SARS CoV2 infection has had a major impact on renal transplant patients with a high mortality in the first months of the pandemic. Intentional reduction of immunosuppressive therapy has been postulated as one of the cornerstone in the management of the infection in the absence of targeted antiviral treatment. This has been modified according to the patient`s clinical situation and its effect on renal function or anti-HLA antibodies in the medium term has not been evaluated.
ObjectivesEvaluate the management of immunosuppressive therapy made during SARS-CoV2 infection, as well as renal function and anti-HLA antibodies in kidney transplant patients 6 months after COVID19 diagnosis.
Material and methodsRetrospective, national multicentre, retrospective study (30 centres) of kidney transplant recipients with COVID19 from 01/02/20 to 31/12/20. Clinical variables were collected from medical records and included in an anonymised database. SPSS statistical software was used for data analysis.
Resultsrenal transplant recipients with COVID19 were included (62.6% male), with a mean age of 57.5 years.
The predominant immunosuppressive treatment prior to COVID19 was triple therapy with prednisone, tacrolimus and mycophenolic acid (54.6%) followed by m-TOR inhibitor regimens (18.6%). After diagnosis of infection, mycophenolic acid was discontinued in 73.8% of patients, m-TOR inhibitor in 41.4%, tacrolimus in 10.5% and cyclosporin A in 10%. In turn, 26.9% received dexamethasone and 50.9% were started on or had their baseline prednisone dose increased.
Mean creatinine before diagnosis of COVID19, at diagnosis and at 6 months was: 1.7 ± 0.8, 2.1 ± 1.2 and 1.8 ± 1 mg/dl respectively (p < 0.001).
56.9% of the patients (N = 350) were monitored for anti-HLA antibodies. 94% (N = 329) had no anti-HLA changes, while 6% (N = 21) had positive anti-HLA antibodies. Among the patients with donor-specific antibodies post-COVID19 (N = 9), 7 patients (3.1%) had one immunosuppressant discontinued (5 patients had mycophenolic acid and 2 had tacrolimus), 1 patient had both immunosuppressants discontinued (3.4%) and 1 patient had no change in immunosuppression (1.1%), these differences were not significant.
ConclusionsThe management of immunosuppressive therapy after diagnosis of COVID19 was primarily based on discontinuation of mycophenolic acid with very discrete reductions or discontinuations of calcineurin inhibitors. This immunosuppression management did not influence renal function or changes in anti-HLA antibodies 6 months after diagnosis.
La infección por SARS CoV2 ha impactado de forma importante en los pacientes con trasplante renal causando una elevada mortalidad en los primeros meses de la pandemia. La reducción intencionada de la inmunosupresión se ha postulado como uno de los pilares en el manejo de la infección ante la falta de un tratamiento antiviral dirigido. Ésta se ha modificado de acuerdo con la situación clínica de los pacientes y su efecto sobre la función renal o los anticuerpos anti-HLA a medio plazo no ha sido evaluado.
ObjetivosEvaluar los cambios de inmunosupresión realizados durante la infección por SARS-CoV2, así como la función renal y los anticuerpos anti-HLA de los pacientes trasplantados de riñón a los 6 meses del diagnóstico de COVID19.
Material y métodosEstudio retrospectivo, multicéntrico nacional (30 centros) de pacientes trasplantados de riñón con COVID19 desde el 01/02/20 al 31/12/20. Se recogieron las variables de la historia clínica y se incluyeron en una base de datos anonimizada. Se utilizó el programa estadístico SPSS para el análisis de resultados.
ResultadosSe incluyeron 615 trasplantados renales con COVID19 (62.6% varones), con una edad media de 57.5 años.
El tratamiento inmunosupresor predominante antes del COVID19 era la triple terapia con prednisona, tacrolimus y ácido micofenólico (54.6%) seguido de los regímenes con inbidores m-TOR (18.6%). Tras el diagnóstico de la infección se suspendió el ácido micofenólico en el 73.8% de los pacientes, el inhibidor m-TOR en el 41.4%, tacrolimus en el 10.5% y ciclosporina A en el 10%. A su vez, el 26.9% recibieron dexametasona y al 50.9% se les inició o aumentó la dosis de prednisona basal.
La creatinina media antes del diagnóstico de COVID19, en el momento del diagnóstico y a los 6 meses fue de: 1,7 ± 0,8; 2.1 ± 1.2 y 1,8 ± 1 mg/dl respectivamente (p < 0,001).
Al 56.9% de los pacientes (N = 350) se les monitorizó los anticuerpos anti-HLA. El 94% (N = 329) no presentaron cambios en los anti-HLA, mientras que el 6% (N = 21) los positivizaron. De entre los pacientes con anticuerpos donante-específicos post-COVID19 (N = 9), a 7 pacientes (3,1%) se les había suspendido un inmunosupresor (en cinco de ellos se suspendió ácido micofenólico y en 2 tacrolimus), a 1 paciente los 2 inmunosupresores (3,4%) y al otro paciente no se le había modificado la inmunosupresión (1,1%), siendo estas diferencias no significativas.
ConclusionesEl manejo de la inmunosupresión tras el diagnóstico de COVID19 se basó fundamentalmente en la suspensión de ácido micofenólico con reducciones o suspensiones muy discretas de inhibidores de calcineurina. Este manejo de la inmunosupresión no influyó en la función renal ni en cambios de los anticuerpos anti-HLA a los 6 meses del diagnóstico.
