Chronic kidney disease (CKD) is increasing in patients older than 65 years and is related to morbidity, frailty, and dependence. Peritoneal dialysis (PD) has classically been associated with young patients with an active life.
HypothesisPD should be offered to patients over 65 years. We search for any unfavorable results that may advice not to recommend PD therapy for this group.
ObjectiveTo describe PD treatment and outcomes in patients > 65 years, to compare their results with patients < 65 years and to identify areas with room for improvement in a real-life study.
StudyProspective, observational, and multicenter study performed in incident PD patients, from January 2003 until January 2018.
ResultsWe included 2,435 PD patients, 31.9% were older than 65 years; there was a difference of 25 years between both groups. Median follow up was 2.1 years. Older than 65 years group had more comorbidity: Diabetes (29.5% vs 17.2%; p < 0.001), previous CV events 34.5% vs 14.0%; p < 0.001), Charlson index (3.8 vs 3.0; p < 0.001).
We did not find differences in efficacy and PD adequacy objectives fulfillment, anaemia management or blood pressure during follow-up. Peritonitis rate was higher in older 65 years group (0.65 vs 0.45 episodes/patient/year; p < 0.001), but there was not differences in germs, admission rate and follow up. Mortality was higher in older 65 years group (28.4% vs 9.4%) as expected. PD permanence probability was similar (2.1 years). The main cause of PD withdrawal was transplant in group < 65 years (48.3%) and transfer to HD in group > 65 years. The main reason was caregiver or patient fatigue (20.2%), and not technique failure (7.3%).
Multivariate Cox regression analysis showed a relation (HR [95%CI]) between mortality and age > 65 years 2.4 [1.9-3.0]; DM 1.6 [1.3-2.1]; CV events 2.1 [1.7-2.7]. Multivariate Cox regression analysis identify a relation between technique failure and age > 65 years 1.5 [1.3-1.9]; DM 1.6 [1.3-1.9] and previous transplant 1.5 [1.2-2.0].
ConclusionPatients older than 65 years fulfilled PD adequacy criteria during the follow up. We believe PD is a valid option for patients older 65 years. It is necessary to try to prevent infections and patient/caregiver fatigue, to avoid HD transfer for reasons not related to technique failure.
La prevalencia de enfermedad renal crónica (ERC) aumenta en población mayor de 65 años y asocia morbilidad, dependencia y fragilidad. La diálisis peritoneal (DP) se ha considerado una técnica de paciente joven y vida active.
HipótesisLa DP puede ser adecuada en pacientes de edad avanzada. Buscamos resultados desfavorables que contravengan esta hipótesis.
ObjetivoDescribir el tratamiento con DP en mayores de 65 años, evaluar su evolución clínica comparada con los menores de 65 e identificar áreas de mejora asistencial.
EstudioProspectivo, observacional y multicéntrico en incidentes en DP, seguimiento hasta evento o fin del estudio (ene-2003 a ene-2018).
ResultadosSe incluyen 2.435 pacientes; el 31,9% (777) eran mayores de 65 años. El tiempo medio de seguimiento fue de 2,1 años para ambos grupos. El grupo de edad avanzada era 25 años mayor, con más comorbilidad: diabetes (29,5% vs. 17,2%; p < 0,001), evento CV previo (34,5% vs. 14,0%; p < 0,001) e índice de Charlson sin edad (3,8 vs. 3,0; p < 0,001). No encontramos diferencias en cumplimiento de objetivos intermedios de eficacia de DP, control de anemia o hipertensión durante el seguimiento. La tasa de peritonitis fue mayor en la cohorte mayor de 65 años (0,65 vs. 0,45 episodios/paciente-año; p < 0,001), aunque la distribución gérmenes, tasa de ingreso y evolución final fue similar en ambos grupos. Lógicamente, registramos mayor mortalidad en el grupo mayor de 65 años (28,4% vs. 9,4%), aunque el tiempo de permanencia en DP fue similar (2,1 años). La principal causa de salida fue el trasplante renal en jóvenes (48,3%), mientras que en los pacientes de mayor edad fue el paso a hemodiálisis, principalmente por cansancio de cuidador/autocuidado (20,2%) y no por fallo de la técnica (7,3%). El análisis multivariante por regresión de Cox identifica los factores de riesgo para mortalidad con los siguientes HR [IC 95%]: Edad > 65: 2,4 [1,9-3,0]; DM: 1,6 [1,3–2,1]; CV: 2,1 [1,7–2,7]. Y para el evento fallo de la técnica y paso a HD: Edad > 65: 1,5 [1,3–1,9]; DM: 1,6 [1,3–1,9]; trasplante renal previo: 1,5 [1,2-2,0].
ConclusiónLos pacientes mayores de 65 años cumplen los objetivos de calidad intermedios y se mantienen en DP el tiempo suficiente como para seguir ofertando esta técnica. Es necesario mejorar la prevención de infecciones y del agotamiento del paciente/cuidador con medidas de soporte para evitar la transferencia a HD por causas ajenas a las complicaciones de la técnica.
