This review discusses the diagnostic value of urinary parameters in the setting of advanced chronic kidney disease and we present the key concepts that summarize the suggestions of the manuscript.
Urinary volumeThe amount of fluid intake may be a non-established risk factor for CKD. For these patients, a urinary output ≥2–3l/day is a reasonable proposal. This recommendation is not applicable to patients with cardiorenal syndrome or fluid overload risk.
NaurThis determination is very useful to monitor salt intake. Reducing urinary Na<120mEquiv./day (≅salt intake≤5–6g) is a reasonable objective.
Urinary urea nitrogen (UUN)This parameter is useful to estimate protein intake (Maroni BJ equation). A protein intake between 48–72g (0.8–0.9g/kg/day according to weight) is equivalent to UUN 7–10g/day approximately.
Acid load and potassiumAcid load reduction may be an additional strategy in the nutritional management of this population. It may be estimated indirectly from a diet survey or by measuring the elimination of UUN and Kur. The limits of this recommendation have not been established, but we propose a cautious and prudent diet of fruit and vegetables.
PhosphorusThere is a significant positive correlation between phosphorus and protein, both in dietary records and urine elimination. Based on this information, we suggest a urinary P excretion<800mg/day or<600mg/day for patients with GFR<25ml/min or <15ml/min, respectively.
ConclusionUrinary parameters provide sensitive and useful knowledge for clinical practice, provide information about the dietary habits of patients and the adherence to our recommendations.
En esta revisión discutimos el valor diagnóstico de los parámetros urinarios en la enfermedad renal crónica avanzada y exponemos los conceptos clave que resumen las sugerencias del manuscrito.
Volumen de orinaLa cantidad de líquido ingerida puede ser un factor de riesgo de enfermedad renal crónica no establecido. Alcanzar una diuresis ≥2-3l al día es una propuesta razonable, no aplicable al síndrome cardiorrenal y riesgo de retención hidrosalina.
Naurdeterminación útil para vigilar la ingesta salina. Reducir la natriuresis<120mEq/d (≅ingesta sal≤5-6g) es un objetivo razonable.
Nitrógeno ureico urinario (NUU)Útil para estimar la ingesta proteica (ecuación de Maroni). Una ingesta proteica entre 48-72g (0,8-0,9g/kg/día según peso) ≅NUU 7-10g/día aproximadamente.
Carga ácida y potasioLa reducción de la carga ácida puede ser una estrategia adicional en el manejo nutricional de esta población. Puede estimarse de forma indirecta desde la encuesta dietética o midiendo la eliminación de NUU y Kur. Los límites en la recomendación no están establecidos; proponemos una liberación prudente de verduras y frutas.
FósforoExiste estrecha asociación entre proteínas y fósforo, tanto en registro dietético como eliminación urinaria. El análisis combinado sugiere que para pacientes con FG<25 mil/min, una fosfaturia<800mg/día, y con FG<15 mil/min, una fosfaturia<600mg son objetivos razonables.
ConclusiónLos parámetros urinarios proporcionan conocimiento sensible y de utilidad para la práctica clínica; aportan información de los hábitos dietéticos del paciente y de la adherencia a nuestras recomendaciones.
There is a notable gap in the guidelines for clinical decisions1–3 regarding the usefulness of urinary parameters in advanced chronic kidney disease (CKD) not only as a tool for diagnostic approximation, but also to assess adherence to medical recommendations. The volume of urine, electrolytes and urea nitrogen can be very useful in daily practice. Will analyze the potential use of these determinations in patients with CKD stages 3–5. A correct collection of 24h urine is one of the main drawbacks for the adequate interpretation of these measurements. Although we will always have a certain margin of uncertainty, the urinary values of creatinine, sodium or urea nitrogen within a adequate range may be helpful in accepting as correct a 24-h urine collection.
The study of tubular function or electrolyte imbalances is beyond the objective of this manuscript.