On March 11, 2020, the World Health Organization (WHO) declared infection with the novel coronavirus SARS-CoV2 as a global pandemic. This virus, whose origin had occurred a few months ago in the Chinese city of Wuhan, is characterized by respiratory infection and systemic involvement and has been called coronavirus infectious disease or COVID19 for its acronym in English.1
On March 2, 2020, a hospital in Madrid diagnosed the first renal transplant patient with SARS-CoV2 infection in Spain and on March 18, 2020, the Spanish Society of Nephrology created the SEN COVID registry2 of patients with renal replacement therapy who were infected by this virus.
Patients on chronic immunosuppressive therapy are at increased risk of infections and in the case of renal transplant patients it is one of the main complications after performing the implant.3 This fact has caused the pandemic to have a significant impact on this group of patients, causing high mortality in the first months of the pandemic2,4 and various studies have confirmed the worst prognosis of the disease in this population.
The management of viral infections in the transplanted population is fundamentally based on prevention, through vaccination, antiviral prophylaxis strategies and general infection control measures. This is the first step of treatment and is of the utmost importance; however, in infectious diseases that appear de novo, such as the current COVID19 pandemic, management includes targeted antiviral treatment, in situations where it is available, and the reduction of immunosuppression to favor the development of specific immunity.5
Since the beginning of the COVID19 pandemic, several cases have been published in kidney transplant patients in which the management of viral infection included the reduction of immunosuppression.6–11 This does not appear to influence short-term patient survival or kidney function or the development of antibodies against donor-specific human leukocyte antigens (HLAs).6–11
Taking advantage of the creation of the SEN COVID registry and that during the SARS-CoV2 pandemic immunosuppression has been modified in these patients according to the clinical situation and at the discretion of each center is an opportunity to learn how immunosuppression has been managed in the different participating centers, as well as the medium-term consequences if any.
We present this study, whose main objective was to analyze the changes in renal function and anti-HLA antibodies in the medium term (6 months) of kidney transplant patients in Spain diagnosed with COVID19, whose immunosuppression was intentionally decreased, who were included in the COVID registry of the SEN, and who agreed to participate. The change in immunosuppression performed and the time that immunosuppression was kept reduced were evaluated as secondary objectives.
Patients and methodsStudy designWe conducted a national observational, retrospective, and multicenter study in which 30 Spanish hospitals participated with follow-up of patients with renal transplantation and who had included patients in the COVID registry of the SEN.
Study populationThe study population was kidney transplant patients diagnosed with SARS-CoV2 infection from March 1, 2020, to December 31, 2020. We excluded patients diagnosed with coronavirus infection by serological methods, those who died during the study period, and patients with incomplete data in relation to the main objective of the study. Patients were followed for a period of 6 months.
Diagnosis of SARS-CoV2 infection was via polymerase chain reaction (PCR) technique of nasal or nasopharyngeal samples.
Patient inclusion covered two periods or waves of the pandemic in Spain; the first from February to July 2020 (N = 274, 44%) and the second from August to December 2020 (N = 341, 55.4%).
Demographic variables were collected (age, sex and race), clinical variables related to chronic kidney disease (CKD) and renal transplantation (etiology of CKD, number of renal transplantations, immunological risk, date of transplantation) as well as maintenance of immunosuppressive treatment, renal function (creatinine, glomerular filtration using the CKD-EPI equation in ml/min/1.73 m2, protein/creatinine index in urine of isolated sample in mg/g Cr) and anti-HLA antibodies prior to diagnosis of SARS-CoV2 infection.
Variables related to the management of immunosuppression after coronavirus infection, renal function (creatinine, glomerular filtration using the CKD-EPI equation in ml/min/1.73 m2 and protein/creatinine index in urine of isolated sample in mg/g Cr) at the diagnosis of the infection, at 7 and 15 days and at 2 and 6 months after the diagnosis of the disease, anti-HLA antibodies at 2 and 6 months after the diagnosis, as well as clinical variables of disease evolution (period of the pandemic, hospital or intensive care unit admission, days of admission, clinical manifestations, and antiviral treatment) were also collected.
Acute renal failure (ARF) was defined and classified according to the KDIGO clinical practice guideline of acute renal damage.12
The clinical and demographic data were obtained from the clinical charts of the patients and collected in the COVID registry of the S.E.N. Subsequently, they were included in an anonymized database on which the analysis was carried out.
The clinical research ethics committee of the Hospital Universitario La Paz (code PI-4332) approved this research as did the committees of each participating center.
ImmunosuppressionThe immunosuppression of patients was modified according to the clinical situation and the criteria of each center, there was no standard management protocol.
In general, immunosuppression was progressively reduced as the patient's clinical situation worsened and consisted, basically, of the reduction, suspension, or non-adjustment of immunosuppression:
- -
In cases of SARS-CoV2 infection with mild or asymptomatic symptoms, immunosuppression was not modified, except in 4 centers where the adjuvant immunosuppressant (mycophenolic acid or m-TOR inhibitors) was reduced or discontinued.
- -
-In cases of moderate infection with pneumonia and hypoxemia requiring oxygen therapy through nasal cannulas, the adjuvant immunosuppressant (mycophenolic acid or m-TOR inhibitors) was discontinued, the dose of calcineurin inhibitor was reduced, except in 3 centers in which both the adjuvant (mycophenolic acid or m-TOR inhibitors) and the main immunosuppressant were discontinued, and the dose of steroids was increased.
