiating hemodialysis in the intensive care unit. To identify patients with HIT, we reviewed all patients who conducted an antigen immunoassay for HIT and/or dropped platelet count ≥50% from baseline. The antigen immunoassay was performed using a latex agglutination test (HemosIL HIT-Ab®, Instrumentation Laboratory, Bedford, MA, USA)1 at the discretion of attending doctors. All patients review were conducted by two experienced physicians (Y.D. and K.K.). Given the lack of functional assay results in most patients, functional assay was not required for the diagnosis of HIT in this study. Based on the Ethical Guidelines for Medical and Health Research Involving Human Subjects in Japan, the opt-out approach through the hospital website was adapted instead of obtaining informed consent. The study was approved by the institutional ethics committee (approval number 737) and conducted in accordance with the Declaration of Helsinki.
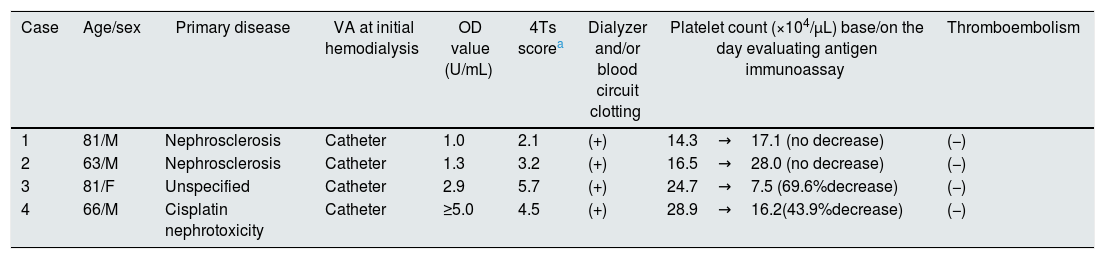
A final total of 330 incident hemodialysis patients anticoagulated with LMWH were included in this study (Table 1). During the study period, antigen immunoassay was performed in 11 patients and another 9 patients with over 50% decrease in platelet count were suspected of HIT. Among those antigen immunoassay performed, HIT was ruled out based on negative results with optical density<0.6 (cutoff<1.0) in 7 patients. The remaining 4 patients with positive results are shown in Table 2. We ruled out HIT in Case 1 and 2 based on low 4Ts score and diagnosed Case 3 and 4 as definitive HIT. In Case 3, emergent hemodialysis was performed because of volume overload. After initiating hemodialysis, dialyzer and blood circuit clotting became apparent with each session accompanied by thrombocytopenia. Positive result of antigen immunoassay was confirmed on day 11. Case 4 had acute kidney injury due to cisplatin for gastric cancer and emergent hemodialysis was performed. On day 8, the dialysis circuit was completely coagulated just after the initiation of hemodialysis and antigen immunoassay was a positive result. Nine patients with over 50% decrease in platelet count but without antigen immunoassay measurement were not considered as HIT due to obvious other causes of thrombocytopenia such as infection, hematologic disease and drugs. Finally, the incidence of HIT in this setting was 0.6%.
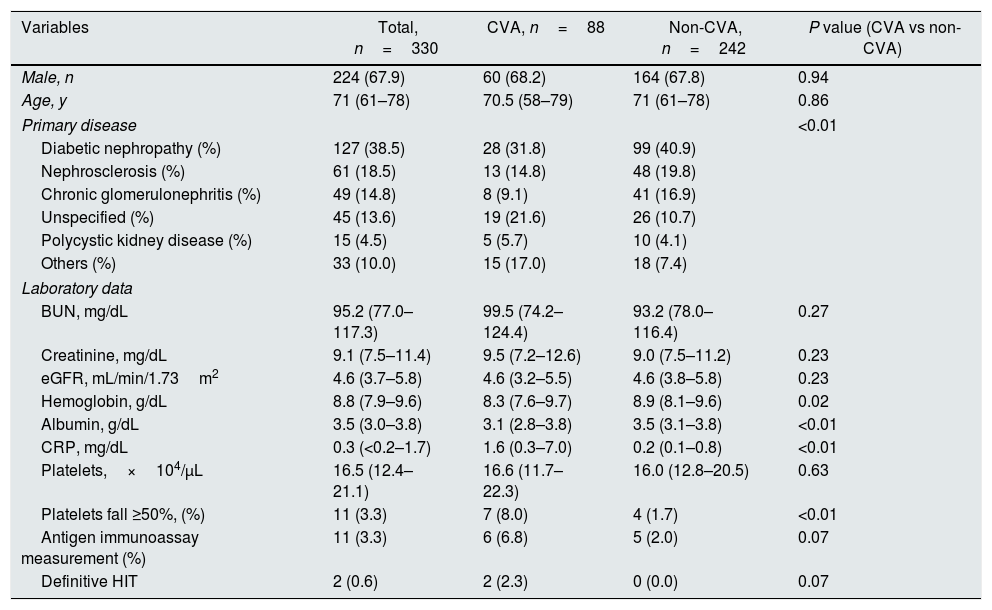
Characteristics of the study population according to vascular access type at hemodialysis initiation (n=330).