Volume and osmolarity of urineThe measurement of 24-h volume of urine provides indirect information on the water management by the kidney and the patient's hydration status. Also, urinary osmolarity (U Osm) reveals the kidney ability to concentrate urine. It is striking that the KDIGO2 Guidelines do not provide guidelines regarding water intake and desirable diuresis in the patient with advanced CKD (ACKD). In no case it is mentioned the potential usefulness of the measurement of U Osm.
Although there is no conclusive evidence, studies in animals and in patients suggest that the amount of fluid ingested may be a risk factor for CKD that has not been clearly established.4 Recently, 2 excellent reviews5,6 have discussed the mechanisms whereby low fluid intake may have adverse effects on CKD. Likewise, another three clinical studies support the benefits of high water intake, both in population studies and in patients with CKD.7–9 The high prevalence of CKD in Central America, known as Mesoamerican nephropathy, is a clear example of kidney damage caused by dehydration.10
Data from the literature suggest that the adverse renal effects of insufficient hydration could be mediated by the increase in vasopressin (ADH).11,12 In polycystic kidney disease, this effect has been well documented, demonstrating that the increase in water intake delays the growth of cysts in animals, through the direct suppression of ADH.12–14
The following theoretical analysis is intended to provide a reasonable argument for answering the question “Dr. How much should I drink?”: A standard diet generates about 650mOsm of solutes, which must be excreted in the urine.15 If it is assumed that the maximum capacity to concentrate the urine is up to 1200mOsm/kg, at least 500ml of urine will be necessary to eliminate such a solute load.15,16 In normal circumstances the urinary Osm is 2–3 times higher than in plasma, which correspond to a 1.2–2.0l daily urine volume in healthy individuals.6
This information may provide reasonable arguments to estimate the minimum necessary diuresis capable to eliminate the solute load in the ACKD patient.
With a significant renal damage, the ability to concentrate urine is reduced and the urine becomes isosthenuric (250–300mOsm/kg). If the mandatory urinary volume is obtained by dividing the daily osmolar excretion by the maximum urinary Osm, a diuresis of at least 2l would be necessary to eliminate the usual solute load. This is achieved, depending on the extrarenal losses, with a liquid intake between 2.5 and 3.5l per day.6 In general, we may assume that 20% of fluid intake comes from solids and 80% from water and other ingested liquids.15
A generous water intake, at least to eliminate the osmotic load, may help preserve kidney function in patients who maintain the ability to generate high volume of urine. The benefits of liquid intake, beyond the demands of thirst, could be key to delay the progression of CKD.5,6,17 This aspect is especially important in elderly patients with a natural loss of sensation of thirst. In fact, it is not unusual to observe an increase in serum creatinine during periods of heat, or during episodes that are associated to dehydration (fever, diarrhea, vomiting, etc.) and that recover after an adequate liquid intake. Given these eventualities, we must also advise the patient for a temporary reduction or suspension of diuretics, blockers of the renin–angiotensin–aldosterone system (RAAS) or antihypertensive drugs, as measures to prevent an acute, possibly irreversible, deterioration of renal function.
Although the capacity to generate high diuresis is maintained until advanced stages of CKD, this recommendation must be handled with maximum caution and individualized. It is not applicable to patients with cardiorenal syndrome or at risk of volume expansion. In addition, a forced water intake may exceed the capacity of dilution of the kidney, and induce hyponatremia.18 Therefore, serum and urinary parameters should be monitored in order to prevent both hyponatremia and dehydration, which is more common in summer and in elderly patients, who are the majority of patients with ACKD coming to the outpatient clinics.
The key concepts of these thoughts are presented in Table 1.
