Antecedentes: La nefropatía inducida por contraste (NIC) constituye una causa importante del fracaso renal agudo en pacientes hospitalizados. Esta tiene lugar tras la administración de un medio de radiocontraste intravenoso. El estrés oxidativo se ha presentado como uno de los mecanismos más importantes de la patogénesis de la NIC. El objetivo de este estudio es determinar el efecto del alfa-tocoferol sobre la reducción del fracaso renal en un modelo de rata de NIC. Métodos: Las ratas Sprague Dawley machos, tratadas previamente con alfa-tocoferol (250 o 500 mg/kg/día) o con tween80 durante los 5 días anteriores a la inducción de CIN, fueron divididas en 6 grupos. Se determinaron los marcadores de la función renal y del estrés oxidativo, el nivel de malondialdehído (MDA), la capacidad antioxidante total (CAT) y la actividad de la superóxido dismutasa (SOD). Se seccionaron los tejidos de los riñones para el examen histopatológico. Resultados: En el grupo de medio de contraste (MC), se detectó un aumento de la urea sérica y de la creatinina. Asimismo, se demostró la presencia de necrosis tubular y de congestión capilar peritubular en este grupo. También se detectó un desequilibrio de los marcadores de estrés oxidativo, un aumento de MDA y una disminución de la actividad de la SOD en el riñón. Por el contrario, en el grupo de ratas con NIC inducida a las que se les administró alfa-tocoferol, se observó una reducción importante de la función renal y de la MDA renal, junto con un aumento significativo de la SOD renal. Curiosamente, en comparación con el grupo MC, en este grupo se demostró una reducción de MDA y un aumento de la CAT sérica, así como la prevención de la lesión tubular. Conclusión: Este estudio ha demostrado que el alfa-tocoferol posee un efecto protector del daño renal en ratas con NIC inducida. Por lo tanto, esta vitamina podría utilizarse como antioxidante para atenuar el daño oxidativo del radiocontraste.
Background: Contrast-induced nephropathy (CIN) is a prominent cause of in-hospital acute kidney injury occurring after the administration of intravenous radiocontrast medium. Oxidative stress has been proposed as one of the more important mechanisms in the pathogenesis of CIN. The aim of the present study has been to determine the effect of alpha tocopherol on the reduction of renal damage in a rat model of CIN. Methods: Male Sprague Dawley rats were subjected into six groups pretreated with alpha-tocopherol (250 or 500mg/kg/day) or the vehicle tweeen80 for 5 days before the induction of CIN. Renal function and oxidative stress markers; level of malondialdehyde (MDA), total antioxidant capacity (TAC), superoxide dismutase (SOD) activity were determined. Kidney tissues were sectioned for pathohistological examination. Results: In the contrast media (CM) group, an increase in serum urea and creatinine was found. Tubular necrosis and peritubular capillary congestion were demonstrated in this group. Also, an imbalance of oxidative stress markers; an increase in MDA and a decreased SOD activity in kidney were shown. On the contrary, in CIN-induced rats administrated with alpha-tocopherol group, a significant reduction of renal function and renal MDA, together with a significant increase of renal SOD, were observed. Interestingly, a reduction in MDA and an increase of TAC in serum, along with prevention of tubular injury, were demonstrated in this group, as compared to the CM group. Conclusion: This present study demonstrated that alpha tocopherol showed protective effect on the rat renal damage induced CIN. Therefore, this vitamin could be used as an antioxidant to attenuate the radiocontrast oxidative damage.