| Variables | Total, n=330 | CVA, n=88 | Non-CVA, n=242 | P value (CVA vs non-CVA) |
|---|---|---|---|---|
| Male, n | 224 (67.9) | 60 (68.2) | 164 (67.8) | 0.94 |
| Age, y | 71 (61–78) | 70.5 (58–79) | 71 (61–78) | 0.86 |
| Primary disease | <0.01 | |||
| Diabetic nephropathy (%) | 127 (38.5) | 28 (31.8) | 99 (40.9) | |
| Nephrosclerosis (%) | 61 (18.5) | 13 (14.8) | 48 (19.8) | |
| Chronic glomerulonephritis (%) | 49 (14.8) | 8 (9.1) | 41 (16.9) | |
| Unspecified (%) | 45 (13.6) | 19 (21.6) | 26 (10.7) | |
| Polycystic kidney disease (%) | 15 (4.5) | 5 (5.7) | 10 (4.1) | |
| Others (%) | 33 (10.0) | 15 (17.0) | 18 (7.4) | |
| Laboratory data | ||||
| BUN, mg/dL | 95.2 (77.0–117.3) | 99.5 (74.2–124.4) | 93.2 (78.0–116.4) | 0.27 |
| Creatinine, mg/dL | 9.1 (7.5–11.4) | 9.5 (7.2–12.6) | 9.0 (7.5–11.2) | 0.23 |
| eGFR, mL/min/1.73m2 | 4.6 (3.7–5.8) | 4.6 (3.2–5.5) | 4.6 (3.8–5.8) | 0.23 |
| Hemoglobin, g/dL | 8.8 (7.9–9.6) | 8.3 (7.6–9.7) | 8.9 (8.1–9.6) | 0.02 |
| Albumin, g/dL | 3.5 (3.0–3.8) | 3.1 (2.8–3.8) | 3.5 (3.1–3.8) | <0.01 |
| CRP, mg/dL | 0.3 (<0.2–1.7) | 1.6 (0.3–7.0) | 0.2 (0.1–0.8) | <0.01 |
| Platelets,×104/μL | 16.5 (12.4–21.1) | 16.6 (11.7–22.3) | 16.0 (12.8–20.5) | 0.63 |
| Platelets fall ≥50%, (%) | 11 (3.3) | 7 (8.0) | 4 (1.7) | <0.01 |
| Antigen immunoassay measurement (%) | 11 (3.3) | 6 (6.8) | 5 (2.0) | 0.07 |
| Definitive HIT | 2 (0.6) | 2 (2.3) | 0 (0.0) | 0.07 |
Data are presented as number (percentage) or median (25th, 75th percentile). Differences between patients with or without CVA were compared using Student's t test or Wilcoxon signed-rank test for continuous variables, and the Chi squared test or Fisher's exact test for categorical variables, as appropriate. CVA, Catheter vascular access; BUN, blood urea nitrogen; eGFR, estimate glomerular filtration rate; CRP, C-reactive protein; HIT, heparin-induced thrombocytopenia.