Hydration: key concepts.
| In ACKD, the ability to generate a high volume of urine is preserved until advanced stages |
| The amount of fluid ingested may be a risk factor for CKD that is not established |
| The intake of water, beyond that necessary to eliminate the osmotic load, can help to preserve kidney function. Achieving a diuresis of 2–3l liter a day, or even more, is reasonable. |
| This recommendation should be used with caution and individualized: |
| Not applicable to patients with cardiorenal syndrome, with risk of extracellular volume expansion and congestive heart failure |
| Forced intake may exceed renal capacity to dilute the urine and produce hyponatremia |
| Complementary control measures (in prevention of the adverse effects mentioned) |
| Periodical measurement of the 24h diuresis by the patient and weight control |
| Monitoring serum and urine Na in the outpatients visits. |
| Measurement of urinary Osmolarity helps to assess the ability to concentrate urine. |
| Emphasis should be placed on the prevention of dehydration, which is more frequent during the summer and older patients, and in the self-control of medication by temporarily reducing or suspending diuretics and blockers of the renin-angiotensin-aldosterone or other antihypertensive medication if appropriate. |
The restriction of Na is an unequivocal recommendation to control of hypertension and volume retention.19 Clinical guidelines1,3 recommend an intake of Na <2g/day, which corresponds to 5g of salt, unless there is a specific contraindication. However, there is no mention of the importance of monitoring the adherence to this prescription and how to do it.
Urinary Na determination is the easiest way to monitor saline intake.20–24 Ancient work has verified that Na is absorbed quickly and effectively in the intestinal mucosa (a 98%), therefore, in a situation of homeostasis urine Na is a good indicator of the intake.21,22 It is postulated that the use of diuretics can be a limitation for the value of natriuresis. However, in a patient on balance, the urinary Na reasonably reflects saline intake.
The Na intake is calculate using a simple equation: Na in mEq/urine 24h×0.058=daily salt intake (g).
In the “real world” the usual intake of salt in the population is 9–12cg/day, and even higher in many regions, which corresponds to a urinary elimination of Na of 160–200mEq./day or even more.25 Achieving a saline intake of 5–6g per day (80–100mEq of Na/day), is possible, but very difficult to achieve even in adherent patients.
Table 2 shows examples of the correspondence between saline intake and urinary Na, and it should be emphasized the importance of measuring Na in 24-h urine to optimize patient management and adherence to our recommendations.
Salt intake and urinary Na.
| The guidelines recommend a Na intake of <2g/day, which is equivalent to 5g of salt (NaCl), unless there is any contraindication. |
| The measurement of urinary Na is a useful to monitor saline intake |
| Calculation: mEquiv. of Na in urine 24h urine×0.058=g of salt intake (1g ClNa⇔17mEquiv. Na) |
| Examples |
| 5g sal→<1.9gNa→<90mEq. |
| 7g sal→2.7g Na→120mE |
| 10g sal→3.9g Na→170mEq |
| 15g sal→5.9g Na→255mEq |
| The resulting protein intake is between 48 and 72g (0.8–0.9g/kg/day, for a weight range of 60–80kg), corresponds to an approximate UUN elimination of 7–10g/day. |
The benefits of dietary protein restriction in CKD are reasonably accepted. The aim is to delay CKD progression of – especially if proteinuria is present – but there are also beneficial in other nutritional aspects.26–31 This concept would not be applicable to patients with polycystic kidney disease, very doubtful in non-proteinuric nephropathies, but especially beneficial in diabetic nephropathy. With the progression of renal damage, nitrogen products accumulate proportionally to the loss of renal function. In fact, protein restriction has prevailed for decades and has been the cornerstone of treatment when dialysis did not exist.
Collecting information from previous studies,32 the KDIGO Guidelines recommend a protein intake of 0.8g/kg/day in adults with GFR<30ml/min/1.73m2, this is being assumed in most guidelines and consensus documents.33 Based on our experience, we consider that achieving a protein restriction of 0.8–0.9g/kg/day is a reasonable objective that is possible to achieve.