INTRODUCTION
Contrast-induced nephropathy (CIN) remains the third most common cause of hospital-acquired acute renal failure, following radiographic procedures. It has been reported that the prevalence of CIN may occur in 1 to 6% of hospitalized patients.1 It is especially the case that incidences of this condition may rise as high as 50% in patients at high-risk for such disorders as renal impairment and dehydration, diabetic nephropathy, volume depletion, hypercholesterolemia and congestive heart failure. This 50% incidence rate has also been seen in elderly patients, as well.2,3 Also, high-volume administration of contrast exposure, contrast osmolarity and the concomitant use of medication have been indicated as risk factors of CIN.4,5
The pathophysiological cellular mechanism of the development of CIN remains unclear. The hypothesis has been proposed that the radiocontrast agents can generate reactive oxygen species and cause a decreased renal blood flow and induced renal arterial vasoconstriction. Such disorders would lead to ischemic tubular injury and can be the cause of direct tubular toxicity.6 A number of animal-model investigations have suggested that oxidative tubular stress plays a substantial role, as supported by contrast agents-induced lipid peroxidation.7
In clinical examinations, many types of prophylaxis have been used in an attempt to prevent CIN. While hydration with isotonic saline before and after exposure to contrast agents is the standard practice, a substantial proportion of patients at risk continue nonetheless to be CIN.8,9 Other agents such as dopamine, captopril, theophylline, atrial natriuretic peptide and calcium channel blockers are also ineffective.10 An additional regimen such as the N-acetylcysteine (NAC) was used.11 Its properties are antioxidant and have the effect of a reactive oxygen radical scavenger; however, many clinical studies that assess the prevention of CIN have reported mixed results.
Vitamin E, particularly in the form of alpha tocopherol, has been proposed for the prevention or treatment of numerous health problems12 by reliance on the vitamin’s lipophilic antioxidant and anti-inflammatory properties13. In particular, it has been found to be the most efficient in scavenging peroxyl radicals for the protection of lipid peroxidation in cell membranes against oxidative damage. Vitamin E supplementation has been found to be protective against oxidative stress as, for example, in preventing cisplatin-induced acute renal failure14 and in protecting aging kidneys for ischemic ARF15. Therefore, alpha tocopherol may assist in the treatment of CIN by scavenging oxygen-free radicals. A recent report in the clinical pilot study by Tasanarong and colleagues16 has shown that the administration of vitamin E in CKD patients before coronary angiograph procedure provides a protective effect against CIN. In this clinical study, subjects in a prospective, randomized, double-blind placebo-controlled trial were randomized for supplementation with 525IU‘s of Vitamin E or placebo, at 48hrs, 24hrs and in the morning before the coronary procedures. It has been found that 23.08% of CKD patients were given the placebo, while, quite interestingly, only 5.88% of CKD patients supplemented with vitamin E had developed CIN, respectively. However, studies on the potential effects of vitamin E in CIN are not sufficient in either aspect of functional and histological changes in the kidneys.
The purpose of this study was to investigate the benefit of antioxidant vitamin E in its preventive effect on CIN in rat models and to examine the preventive effect of difference doses of vitamin E on CIN.
MATERIAL AND METHODS
Chemicals
Thiobarbituric acid (TBA), 2, 2'-azinobis (3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS), Trolox and all other chemical reagents were purchased from Sigma-Aldrich (Singapore). The low-osmolar, nonionic contrast.-media agent (Iopromide) was obtained from Schering AG (Germany). Vitamin E (alpha-tocopherol) was provided by Pharma Nord Aps (Vejle, Denmark)
Animals
Adult male Sprague Dawley rats obtained from the National Animal Center were used. All experiments were approved by the Animal Ethics Committee of the Faculty of Medicine, Thammasat University. All rats were housed in a temperature-controlled room (24oC±1oC) and were given standard food pellets and tap water until they weighed 250-300g, prior to the induction of CIN. The doses of vitamin E used in this study were determined on the basis of the several published studies that demonstrated these concentrations to be effective dose levels for the decrease in lipid peroxidation and oxidative renal injury in experimental animal models.17,18 Vitamin E was reconstituted in 1% Tween80 in PBS. Rats were divided into six groups of 6-8 animals to each group as follows: Group1, control rat administered 1 ml of 1% Tween80 in PBS (control); Group2, contrast media (CM), 250 or 500mg/kg/day of vitamin E given to rats in group3 (250mg Vit E) and in group4 (500mg Vit E), respectively. Rats administrated with 250 or 500mg/kg/day of vitamin E followed by CM injection were defined as group 5 (250mg Vit E+CM) and as group6 (500mg Vit E+CM), respectively. The animals were orally administered with antioxidant for 5 days, and then rats in Group2, Group5 and Group6 received contrast medium on the next day. At the end of the study (24h after CIN induction), all animal test subjects were euthanized by inhalation of ether anesthesia.