In cases of severe infection with pneumonia requiring mechanical ventilation, both the adjuvant immunosuppressant (mycophenolic acid or m-TOR inhibitors) and the main one were discontinued, and the dose of steroids was increased.
There was a percentage of patients in whom the initial suspension of the immunosuppressant did not materialize due to interactions of immunosuppressants with other drugs and who maintained their immunosuppressant levels in the blood during admission. This group of patients were considered, for the analysis of the management of immunosuppression, as patients in whom the medication was not adjusted or failed suspension.
Antiviral and anti-inflammatory treatment against SARS-CoV2Each center carried out treatment aimed at controlling the infectious disease according to its own protocols, which varied according to the scientific evidence available in each period of the pandemic.
In relation to antiviral treatment:
82.5% of patients (N = 226) received hydroxychloroquine during the first period of the pandemic while only one patient received it in the second period.
19.3% (N = 53) of patients received lopinavir/ritonavir in the first period and only 3 patients in the second period.
1.1% (N = 3) received remdesivir during the first period and increased to 5.9% (N = 20) during the second period.
In relation to anti-inflammatory treatment:
A 40.9% of patients (N = 112) received dexamethasone or methylprednisolone during the first period and 51.5% (N = 173) received it during the second period.
Interferon was used in 4% of patients (N = 11) in the first period and in a smaller proportion of patients in the second (N = 5, 1.5%).
Tocilizumab was used in 11% of patients (N = 30) in the first period and in 5.9% (N = 20) in the second period.
Anakinra was used in 11 patients, 8 during the first period and 3 patients in the second period.
Plasma from convalescents was used only in the second period of the pandemic in 7 patients (2.1%).
Statistical analysisThe results of the quantitative variables are expressed as mean ± standard deviation or as median. Comparison of means of continuous quantitative variables was performed using Student's t-test for paired and unpaired samples, as appropriate. If variables were not normally distributed, we used non-parametric tests: the Mann–Whitney U test to compare independent samples and in the case of paired samples, the Wilcoxon test was used to compare sets of 2 samples and the Friedman test when comparing more than 2 paired samples. The results of categorical variables are expressed with absolute and relative frequency. The statistical chi-square test or Fisher's exact test was used, as necessary, for the comparison of categorical variables. We treated p-value <0.01 as a statistically significant result. The statistical program SPSS version 20.0 was used for the analysis.
Results615 kidney transplant patients diagnosed with SARS-CoV2 infection were included. 75.9% (N = 467) were admitted to the hospital and 24.1% were followed on an outpatient basis (N = 148).
Table 1 shows the demographic characteristics of the study population, as well as the clinical variables related to renal transplantation and those related to the evolution of infectious disease.
Demographic characteristics and clinical variables related to renal transplantation and the evolution of SARS-CoV2 infection in the study population.
| Age (x ± sd) – years (min–max) | 57.5 ± 12.7 (14−90) |
| Sex, N (%) – male/female | 385 (62.6)/230 (37.4) |
| Ethnic group N (%) | |
| White | 449 (73) |
| Latin | 77 (12.5) |
| Asian | 6 (1) |
| Other | 38 (6.2) |
| Unknown | 45 (7.3) |
| Etiology CKD, N (%) | |
| Diabetic nephropathy | 72 (11.7) |
| Nephroangiosclerosis | 52 (8.5) |
| Glomerular | 161 (26.2) |
| Chronic TINI | 51 (8.3) |
| HR polycystic disease | 103 (16.7) |
| Systemic disease | 21 (3.4) |
| Other | 143 (23.3) |
| Unknown | 12 (2) |
| No. kidney transplant, N (%) | |
| First | 523 (85) |
| Second | 61 (9.9) |
| Third | 4 (0.7) |
| Four | 3 (0.5) |
| Not specified | 24 (3.9) |
| Immunological risk, N (%) | |
| Not sensitized (PRA 0%) | 453 (73.7) |
| Sensitized (PRA 0−50%) | 72 (11.7) |
| Highly sensitized (PRA > 50%) | 44 (7.2) |
| Not specified | 46 (7.5) |
| Months since kidney transplant (x ± sd) | 100 ± 85.4 |
| <2 months, N (%) | 25 (4.1) |
| 2−12 months, N (%) | 42 (6.8) |
| >12 months, N (%) | 548 (89.1) |
| Pandemic periods, N (%) | |
| First period (from March to July 2020) | 274 (44.6) |
| Second period (from August to December 2020) | 341 (55.4) |
| Hospital admissions, N (%) | 467 (75.9) |
| Admission days (x ± ds) | 18 ± 18.2 |
| Mean | 13 |
| Minimum–maximum | 1−190 |
| Respiratory clinic (cough, expectoration, rhinorrhea), N (%) | 384 (62.4) |
| Fever, N (%) | 430 (70) |
| Dyspnea, N (%) | 226 (36.7) |
| G.I symptoms (nausea, vomiting, diarrhea), N (%) | 228 (37.1) |
| Asymptomatic, N (%) | 59 (9.6) |
| Pneumonia, N (%) | 388 (63.1) |
| Without oxygen, N (%) | 113 (29.1) |
| Nasal oxygen, N (%) | 157 (40.5) |
| VMK, N (%) | 61 (15.7) |
| VM (invasive or non-invasive), N (%) | 49 (12.6) |
| Not specified, N (%) | 8 (2.1%) |
| Admission toICUN (%) | 44 (7.2) |
N = 615.
x: mean; sd: standard deviation; CKD: chronic kidney disease; M: male; HR: hepatorenal; F: female; min: minimum; max: maximum; TIN: tubulointerstitial nephritis; PRA: reactive antibody panel; VM: mechanical ventilation; VMK: ventimask; ICU: intensive care unit.