Now, returning the ACKD scenario, we would like to point out the importance of knowing the patient's protein intake and adherence to our recommendations. It has been verified that the determination of urinary urea nitrogen (UUN) is a useful parameter to estimate protein intake. The classic equation described by Maroni et al.34 is useful to evaluate the protein intake in patients in metabolic equilibrium. The protein intake is derived from the estimation of UUN, adjusting for weight and proteinuria: UUN (g)+0.031×weight (kg)+proteinuria (g). This result multiplied by 6.25 gives us approximate information on protein intake. This formula has been reasonably verified by other authors,35 and even with its limitations we consider that it should be mentioned in the clinical practice guidelines. In Table 3, based on unpublished results of our ACKD outpatient clinic, we show the approximate relationship between the values of UUN and protein intake, where we observe that the median of NUU (7.8g) corresponds to a protein intake of 49–55g depending on the weight used in the equation.
Protein intake and urinary urea nitrogen (UUN): key concepts.
| A protein intake of 0.8–0.9g/kg ideal weight/day is a feasible objective that can provide subtle renal protection benefits in patients with GFR<30ml/min |
| The determination of UUN has been verified to be a useful parameter to estimate the protein intake using the Maroni equation34: |
| Protein intake (g)=6.25×(UUN [g]+0.031×weight (kg)+proteinuria [g]) |
| These are the results obtained from our ACKD outpatient clinic in 194 patients with NUU data in 24h urine, weight and proteinuria (g). |
| NUU 24h (g) | Protein intake (g) | |
|---|---|---|
| Median | 7.8 | 49–59 |
| Percentile 25 | 6 | 37–45 |
| Percentile 75 | 9.6 | 60–72 |
| The resulting protein intake is between 48 and 72g (0.8–0.9g/kg/day, for a weight range of 60–80kg), corresponds to an approximate UUN elimination of 7–10g/day. |
The evaluation of the acid load and urinary potassium (K) are not frequently used in clinical practice. However, recent data emphasize the importance of controlling the acid load of the diet as a modifiable risk factor for the progression of kidney disease.36
The endogenous production of acids derives mainly from the intake of proteins and phosphate. The production of alkali comes from organic anions such as citrate and acetate, naturally bound to cations such as K which are found primarily in vegetables and fruits. The difference between the two results in the acid load, which is normally eliminated by the kidneys as ammonium and titratable acid, and in a standard diet is approximately 1mEq/kg/day.
Scialla et al.37 described a direct association between dietary acid load and renal deterioration. In CKD, with the reduction of nephron mass, each of the remaining nephron increases the ammoniogenesis and distal excretion of acid mediated by RAAS and endothelin-1, which results in kidney damage.36,38,39 In fact, the KDIGO Guidelines recommend giving oral bicarbonate supplements in case the serum concentration is less than 22mmol/l.
Based on these premises, it is postulated that the reduction of the acid load may be an additional strategy in the nutritional management of this population. The acid load is determined by the balance of the foods that induce acid (meats, eggs, dairy products, cereals) and those that induce alkali (fruits and vegetables). A greater intake of the latter may contribute to a reduction of the acid load without the need for excessive protein restriction and bicarbonate intake.36,37,40–42 A prudent intake of foods rich in K, may contribute to achieve a healthier diet in this population. This subject has been recently reviewed by Kalantar-Zadeh et al.43
Obviously, in ACKD the risk of hyperkalemia is an important limitation. Currently there are no clinical studies that guarantee the safety of a more free diet in vegetables and fruits in this population. However, in well-nourished patients, with good urinary output and in a non-catabolic state, their judicious intake of fruits and vegetables avoiding prolonged fasting44–46 (an important and frequently forgotten recommendation), may be a measure both nutritionally healthy and renoprotective (37, 47–49). Obviously, diabetic patients or those treated with beta-blockers and/or RAAS blockers will be subject to greater vigilance, especially those who receive K-sparing diuretics.50
The acid load may be estimated indirectly from the intake of nutrients or from the urinary elimination of ions, for which various equations have been postulated36,37,47,48,51; perhaps the simplest is the following:
Acid load (mEquiv./day): −10.2+54.5×(protein intake [g/day]/K intake [mEq/day]).36,37,48
The protein and K intake may be obtained from the elimination of UUN and K (1mEquiv. of K⇔39mg), which, with all its limitations, are considered indirect indicators of protein intake and alkaline load, respectively.37,41,52 Whereas the determination of the UUN to assess protein intake is well established,34,35 the value of the urinary K as indicator of the alkaline load is not that well recognized.