Induction of CIN in Rats
CIN-induced rats were subjected to CIN protocol as described by Agmon et al.19 Briefly, pentobarbital sodium (60mg/kg) anesthesia by intraperitoneal injection was followed by CIN induction, which was performed with drug administration into the tail vein. Drugs administered consisted of Indomethacin at a dose of 10 mg/kg, followed at 15min and at 30min later with Nw-nitro-L-Arginine methyl ester (L-NAME) at a dose of 10mg/kg and with the low-osmolar, nonionic contrast medium agent (Iopromide, Schering AG, Germany) at a dose of 1600mg I/kg. This quantity is the dose of contrast medium that is standard for clinical use and for other relevant experiments in rat models.20 As controls, rats were injected with PBS alone at each time.
Histopathologic examination of renal tissues
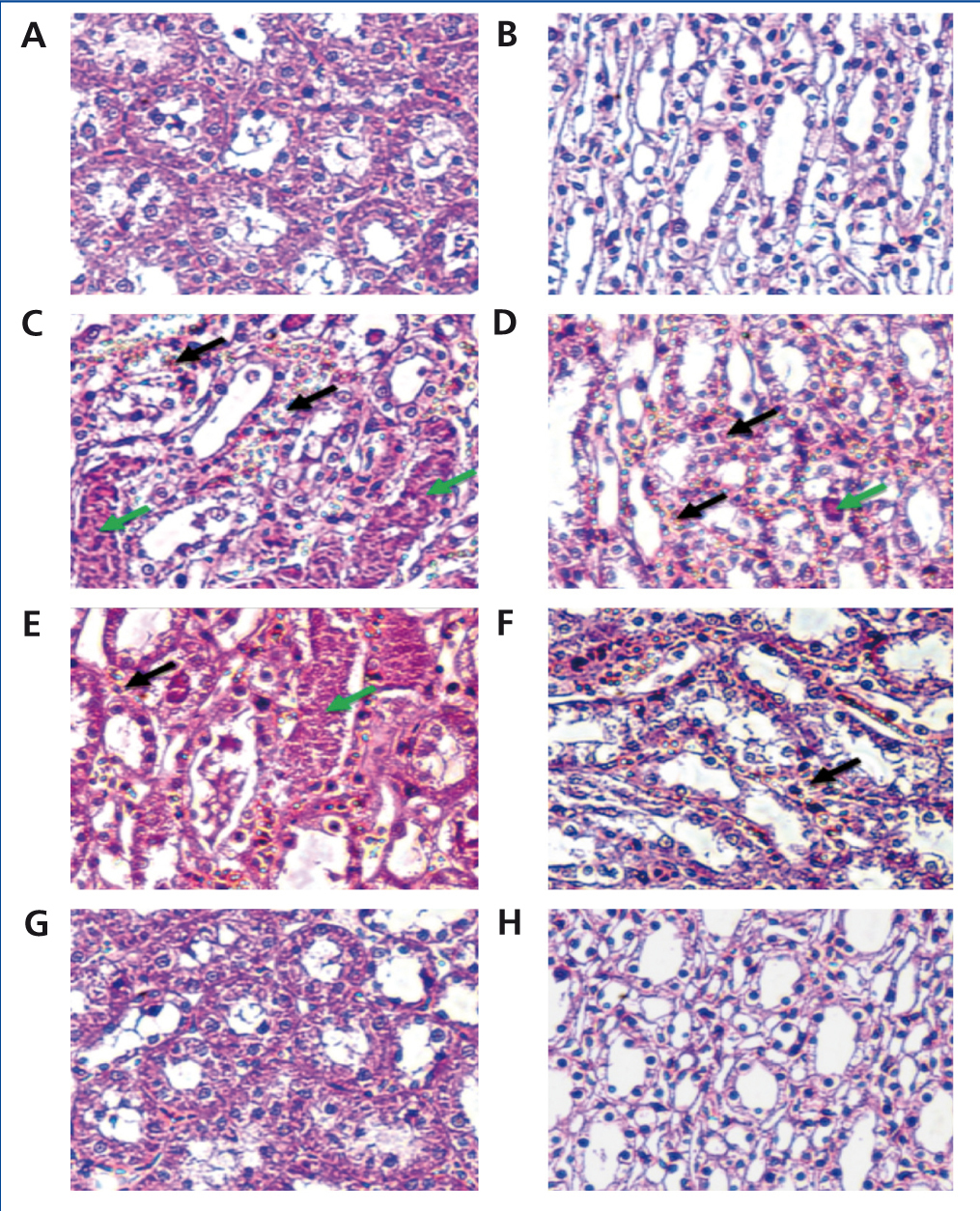
Both kidneys were excised immediately and cut into four equatorial sections; they were then washed twice with cold PBS. Two pieces of kidney were placed in 10% formaldehyde for histopathological examination. The other pieces were flash-frozen in liquid nitrogen, and stored at -70oC until used for subsequent tissue analysis. Histological slides of the formalin-maintained samples were prepared and then counterstained with hematoxylin, eosin (H&E) and periodic acid schiff staining (PAS) using standard procedure. These steps were then followed by semi-quantitative analysis of the kidney sections by a pathologist functioning in a blind manner. Tubular necrosis and proteinaceous casts were graded according to a previous methodology21 as follows: 0=no damage; 1=mild (unicellular, patchy isolated damage); 2=moderate (damage <25%); 3=severe (damage between 25 and 50%) and 4=very severe (>50% damage). The degree of medullary congestion was defined as: 0=no congestion; 1=mild (vascular congestion with identification of erythrocytes by x400 magnification); 2=moderate (vascular congestion with identification of erythrocytes by x200 magnification); 3=severe (vascular congestion with identification of erythrocytes by x100 magnification) and 4=very severe (vascular congestion with identification of erythrocytes by x40 magnification). Moreover, interstitial edema was graded as follows: 0=no damage; 1=mild (unicellular, patchy isolated damage); 2=moderate (damage <25%); 3=severe (damage between 25 and 50%) and 4=very severe (>50% damage).