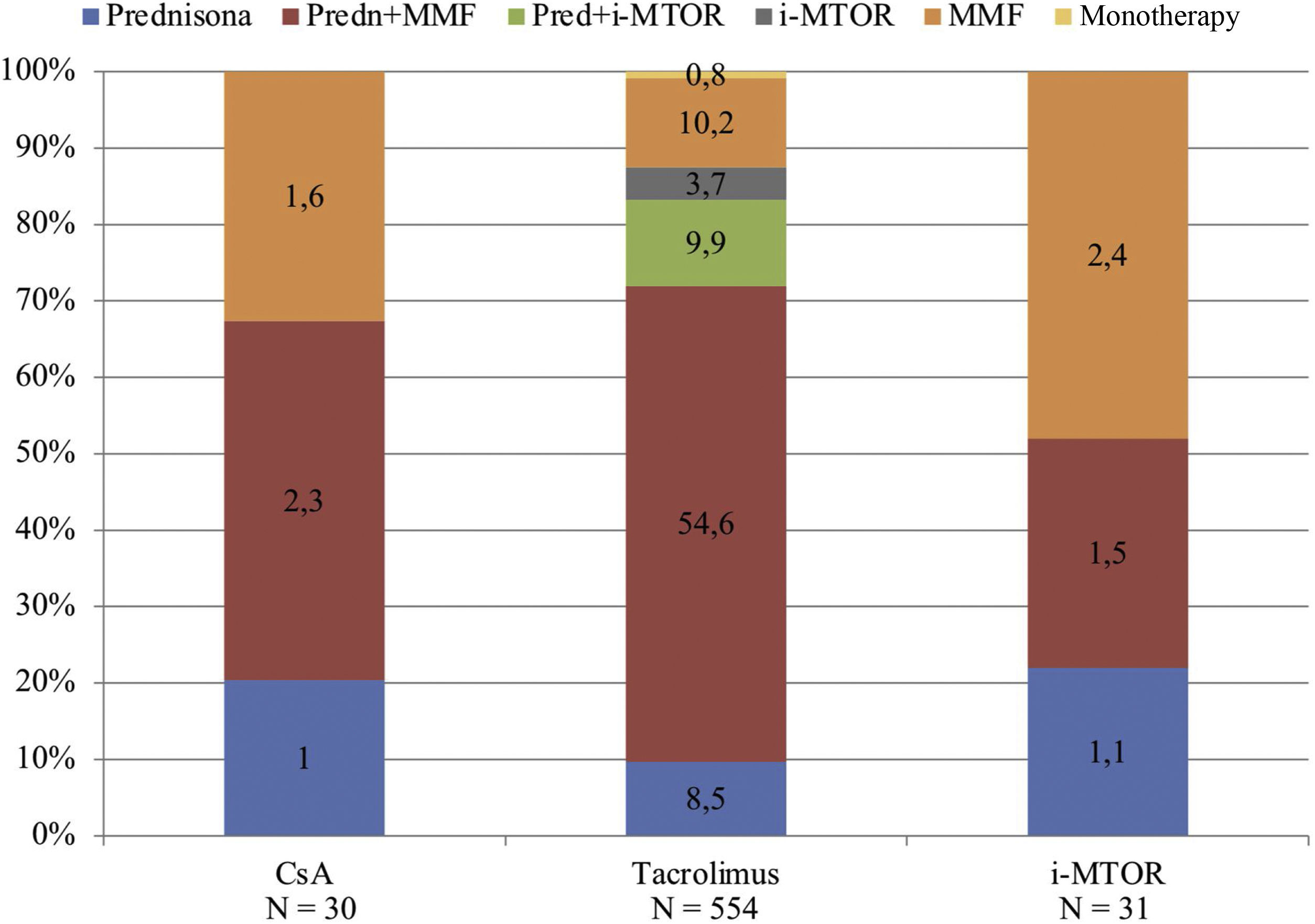
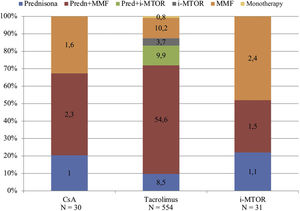
The predominant immunosuppressive (IS) treatment prior to COVID19 was triple therapy, being present in 68.3% (N = 420) of patients, followed by dual therapy (28.5%, N = 176) and tacrolimus monotherapy regimens in a very small proportion of patients (0.8%, N = 5).
The prednisone, tacrolimus and mycophenolic acid regimen was the majority (54.6%, N = 336), followed by the dual therapy regimen with tacrolimus and mycophenolic acid (10.2%, N = 63) and triple therapy with prednisone, tacrolimus, and m-TOR inhibitors (9.9%, N = 61). The rest of the guidelines were represented by a low proportion of patients, as shown in Fig. 1.
After the diagnosis of SARS-CoV2 infection, treatment management basically consisted of the reduction, suspension, or non-adjustment of immunosuppression.