On a standard diet, the range of urinary elimination of K is wide, 50–200mEq/day, which is equivalent to 2000–7800mg/day. In hemodialysis patients the adverse effect of K retention is well known. Kalantar-Zadeh et al.53 suggest that the K intake should be <3g/day; the authors also emphasizes the importance of a healthy diet, with prudent intake of fruit and vegetable, without proposing specific amounts.43
Several authors have explored the impact of acid loading, both in population studies37 and in ACKD patients, even using a diet relatively free on fruits and vegetables.51 The latter described a decrease in the acid load, with a urinary elimination of K of 69.9±12.39mE/day, equivalent to an average of 2800mg/day, without relevant change in serum K concentration (4. 1±0.1mEq/l).
Since the limits in this recommendation have not been established, we propose a prudent intake of vegetables and fruits, around 2800–3000mg of K/day (see http://www.revistanefrologia.com/es-monoografias-nefrologia-dia-articulo-manejo-nutricional-enfermedad-renal-cronica-99), trying to maintain urinary K levels below 70–75mEq/day.37,43,47–49,51,54 The key concepts are presented in Table 4.
Acid charge and urinary K: key concepts.
| The acid load is derived mainly from proteins and phosphate intake, which are more frequent in dairy products, meat products and legumes. The alkaline production comes from anions, linked to cations like K and found primarily in vegetables and fruits. The difference between both is the acid load; that is renal excretion as NH4 and titratable acidity. In a standard diet the acid load is approximately 1mEquiv./kg/day. |
| The nutritional reduction of the acid load may be an additional strategy in the management of ACKD reduce the deterioration renal function. |
| The acid load can be estimated indirectly from the dietary survey or in a simplified way by measuring the elimination of UUN and urinary K. The simplest equation published is the following: |
| Acid load (mEquiv./day): −10.2+54.5 (protein intake [g/day]/K intake [mEq/day]).34,35,45 |
| In well-nourished patients, with enough urinary output and in a non-catabolic state, the judicious and monitored use of a more liberal diet of vegetables and fruits (avoiding prolonged fasting), along with protein restriction is healthy and renoprotective. |
| Since the limits in the recommendation have not been established, we propose to recommend a prudent intake of vegetables and fruits, around 2800–3000mg of K/day that would correspond to a urinary K excretion of less than 70–75mEq/day. |
In the normal adult, the calcium (Ca) balance is relatively neutral when the intake is between 750 and 1740mg/day.55 In this context, the urinary excretion of Ca ranges between 200 and 300mg/day, thus there is variable intestinal absorption from 20 to 60%, depending to a large extent on the amount of circulating levels of 1,25(OH)2D3.
In ACKD, urinary excretion of Ca is much lower, in a range of 20–60mg/day,56–58 although fractional excretion (percentage of filtered Ca that is eliminated in the urine) is slightly elevated (1.1–1.4%, normal <1%). Hill et al.,56 in an interesting study in patients with CKD stages 3–4, described an intestinal absorption of Ca truthfully low, around 17–22%, and urinary Ca excretion of 40–42mg/day.
In ACKD, calciuria is not a parameter that provides relevant information in clinical practice, but it is useful in the experimental setting, especially in studies that involve the balance of Ca and the use of drugs such as calciomimetics, vitamin D analogs or phosphate binders (P).