Biochemical assay
In order to measure the oxidative stress markers including the oxidation of lipids, the antioxidant enzyme, total antioxidant capacity (TAC) and the renal function test, the serum was separated from the blood that had been obtained from the heart and was kept at -20oC until used. Renal function was investigated in serum blood urea nitrogen (BUN), and serum creatinine levels were determined using an automatic analyzer. Also, oxidative stress markers were measured in the kidneys. Renal tissue samples were prepared from 0.1g of frozen renal tissue which were homogenized in 1 ml of PBS (pH 7.2), and centrifuged at 10,000g for 10 min at 4oC; supernatant was then taken and kept at -80oC until used. Protein contents in tissue homogenates were estimated by the Bradford method using bovine serum albumin as a standard.
Lipid peroxidation, in term of malondialdehyde (MDA), was measured in the serum and in the renal tissue using thiobarbituric acid reactive substances (TBARS) assay, a method modified from that of Wong et al.22 Briefly, 100ml of serum or renal tissue samples were subjected to 0.75ml of phosphoric acid solution (0.44M) and incubated at room temperature for 10min; 0.5ml of TBA solution was then added. The mixtures were heated at 100oC for 30 min. The samples were centrifuged at 4000g for 10 min. The absorbance of the supernatant was read at 532nm. The concentration of MDA was calculated from the 0–20mM of standard MDA using 1,1,3,3 tetraethoxypropane. The results of the serum or renal MDA were expressed as mM or mM per mg of protein, respectively.
TAC in serum and in renal tissue samples was determined by modified ABTS decolorization method as described by Re et al.23 ABTS (7mM in final concentration), with potassium persulfate added (2.45mM final concentration), was kept overnight in the dark at room temperature. The initial absorbance of diluted ABTS radical cation solution was approximately 0.7±0.02 at 734 nm. After addition of 10 ml of sample or Trolox standard to 1ml of diluted ABTS, the absorbance was measured at 734nm at 1min after initial mixing. The TAC value was estimated by the reduction of the absorbance at 734nm and calculated against a standard curve of the Trolox (0–10mM). The values of TAC were expressed in an equivalence of mM Trolox or mM Trolox/mg of protein, when measured in the serum or renal tissue samples, respectively.