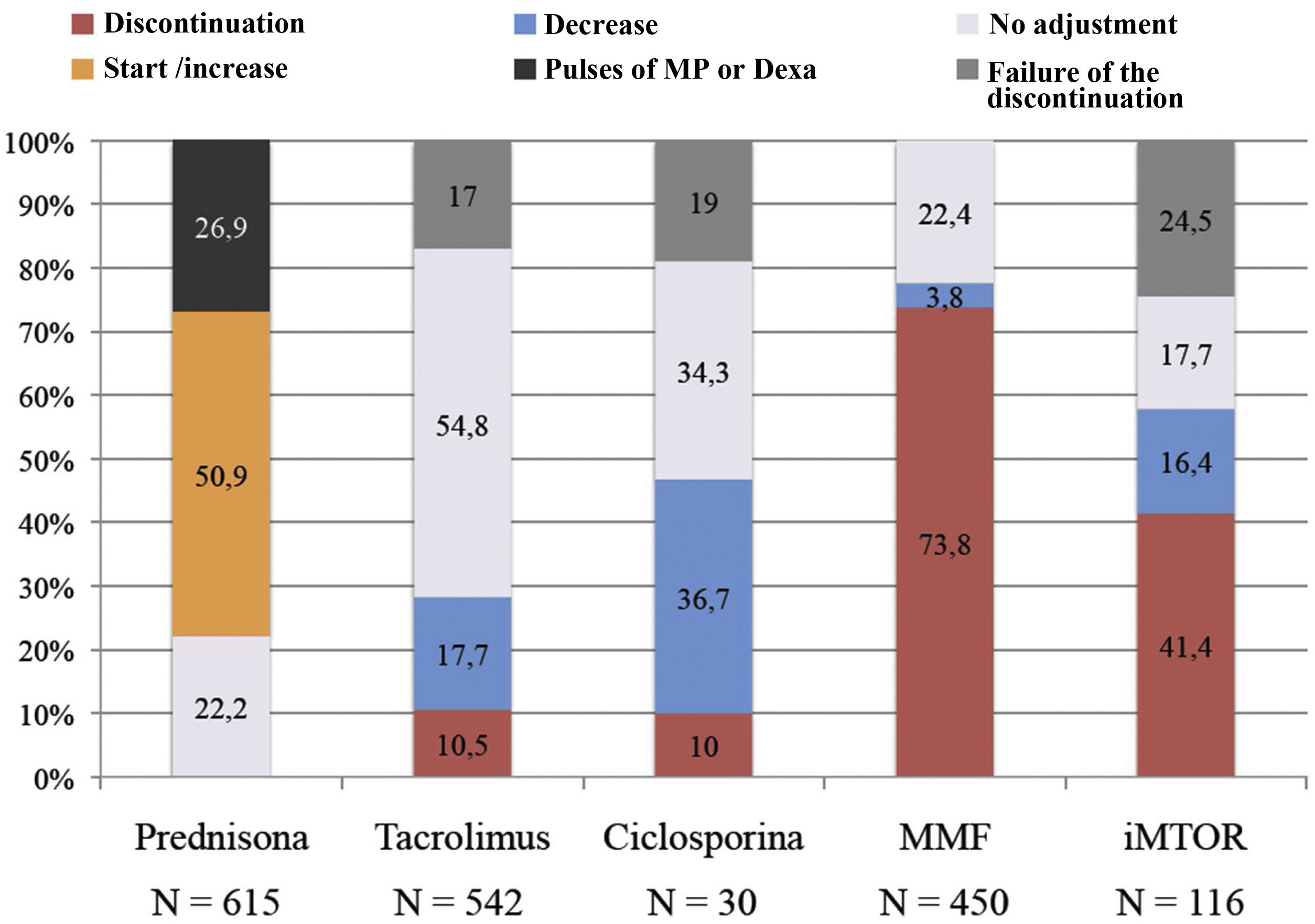
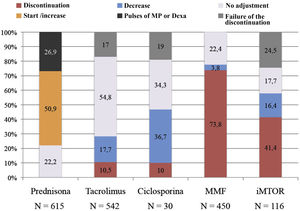
Fig. 2 shows the final management of each immunosuppressive drug, including in the graph the group of patients in whom the suspension of immunosuppression did not occur during admission as a result of the interactions of immunosuppressants with other drugs used (failed suspension) and the group of patients who received dexamethasone or pulses of methylprednisolone as part of the anti-inflammatory treatment of the disease.
Mycophenolic acid treatment was mostly discontinued: 73.8% (N = 332) of patients previously taking it (N = 450) discontinued treatment. Cyclosporine A was proportionally the drug that was most reduced, doing so in 36.7% (N = 11) of patients that were taking it before infection (N = 30). Tacrolimus was the least adjusted drug during infection remaining in 71.8% of patients (in 323 patients [54.8%] it was unchanged and in 66 patients [17%] the planned discontinuation failed). And m-TOR inhibitors were discontinued or not adjusted in the same proportion of patients (41.4% vs. 42.2% respectively, although in the group of patients without adjustment of the m-TOR inhibitor there was a high percentage of patients with failed suspension [24.5%]).
Baseline prednisone dose was increased in 50.9% of patients (N = 313) and of these a 66.5% also received dexamethasone or methylprednisolone pulses. In patients in whom the prednisone dose was not adjusted (N = 301, 49.1%), 26.8% (N = 81) received dexamethasone.
The levels of the immunosuppressants were monitored during admission in each patient and were lower, as expected, in patients in whom the dose was intentionally reduced compared to those who maintained their usual dose. The group of patients in whom the planned discontinuation of the immunosuppressive drug was unsuccessful had mean blood drug levels higher than those of patients in whom the medication was not adjusted (Table 2).