PhosphateRelation of phosphorus intake and protein intakeStudying P balance bear difficulties, such as inaccuracy in dietary records or variable intestinal absorption. In addition, studies based on dietary surveys were conducted without counting on effect of additives with a high content of absorbable P.59,60
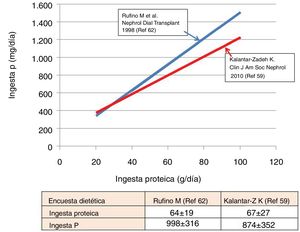
The relationship between P intake and protein intake has been verified both in population studies61 and in hemodialysis patients,59,62 as shown in Fig. 1. Summarizing, roughly, an intake of 60g of proteins entails an ingestion of 750–1000mg of P. In patients with ACKD, with protein restriction diets, an average intake of 40–50g of protein is associated with an obligatory P intake of 640–800mg.62 However, to this values it is necessary to add the content of inorganic P from the additives.
The P/protein ratio in the diets has also been explored by several authors. Morimoto et al.61 described a ratio of approximately 15mg of P/g protein in the diet of healthy adults. The surveys of Kalantar-Zadeh et al.59 in hemodialysis patients reveals values of 13mg of P/g protein (range 10–16); while Noori et al.63 suggest that a quotient greater than 14 (that is, 840mg of P for every 60g of ingested proteins) was associated with an increase in mortality. Therefore, reducing the P/protein ratio in the diet is a desirable goal. Generically, the highest ratio is found in dairy products (15–25mg/g proteins), followed by animal proteins and legumes (10–15mg/g proteins).59 In this sense, it is essential to limit the preservatives, frozen foods and soft drinks with additives that are very rich in easily absorbable inorganic P.59,60
Urinary excretion of phosphorus and urea nitrogenThe next difficulty in estimating the P balance is intestinal absorption. The net absorption is very variable, between 40% and 80%. Therefore, for an intake between 800 and 1600mg/day, the absorption may vary from 320 to 1280mg, being somewhat lower in ACKD and in hemodialysis.64–67
In this sense, the values of urinary P (Pur), along with those of UUN may provide relevant information, since the Pur must reliably reflect the amount P absorbed, not the ingested amount. If we accept that the absorption is of 60–70%, this parameter can be useful to advise the patient in his diet and monitor his adherence.
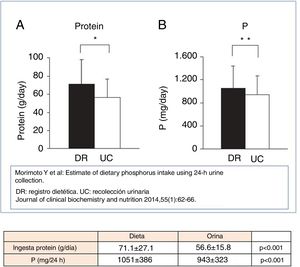
Morimoto et al.61 found in the general population a close relationship between the P and protein intake obtained from the dietary survey and the urinary parameters of P and UUN, respectively (Fig. 2). The latter were derived from the following equation: intake of P=Pur×0.65 (assumes that the intestinal absorption is 65%) and protein intake is calculated from the UUN using the Maroni equation.34 Despite the biases described by the authors (inaccuracy of the dietary record and variable intestinal absorption of P) this analysis highlights the information that can be extracted from the determination of the Pur and UUN.
Association between intake of protein and phosphorus, assessed by dietary record (RD) and urinary collection (UC).
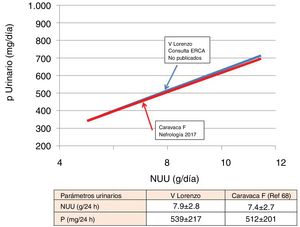
In ACKD it has been possible to verify the close association between the UUN and the Pur. The data published by Caravaca et al.68 can be superimposed to those obtained in our population, as illustrated in Fig. 3. If we assume that the intestinal absorption of P was 60–70%, we can indirectly estimate that the actual intake of P was within a range of 650–900mg. These data are very consistent with those obtained from dietary surveys (Fig. 1) in hemodialysis patients. Slightly higher values were published in CKD E3 patients,69 where a mean Pur of 792mg was collected (95% CI: 646–938).
The information provided by the ratio P/protein in urine described by Caravaca et al.68 is very interesting; it is provides values of 8.22±2.34mg of P/g protein. These values are lower than those published from the dietary registry59 that presented the P ingested, not the absorbed.
Finally, Caravaca et al.68 conducted a novel and very interesting analysis based on the corrected Pur for glomerular filtration rate (GFR). A Pur of 35–40mg for each ml/min of GFR marked the limit for the development of hyperphosphataemia (serum P>4.5mg/dl). That is to say, for patients with GFR<25ml/min phosphaturia should be less than 800mg.