Renal SOD activity was also determined using the SOD kit (Sigma), following the manufacturing protocol. The SOD activity was measured and expressed as units per mg protein.
Statistical analysis
Results were presented as the means±SE. Student’s t-test (when two groups were considered) and one-way ANOVA were used to determine the significance of differences in group comparisons using SPSS software. A p value of less than 0.05 was considered to be as statistical significance.
RESULTS
Effect of vitamin E on the attenuation of renal dysfunction on CIN in rats
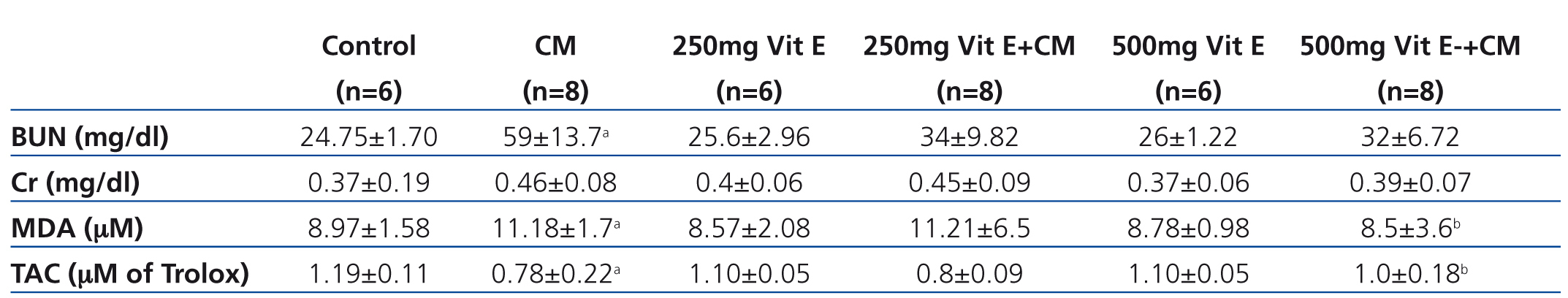
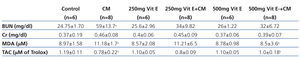
All rats in the CM group exhibited a significant increase in BUN concentrations when compared to controls, and also the increase of BUN in this group was higher than that in the other four groups, as shown in (Table 1). No significant change in the serum creatinine level was observed between groups. However, a 25% increase in serum creatinine levels from baseline in the CM group (0.46±0.08) was demonstrated, when compared to control group (0.37±0.19), and thus indicated that rats in CM group were characterized as CIN.24 In addition, a decrease in serum creatinine in 500mg Vit E+CM group (0.39±0.07) was shown.
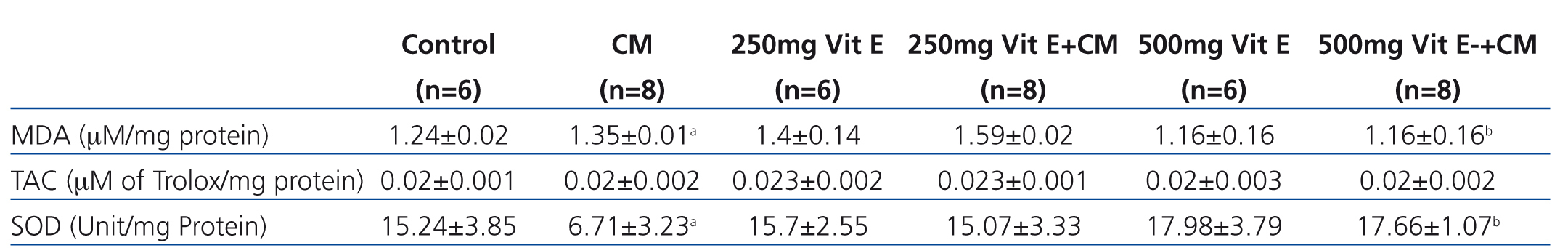
Effect of vitamin E on attenuation of oxidative stress on CIN in rats
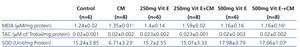
Serum MDA and renal MDA of all rats in the CM group were significantly increased when compared with those of the control group, whereas those MDA levels were significantly decreased in the 500mg Vit E+CM group when compared with CM group, (Table 1 and Table 2). The serum TAC levels in the CM group were significantly lower than those in control rats and all rats administered with vitamin E. The increase in serum TAC levels in the 500mg Vit E+CM group was observed and TAC levels were significantly higher than those in the CM group. However, there was no significant difference in renal TAC levels among groups, as shown in Table 2. The renal SOD levels were significantly decreased in the CM group when compared with the all other groups. Also the increase in renal SOD levels in a dose-dependent manner of vitamin E was presented, as shown in Table 2.