Mean immunosuppressant levels during SARS-CoV2 infection admission.
| Reduction tacrolimus (N = 63 of 96) | 3.91 ± 1.2 (1.35−7.4) | |
| Non-adjustment of tacrolimus (N = 239 of 297) | 11.5 ± 7.5 (2−50) | 0.08 |
| Failed suspension of tacrolimus (N = 66 of 92) | 15.4 ± 9.8 (6−41) | |
| Cyclosporine A reduction (N = 11) | 63.5 ± 23.7 (27.8−90) | |
| Non-adjustment of cyclosporine A (N = 6 de 13) | 155 ± 137.4 (55−485) | NS |
| Failed cyclosporin A suspension (N = 3) | 157 ± 76.9 (77−252) | |
| Reduction of I-mTOR (N = 19) | 3.1 ± 1.1 (0.7−4.8) | |
| No adjustment of i-mTOR (N = 15 de 49) | 9.4 ± 3.8 (0.9−20) | NS |
| Failed suspension of I-mTOR (N = 12) | 11.4 ± 3.2 (6−20) |
The mean levels of the “did not adjust” patient group are compared to the “failed suspension” patient group. Student’s t-test or Mann–Whitney U test was used for independent samples as needed.
i-MTOR: m-TOR inhibitors.
Immunosuppression was progressively introduced when the clinical situation of the patients allowed it, so that treatment with mycophenolic acid, tacrolimus, cyclosporin A or m-TOR inhibitors was restarted at 33, 17, 9 and 12 days on average, respectively. The usual prednisone dose was recovered at 36 days median (50.2 ± 48.8 days).
Six-point two percent of patients (N = 38) who, having discontinued some immunosuppressive drug (mainly mycophenolic acid) at the onset of the disease, did not recover from previous treatment once they clinically recovered from the infection.
Renal function and anti-HLA antibodies before, during and after SARS-CoV2 infectionMean creatinine before COVID19 diagnosis, at diagnosis, at 7 and 15 days and at 2 and 6 months was 1.7 ± 0.8 mg/dl; 2.1 ± 1.2 mg/dl; 1.8 ± 1.1 mg/dl; 1.8 ± 1 mg/dl; 1.7 ± 0.9 mg/dl and 1.8 ± 1 mg/dl, respectively (p < 0.001).
Proteinuria was only available in most patients before diagnosis and at 2 and 6 months after diagnosis, with mean values of 217 ± 530 mg/g Cr, 220 ± 510 m g/g Cr and 178.1 ± 447 mg/g Cr, respectively (p = 0.64).
Fifty-seven percent of patients (N = 266) admitted to the hospital and 8.1% of patients (N = 12) followed on an outpatient basis presented with ARF. According to the ARF KDIGO classification, 78.8% (N = 219) were stage 1, 1.8% (N = 5) stage 2, and 16.5% (N = 46) stage 3. In 8 patients the stage of ARF could not be established. 8.6% of patients required renal replacement therapy (RRT) during the infectious disease process (N = 24).
Among admitted patients the most frequent cause of renal failure was functional impairment (70%, N = 186) followed by obstructive and immunological causes in 1.5% (N = 4) and 1.5% (N = 4), respectively; other causes were diagnosed in 4.1% (N = 11). In 22.9% (N = 61) of patients, no cause for deterioration was specified. Among outpatients ARF was of functional origin in all of them and all recovered their basel renal function.
There were 10 renal biopsies performed in the context of ARF and SARS-CoV2 infection. In 4 of them the histological diagnosis was of an immunological nature, in 2 of them acute tubular necrosis and in the other 4 the causes were toxicity by calcineurin inhibitors in 2 patients, cortical ischemia in one and chronic vascular lesions in another patient. There was no evidence of the presence of viral particles in any of the renal biopsies performed.
In the 4 patients with histological diagnosis of an immunological nature, T cell-mediated rejection was observed in 3 of them and in one of them had active antibody-mediated rejection. The patients who developed cell rejection had been transplanted for 15 days, one month and 4.7 months, which means an incidence of cell rejection in the first 6 months after transplantation of 6.9%; while the patient with humoral rejection had been transplanted for 7.2 years. Management of immunosuppression during SARS-CoV2 infection in these patients consisted of in discontinuation of mycophenolic acid in all 4 patients. Tacrolimus treatment was not discontinued in any of them (2 of them maintained the same dose of tacrolimus and the other 2 had their dose reduced). In relation to steroids, 3 of them had their dose increased. Only one of the patients with acute rejection presented with donor-specific anti-HLA antibodies within 2 months of COVID19 diagnosis. In the other 3 patients, in one of them the anti-HLA antibodies remained negative at 2 and 6 months after the diagnosis of COVID, in another patient the anti-HLA antibodies became negative after the treatment received, and the other patient maintained positive anti-HLA antibodies.
In hospitalized patients, renal function was recovered in 93.2% of patients with ARF (N = 205), in 2.3% it was partially recovered, and 3.6% (N = 8) did not recover from acute kidney damage.