Clinical profiles of patients with positive antigen immunoassay.
| Case | Age/sex | Primary disease | VA at initial hemodialysis | OD value (U/mL) | 4Ts scorea | Dialyzer and/or blood circuit clotting | Platelet count (×104/μL) base/on the day evaluating antigen immunoassay | Thromboembolism |
|---|---|---|---|---|---|---|---|---|
| 1 | 81/M | Nephrosclerosis | Catheter | 1.0 | 2.1 | (+) | 14.3→17.1 (no decrease) | (−) |
| 2 | 63/M | Nephrosclerosis | Catheter | 1.3 | 3.2 | (+) | 16.5→28.0 (no decrease) | (−) |
| 3 | 81/F | Unspecified | Catheter | 2.9 | 5.7 | (+) | 24.7→7.5 (69.6%decrease) | (−) |
| 4 | 66/M | Cisplatin nephrotoxicity | Catheter | ≥5.0 | 4.5 | (+) | 28.9→16.2(43.9%decrease) | (−) |
M male, F female, VA vascular access, OD optical density.
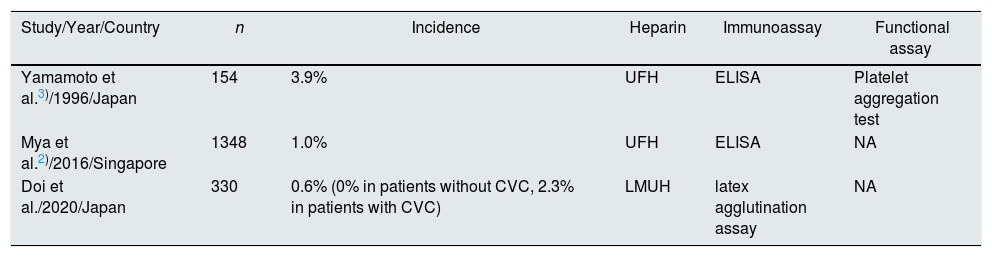
Our result showed a mild decrease in HIT incidence with LMWH, compared to previous reports with UFH (3.9% and 1.0%, Table 3).2,3 But small sample size, low HIT incidence and the difference in patient population and clinical practice pattern make a direct comparison difficult. Most interestingly, no patients with non-catheter vascular access (CVA) had HIT, but 2.3% of patients with CVA developed HIT, resulting in a marginally significant difference (p=0.07). A previous paper reported that all 13 patients with HIT used CVA in incident hemodialysis patients.2 A recent case series from Spain also reported that all 5 hemodialysis patients of HIT used CVA as vascular access.4 Underlying mechanisms are uncertain, but it may be reasonable to consider that physical status requiring temporary central venous catheter (CVC) for emergent hemodialysis and/or complications of CVC play an important role in HIT development. The inflammation seems to facilitate HIT antibody formation and clinical HIT.5 Those requiring emergent hemodialysis tend to have extracellular fluid overload, which is a strong association between malnutrition–inflammation complex syndrome.6 In line with this context, our data showed lower serum albumin and higher C-reactive protein in the CVA group. In addition, catheter-related bloodstream infection (CRBSI) is a frequent, fatal complication related to CVA,7 and we have recently reported that CRBSI triggered the development of HIT.8 Furthermore, CVC itself reported to be a contributor to increased inflammation independently of infection.9
Incidence of HIT as reported in incident hemodialysis patients.
| Study/Year/Country | n | Incidence | Heparin | Immunoassay | Functional assay |
|---|---|---|---|---|---|
| Yamamoto et al.3)/1996/Japan | 154 | 3.9% | UFH | ELISA | Platelet aggregation test |
| Mya et al.2)/2016/Singapore | 1348 | 1.0% | UFH | ELISA | NA |
| Doi et al./2020/Japan | 330 | 0.6% (0% in patients without CVC, 2.3% in patients with CVC) | LMUH | latex agglutination assay | NA |
CVA, catheter vascular access; UFH, unfractionated heparin; LMWH, low molecular weight heparin; ELISA. enzyme-linked immunosorbent assay; NA, not applicable.
In conclusion, the incidence of HIT among incident hemodialysis patients anticoagulated with LMWH was 0.6%. CVA at the initiation of hemodialysis therapy might be a risk for the development of HIT. Given that the incidence of HIT is very low, large prospective observational studies are warranted to validate these findings.
Authors’ contributionsStudy concept and design by Doi Y and Ishihara T; writing by Doi Y, and Sugawara A; data collection by Doi Y, and Sugioka S; analysis and interpretation of data by Doi Y, Inoue Y, Arisato T, Koga K, and Nishioka K; critical revision of the manuscript for important intellectual content by Sugawara A.
FundingNone.
Conflict of interestsThe authors declare that they have no competing interests.
We thank Takeda T for secretarial assistance.