Effect of vitamin E on attenuation of tubular injury and medullary congestion on CIN in rats
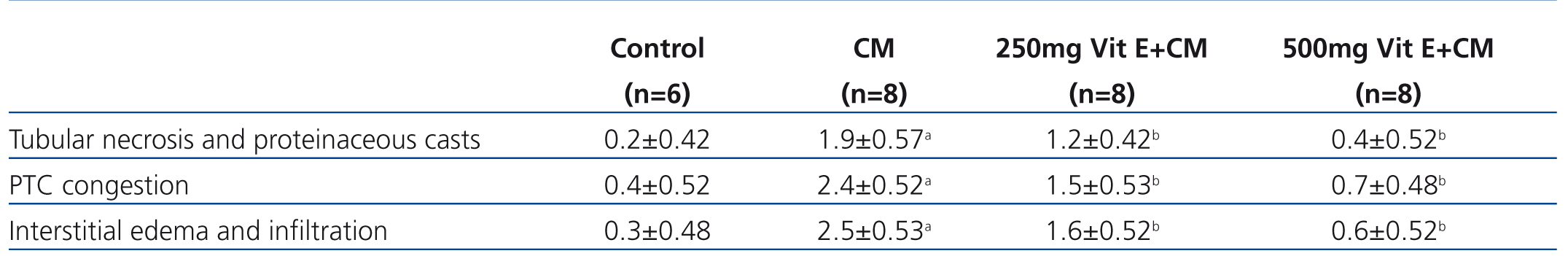
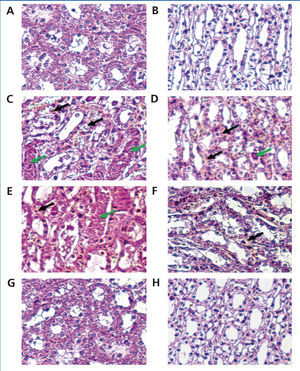
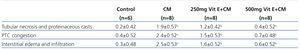
In the CM group, severe damage consisting of tubular necrosis and proteinaceous cast scored as high as 1.9±0.57. Damage was observed in the proximal tubular epithelial cells (TEC) in the outer stripe of the outer medulla (OSOM) and cortex (Table 3 and Figure 1). The marked structural damage, which included the widespread loss of brush border, a denudation of tubular cells, tubule dilatation and intratubular obstruction by granular casts, was presented. Moreover, the mean score of PTC congestion and interstitial edema in the OSOM and cortex were 2.4±0.52 and 2.5±0.53, respectively. These lesions were markedly different from those in the control kidney (p<0.05, Table 3). In contrast, the severity of TEC necrosis and proteinaceous cast, PTC congestion and interstitial edema were significantly reduced in the 250mg Vit E+CM group when compared with the CM group (p<0.05). Surprisingly, treatment with higher doses of vitamin E (500mg/kg/day) in CM induction reduced severity of kidney injury, in which histopathological changes in this group were demonstrated to be similar to those found in the control group.