The combined analysis of these data suggests that for patients with GFR<25ml/min, and prescribing a diet of 0.8–0.9g/kg/day of protein, a reasonable target is a urinary P less than 800mg/day, and with GFR<15ml/min, urinary P excretion should be less than 600mg.
It is still pending to quantify the impact of inorganic P contributed by additives and soft drinks, being also the Pur and the urinary quotient P/UUN the parameter that could best identify those patients who abuse these products.
Fractional excretion and tubular reabsorption of phosphorusThe fractional excretion (FEP=Pur×Serum Cr/Serum P×Urine Cr) and its complementary parameter, the tubular reabsorption of P (TRP [%]=1−FEP) also provides relevant information. With the progressive loss of functioning renal mass the TRP is reduced and it increases the FEP, with an increase of FGF23 and PTH. In our population with CKD stage 4 and 5 we observe an average TRP of 59% (corresponding to FEP of 41%), which suggests P retention, despite documenting normal serum levels of P. In the study by González Parra et al.,69 performed in patients with CKD stage 3, it was observed a TRP of 66–74%. Thus TRP emerges as an valuable marker of abnormal mineral metabolism in CKD.67,70
CreatinineUrinary creatinine concentration is an essential parameter in renal patients. It is used to calculate creatinine clearance and the albumin/creatinine ratio in morning urine samples. In general, urinary excretion of creatinine in the normal adult is very variable ranging from 500 to 2000mg/day, depending largely on age and muscle mass. A practical way to express the normal range of urine creatinine excretion is as follows: male 14–26mg/kg/day, female 11–20mg/kg/day. Urinary creatinine values within this range suggests that 24h urine collection has been adequate.71
ConclusionAs a final reflection, urinary parameters provide reliable and useful information in routine clinical practice. They provide information on the patient's dietary habits and adherence to the treatment. Some recommendations are suggested in this review (Tables 1–4) that should be verified by other colleagues experience, and it can be a starting point for those nephrologists open to a more liberal dietary prescription on water, vegetables and fruits in patients with ACKD.
Key conceptsUrinary parameters provide reliable and useful knowledge for routine clinical practice; they provide information on the patient's dietary habits and adherence to our recommendations.
HydrationIn ACKD, the ability to generate a high volume of urine is preserved until more advanced stages of CKD.
The amount of liquid ingested may be a risk factor for CKD that is not established.
Achieving an adequate and individualized high urinary flow may help to preserve kidney function.
Controlling the diuresis of 24h is a necessary parameter to prevent the undesired effects of inadequate hydration (Table 1).
SaltThe restriction of Na is a primary recommendation for Blood Pressure control and prevention of hydrosaline retention.
The urinary Na is a useful and affordable determination to monitor saline intake (Table 2).
Urinary urea nitrogenProtein restriction is considered a first step to protect renal function in ACKD.
The determination of UUN has been verified as a useful parameter to estimate the protein intake (Table 3).
Acid charge and urinary potassiumThe reduction of the acid load can be a complementary nutritional strategy in the management of CKD patients.
The acid load can be estimated indirectly from the dietary survey or by measuring the urinary excretion of UUN and K.
A more liberal diet of vegetables and fruits (avoiding prolonged fasting), along with protein restriction, may be nutritionally healthy and renoprotective.
PhosphateThere is a close correlation between intake of protein and P.
The combined analysis of both may help to minimize the P load, without inadequately low protein restriction.
This correlation has been verified from the urinary parameters of UUN and P, which reflect the P absorption versus intake.
Monitoring phosphaturia and UUN may help to assess the intake of P in the form of additives and soft drinks.
Conflict of interestsThe authors have no conflicts of interest to declare.
Please cite this article as: Lorenzo Sellarés V. Utilidad de los parámetros urinarios en la enfermedad renal crónica avanzada. Nefrologia. 2019;39:124–132.