The average hospital stay of patients with ARF was significantly higher than that of patients without ARF (22.1 ± 22.3 days vs. 14.2 ± 12.1 days, p < 0.001). Patients who developed ARF had significantly higher mean creatinine values before diagnosis of coronavirus infection, at diagnosis and at 2 and 6 months post-infection than patients who did not develop acute kidney damage (before: 2 ± 0.9 vs. 1.5 ± 0.5; at diagnosis: 2.7 ± 1.3 vs. 1.6 ± 0.8; at 2 months: 2.1 ± 1.1 vs. 1.5 ± 0.6; at 6 months: 2.1 ± 1 vs. 1.5 ± 0.7, p < 0.001, respectively). Likewise, during the infection process, patients admitted with ARF presented a mean maximum value of creatinine significantly higher than that of patients admitted without ARF (3.1 ± 1.5 vs. 1.6 ± 0.8, p < 0.001), with the mean peak creatinine values according to the etiology of ARF being the following: 3.1 ± 1.4 in functional cases, 4.1 ± 3.1 in obstructive cases, 6.1 ± 3.1 in immunological cases, 5.1 ± 2.1 in other causes and 3.5 ± 1.2 in cases of ARF without specified cause. In the case of patients followed as outpatients, the mean maximum creatinine during infection was 2.4 ± 1 in patients with ARF and 1.7 ± 1.2 in patients who did not develop ARF, but these differences were not statistically significant (p = 0.13). The group of patients who required RRT during infection had a significantly higher baseline creatinine before the diagnosis of COVID19 than patients who did not require RRT (4.04 ± 1.9 vs. 2.6 ± 1.2, p < 0.001).
Twelve patients (1.9%) lost renal graft and 11 of them had presented with an ARF during admission. The causes of graft loss were: graft CKD progression in 5 patients, vascular in 2 patients, immunological in one patient, and other causes in 4 patients. The mean baseline creatinine of these 12 patients was 3.8 ± 2 mg/dl (minimum 1.19 mg/dl–maximum 6.99 mg/dL). Both the group of patients who required TRS-CHK and the group of patients who lost the graft during the course of the infectious disease already had advanced renal function before diagnosis, which could influence these results.
Renal function was also analyzed in the group of patients on tacrolimus receiving antiviral treatment with lopinavir/ritonavir (N = 46). Of these patients, only 36 had data on immunosuppression levels during admission. A tacrolimus trough level ≥10 ng/ml was considered as a cutoff point for high tacrolimus levels. Eighteen patients treated with lopinavir/ritonavir presented high levels of tacrolimus and of these 77.8% (N = 14) presented ARF with maximum creatinine values during admission of 2.2 ± 1.1 mg/dl, while the other 18 patients presented tacrolimus levels <10 ng/mL, representing 83.3% (N = 15) of ARF patients with maximum creatinine figures of 2.8 ± 1.4 mg/dl (there was no significant difference between maximum creatinine concentrations in the two groups).
Fifty-six point nine percent of patients (N = 350) had anti-HLA antibody monitoring before and after SARS-CoV2 infection and in 94% of these patients (N = 329) presented no changes in anti-HLA antibodies after infection.
Twenty-one patients (6%) tested positive for anti-HLA after infection, of which 9 (2.6%) patients had donor-specific anti-HLA antibodies. Of these patients, 7 had discontinued an immunosuppressive drug during infection (mycophenolic acid was discontinued in 5 of them and tacrolimus discontinued in 2), one patient had discontinued the 2 immunosuppressive drugs and the other patient had not changed in immunosuppression.
DiscussionIn this collaborative multicenter study, we have shown that the management of immunosuppression in kidney transplant patients with COVID19 consisted of the suspension of mycophenolic acid in 73.4% of patients, an increase in the dose of steroids in 50.9%, the use of methylprednisolone as part of the anti-inflammatory treatment of infectious disease in 26.9% reduction and discrete reductions (17.7% for tacrolimus and 36.7% for cyclosporine A) or discontinuation (10.5% for tacrolimus and 10% for cyclosporine A) of very discrete reduction of calcineurin inhibitors.
These supervised and intentional changes in immunosuppressive therapy have not resulted in functional or immunological damage to the renal graft in the majority of patients. It is possible that the reduction in immunosuppression performed in parallel with the increase in the dose of steroids may offset the risk of rejection or graft loss resulting from this practice.
Although there is no consensus on the management of immunosuppression in these patients, the reduction of immunosuppression has been postulated as the mainstay of the therapeutic management of these patients in the absence of specific antiviral treatment, and this reduction is usually done according to the clinical situation of the patient.13,14
In fact, in an international study that surveyed immunosuppression management during the first months of the pandemic in 513 transplant programs in 71 different countries, patients with moderate COVID19 symptoms (in need of hospital admission, not ICU) treatment with antimetabolites and calcineurin inhibitors was decreased or suspended in 76% and 45.4% of patients respectively. In the case of patients with severe symptoms (admission to the ICU), these percentages of suspension or reduction of antimetabolites and calcineurin inhibitors amounted to 79.5% and 68.2%, respectively. In relation to steroids, there was a parallel increase in dose of up to 30.6% and 46% of patients with moderate and severe symptoms of COVID19, respectively.15
Compared to our study, Sandal et al.15 reported a greater tendency to reduce or discontinue calcineurin inhibitors with similar percentages in the change of antimetabolites and steroids. This may be due to the design of the study, carried out through a survey with questions on the management of immunosuppression without collecting data on the final management carried out in each center. In fact, in our study there is a percentage of patients in whom having considered suspending or reducing calcineurin inhibitors it was not formally achieved and were considered as patients without changes in immunosuppression or with failed suspension.