DISCUSSION
To the best of our knowledge, the present study is the first report to investigate vitamin E effects in both renal function and structure on oxidative stress in radiocontrast-induced nephrotoxicity in rats. We found that vitamin E has a protective effect in preventing the development of renal dysfunction and improving oxidative stress injury in CIN-induced rats. These findings were demonstrated by biochemical markers. Also vitamin E has markedly alleviated incidences of renal tubular injuries, as shown by histological analysis.
Increased oxidative stress is one of the key underlying mechanisms in CIN pathogenesis.4 Impairment in the antioxidant defense mechanism also has been reported, including increased lipid peroxidation and inactivated antioxidant enzyme.11,25 The cellular antioxidant defense mechanisms consist of enzymatic antioxidant components such as catalase, glutathione peroxidase and superoxide dismutase, and of non-enzymatic antioxidant components such as vitamin E, vitamin C and glutathione. Many studies have been reported that supplements containing antioxidant vitamins reduce free radical production, trap free radicals or interrupt their peroxidation. Ascorbic acid (vitamin C), a water-soluble antioxidant vitamin, reacts directly with hydroxyl radicals and hydrogen peroxide to reduce lipid peroxidation.26 Its role has been investigated and it was found that this supplementation offers partial protection against contrast-induced renal failure.27 In clinical investigation, N-acetylcysteine or NAC, a glutathione precursor, has been used to prevent the CIN through its antioxidant property. However, conflicting results have been found in several studies.5-7 In a recent report, erdostein, a mucolytic drug that acts as an antioxidant agent,28 has been assessed for its preventive effects on CIN-induced rats, although the molecular mechanisms that provide the protective effects need to undergo further investigation. Until now, no medical agents seem to be very promising for practical regimens.
Vitamin E, an example of a non-enzymatic antioxidant, is the main lipophilic antioxidant found in the blood and organs, and is carried in lipoprotein and cell membranes, respectively.29 Importantly, its property acts to reduce free-radical and lipid peroxidation formation on cell membranes, consequences initiated by oxidant-antioxidant imbalance, and to decrease cell damage.30,31 Vitamin E has been addressed to be useful for treatment in many nephropathy, such as cisplatin-induced acute renal failure32 or cadmium.33 These components consist of renal toxicity with their pathogenic effects on oxidative stress. In the present study, the increased lipid peroxidation caused by the increased oxidative stress in the kidneys induced by radio-contrast media was assessed and investigated. It was found that the lipid peroxidation products, in terms of MDA, increased in both renal tissue and serum. A consistency in the decrease of serum TAC and renal SOD activity was observed in CIN-induced rats. These results are in agreement with the observation of many studies.26 SOD, an enzymatic antioxidant, can act to scavenge superoxide ions. This present study shows the significantly lower levels of SOD activity in CIN-induced rats, as compared with those of the control group. The decreased activity of SOD in renal tissue of CIN-induced rats may be the result of oxidative stress induced by inactivation and/or excessive antioxidant consumption,34 and is a causative factor in oxidative damage. In the current study, vitamin E supplements, particularly in high doses, significantly attenuated the increase of MDA concentration in renal tissue and serum and significantly increased renal SOD activity. Nevertheless, the unaltered TAC level in renal tissues exposed to contrast media was observed, whereas the TAC level in the plasma of this group significantly decreased, when compared with the control group. From these findings, it was hypothesized that the compensatory mechanism of a tissue antioxidant state that develops in response to local oxidative stress by recycling of endogenous non-enzymatic antioxidant vitamins may have involved and/or associated a lesser sensitivity in the estimation of total antioxidant capacity value in tissue samples.35,36 However, the antioxidant property of vitamin E is not only to scavenge ROS, but also to up-regulate antioxidant enzymes through regulation of the gene expression or activity of antioxidant enzymes.37-39 In this study, the supplements of vitamin E exhibited the elevated renal SOD activity which indicates an elevated antioxidant status. These data were similar to a number of reports which have shown that the increased activity of intracellular antioxidant enzymes can also be correlated with a decrease in lipid peroxidation increases.40,41
Taken together, the evidence presented in this study has shown that imbalance in antioxidative scavenger-promoted generation of MDA, resulting in appearance of renal tubular injury, is consistent with histological assessment. This study showed that outer medullary congestion of renal cells, one of the vascular hallmarks of CIN,42 was significantly higher in the CIN-induced rats. After vitamin E supplementation, we found that medullary congestion significantly decreased, in comparison with the CM group. Furthermore, high incidence of tubular necrosis in the CM group was presented, whereas a low score of tubular necrosis in both of the CM groups administrated with vitamin E was found. These results support the fact that vitamin E treatment has a preventive effect in the improvement of histopathological changes, and that these observations have paralleled the decrease in MDA levels and increase in SOD activity. Moreover, all of the oxidative stress marker concentrations found in this experiment are likewise related to the pathogenesis of CIN and its pathologic markers, including BUN and serum creatinine. The increase in BUN and serum creatinine have been found in CIN-induced rats, but not in CIN-induced rats supplemented with vitamin E. It implied that vitamin E treatment can reduce the renal dysfunction caused by radio-contrast media.
In this study, an effective dose of vitamin E also has been investigated at dosages of 250 and 500mg/kg/day. It was found that a low dose cannot reduce the MDA level, but a higher dose can reduce, in particular, the serum MDA and the renal MDA; increase the renal SOD activity; and attenuate renal dysfunction. In present results, supplementation with vitamin E 250mg/kg/day exhibited the increasing MDA and SOD activity in renal tissue were demonstrated when compared to control group. In our opinions, it is possible to assume that low dose of vitamin E can have pro-oxidant effects on renal tissue and accompany an inappropriate dose to generate antioxidant effect. It has been reported that vitamin E acts as a pro-oxidant by activation of enzymatic antioxidant (SOD and GPx), leading to increase lipid peroxidation43 and also its capacity of free radical scavenger is a dose dependent manner.39 The high dosages of vitamin E found to be effectively protective of the renal function from oxidative damage in rat models have been demonstrated in several studies.44 Results of the present study imply that high doses of vitamin E provide a beneficial effect against CIN. However, the high dosage used in this study (approximately equivalent to human doses at 2500mg in an adult of 60kg body weight) was found to be higher than that of a tolerable upper intake level (UL) of vitamin E (1000mg/day), as recommended by the food and Nutrition Board of the U.S. Institute of Medicine. Non-toxic intake of vitamin E in the highest dosage of 3200mg/day also has been shown.45 Therefore, in further study, the short-term administration of an adjusted dosage of vitamin E (1000mg/d) prior to radio-contrast procedures in high-risk patients should be considered. Indeed, in CKD patients, several clinical studies have reported that the imbalance of oxidative stress and anti-oxidant levels are a common feature of CKD. Factors contributing to increased oxidative stress in CKD patients, such as diabetes, chronic inflammation and uremia, have also been reported.46,47 For example, the uremic patients had higher levels of plasma oxidative markers and decreased antioxidant activities, as correlated with their levels of renal dysfunction.48,49 Thus, as a means of preventing CIN, these patient groups should have the benefit of pre-treatment with antioxidant vitamin E in cases of radio-contrast administration.
In conclusion, the present study has emphasized the protective role of vitamin-E supplementation, which improves the renal dysfunction and morphological tubular injury in rats caused by CIN induction through its antioxidant activity. These results were associated with a significant decrease in lipid peroxidation and a significant increase in the antioxidant enzyme, particularly SOD activity in renal tissue. Therefore, based on our results, we could suggest that vitamin-E administration prior to radio-cardiography may have the potential of protecting CIN in patients. However, further clinical trials need to be optimized in order to produce a better protection.
Acknowledgement
This work was supported by a research grant by the Faculty of Medicine, Thammasat University, Thailand.
Conflict of interest
The authors declare that there is no conflict of interest associated with this manuscript.
Figure 1. Histological with H& E staining of contrast-induced nephropathy in rats
Table 1. Biochemical data of the serum in the six experimental groups
Table 2. Biochemical data of the renal in the six experimental groups
Table 3. Histological analysis of tubular necrosis, PTC congestion and interstitial edema in the four experimental groups