Other studies, published mainly after the first wave of the pandemic, also show that changes in immunosuppression during infection did not influence kidney function or the immunological status of patients,7 as is the case in our study. Although the follow-up time after COVID19 cure is short (3 months in the published studies7–11 and 6 months in our study) for analyzing the effect of stopping immunosuppression on graft survival, it does allow us to know its effect on renal function and HLA sensitization that directly influence long-term graft survival.
In our study, the incidence of ARF after SARS-CoV2 infection in renal transplant patients is common, occurring in 57% of patients admitted to the hospital and almost 10% of outpatients. Similarly, a systematic review16 analyzing the evidence on clinical evolution, mortality and ARF in renal transplant patients with COVID19 shows that the incidence of ARF is common and occurs in 50% of cases (95% CI: 44–56%). Although the incidence of ARF seems lower in the meta-analysis compared to our study, the variability between studies is large (I2: 66%), which indicates that there may be clinical or methodological differences between the studies.
The high incidence of ARF in renal transplant patients with COVID19 is probably of multifactorial origin and is influenced by factors associated with the severity of the infectious disease - such as hemodynamic instability, respiratory distress, exposure to nephrotoxic drugs, secondary infections, hypoxia and hypovolemia or cytokine storm - as well as factors related to the management of immunosuppression that may favor the setting in motion of immunological rejection mechanisms in the renal graft. In our study, the main cause of ARF was functional etiology, which led to recovery from ARF in most patients; however, in those cases (10 patients) with renal graft biopsy, the most common histological diagnosis was acute rejection in 4 of them, with T-cell-mediated acute rejection being the most frequent (in 3 patients). In all 4 cases, the reduction in immunosuppression consisted of the suspension of mycophenolic acid and in addition in 2 of them the dose of tacrolimus was reduced. Only one of the patients developed donor-specific antibodies within 2 months of infection. Histological evidence of acute rejection in the context of COVID19 has been previously described in other multicenter studies, with cellular rejection being the most common of the rejection diagnoses17,18
In our study, once the ARF was resolved, the patients' renal function returned to its previous creatinine levels and remained stable after 6 months of COVID19 infection, with no changes in the same in the medium term. However, in the general population, patients who develop COVID19−associated ARF are described as having a higher risk of progressive loss of renal function between 7 and 90 days after the initial ARF event than those patients who develop non-COVID19-associated ARF. It is very likely that the close follow-up of transplant patients after the COVID19 episode, in our study, positively influenced that patients who had an ARF did not develop in the medium term the deleterious effects of the same on renal function, as described in the work of Nugent et al.19 Therapeutic measures such as optimal blood pressure control, avoiding the use of nephrotoxic drugs, the introduction of drugs that block the renin-angiotensin-aldosterone system, and adequate control of immunosuppression after resolution of infection have been able to help achieve these results in our cohort of transplant patients, and many of them could serve as opportunities for improvement in the early follow-up of patients in the general population who have developed ARF associated with COVID19.
Although the presence of ARF in COVID19 is associated with a worse prognosis of the disease and a higher risk of mortality, in our work we have not been able to perform this analysis since only patients who survived the disease have been included in the study, excluding patients who died during the disease.
Our study includes a very large number of kidney transplant patients, indeed one of the largest samples described in the literature and covered most of the kidney transplant patients infected by COVID19 in Spain during the first and second waves of the pandemic. Given its descriptive nature, it is of great value for the administration of health resources and for the generation of working hypotheses for other studies in this population of patients so vulnerable to infections. The clinical data come from a national registry created only 2 weeks after the first case of COVID19 was diagnosed in a kidney transplant patient, which allowed the rapid and homogeneous collection of the main study variables by each researcher, providing validity to the study. However, it has some limitations; its retrospective design does not allow establishing causal associations and in addition, the special characteristics of the study period (pandemic status with hospitals dedicated to the care of numerous patients with a new infectious disease) meant that some of the study variables, in particular the determination of proteins in urine or the levels of immunosuppressants, were not analyzed in all the established time periods, but did not affect the final results of the study.
In conclusion, the management of immunosuppression after the diagnosis of COVID19 was mainly based on the suspension of mycophenolic acid with discrete reduction or discontinuation or suspensions of calcineurin inhibitors. This management of immunosuppression did not influence renal function or changes in anti-HLA antibodies at 6 months since diagnosis.
FundingThis research has not received specific support from public sector agencies, the commercial sector or non-profit entities.
Conflict of interestsThe authors have no conflicts of interest to declare.
This work would not have been possible without the collaboration of the SEN transplant working group (SENTRA) who participated in the data collection. The names are listed in Appendix A.
Cristina Canal, Nuria Serra, Lluis Guirado, Ana Vila, Rosana Gelpi, Natalia Ridao, Carmen Díaz, Esther González, Amado Andrés, Ana Hernández, Natalia Polanco, Alba Santos García, Luis Alberto Sánchez Cámara, Nicolás Macías Carmona, Laura Llinás, Anna Buxeda, Carlos Arias, Carla Burballa, Julia Farrera, Miren Iriarte, Marta Crespo, Francisco Llamas Fuentes, Inmaculada Lorenzo González, Pilar Fraile, Elena González García, M. Auxiliadora Mazuecos, Emilio Rodrigo, Pilar Fraile, Beatriz Rodríguez Cubillo, Natividad Calvo Romero, M. Ángeles Moreno de la Higuera, Ana Sánchez Fructuoso, Patricia Martínez.