Stage 5 chronic kidney disease ( requiring renal replacement therapy (RRT) is associated with high morbidity and mortality and implies a change in the type of life in both hemodialysis (HD) and peritoneal dialysis (PD). Patients are forced to modify their lifestyle habits to adapt to the RRT they have chosen, so the emotional impact of the disease is important. PD favors the principles of autonomy and independence and avoids trips to the hospital, with an improvement in the patient's self-perception and in their quality of life.1
Classically, PD has been associated to young patients that are able to maintain their employment and to reconcile work and personal life.2 As we have gained experience with the favorable evolution of aging of PD patients, we have learned to offer RRT increasingly elderly patients, thus the age of PD patients has been increasing in parallel to that of the general population.3 This is why many end stage renal disease units (ESRD) proposed PD therapy to the elderly patients after an individualized assessment.4
The latest registry of kidney patients proposes age limits at 45, 65 and 75 years and confirms that the percentage of patients who choose PD decreases as the age of the patient increases. But a review the actuarial trend of the last 10 years, shows that the prevalence of PD in elderly patients has grown more than in young patients. The Spanish registry (REER) reflects an increase of 17% in the incidence of PD in patients older than 65 during the last 5 years.5 In this scenario, some nephrologists still consider that there is no evidence to offer the PD to the elderly patient and we do not have structured and specific recommendations to encourage PD as a free choice in this population.6
The Peritoneal Dialysis Center Group (GCDP) includes 25 hospitals in the country. Each hospital unit incorporates all patients starting PD from a area equivalent to 9.5 million people.7 This group constitutes an excellent base of current and forthcoming information on the actual use and the clinical results of PD in our country.
Although we assume that a patient aged 65 can remain active with a good quality of life and a life expectancy of more than 20 years, we have chosen this cut-off point because it is common in all published records and constitutes the limit established by the Spanish Society of Geriatrics and Gerontology. The aim of this paper is to describe the clinical characteristics, management and evolution of patients older than 65 years that start PD. We intend to analyze the guidelines, the final clinical results of survival of these patient and the technique and the intermediate results of efficacy, control of anemia and incidence of peritonitis, as well as to explore related prognostic factors.
Material and methodsType of studyThis is a prospective, observational and multicenter study of incident patients on PD carried out by the GCDP.
Study populationA consecutive systematic inclusion of all patients older than 18, incidents in the PD technique, was carried out in the database previously described.7 The recruitment period began in 2003 and ended in December 2017. They were categorized into two groups depending on whether they were older or younger than 65 years. The patients were followed up until the end in this technique due to transplantation (Tx), transfer to HD or death. There was a 2.2% loss to follow-up due to transfer to hospitals outside the GCDP. The patients gave their consent for inclusion in PD and the study was approved by the Research Ethics Committee (CEIm) of the Puerta de Hierro University Hospital (PI 139/15).
VariablesAt the beginning of the treatment with PD, the following characteristics of the patients were collected: age, gender, body mass index, etiology of kidney disease, history of diabetes and cardiovascular events (CV), Charlson comorbidity index without age, inclusion in the waiting list for kidney transplantation and treatment before starting PD (ESRD unit, transplantation, HD). Likewise, we collected the modality of PD they started with: manual (CAPD) or automated (APD).
During the follow-up, the following information was collected: peritoneal functionality profiles, dialysis dose, modality (APD or CAPD) and intermediate indicators such as anemia, blood pressure, need to be transfer to HD and reason for discharge. Blood pressure goals were defined as SBP < 140 mmHg and DBP < 90 mmHg as previously described.6
The final events of death, failure of the technique or transplantation were incorporated into the database by adding the cause of death detailed and categorized into groups (cardiovascular, infectious, tumor, others) and the reason for failure of the PD technique.
Peritoneal infection episodes were also recorded as intermediate events to estimate incidence rates. The causative germ and the final evolution of the episode were collected, stratified as cure, catheter removal, recurrence and death from peritonitis. In relation to death from peritonitis, there were considered both those described by the clinician with that as cause of death and any death within 30 days of the onset of infection.
We present these variables globally and stratified by age groups in > 65 years and ≤ 65 years.
Follow-upThe follow-up time was from the inclusion of patients in PD therapy until their discharge due to kidney transplantation, change to HD, death or the end of follow-up in December 2018.
Statistical analysisThe quantitative variables were described as mean and standard deviation (SD) if they had a normal distribution and as median and interquartile range (IQR) if they did not have a normal distribution. The categorical variables were expressed as percentage. For the comparison of variables, a Chi-square, t-Student or t-Student for paired samples was used according to the nature of the variables. An analysis was carried out according to the Kaplan-Meier model to estimate the probability of survival and maintenance in technique. The comparison of events over time was performed with a univariate regression and subsequently with a multivariate model by backwards steps, retaining variables with pIN and pOUT of 0.1. Statistical significance was accepted with a p < 0.05. For data processing and analysis, the Stata program (v14 Stata Corp, TX, USA) was used.
ResultsPatients characteristicsWe included a total of 2,435 patients, of whom 777 (31.9%) were older than 65 years and 1,658 (68.1%) were younger than 65 years. The descriptive characteristics of the entire population and stratified by age groups can be seen in Table 1.
Baseline characteristics of the study population stratified by age groups.
| Total (N = 2.435) | ≤ 65 years (N = 1,658; 68%) | > 65 years (N = 777; 32%) | p | |
|---|---|---|---|---|
| Age (years) | 55.9 (15.4) | 47.9 (11.7) | 72.9 (5.4) | < 0.001 |
| Males (%) | 64.5 | 63.3 | 67.1 | 0.06 |
| Previous CV event (%) | 20.5 | 14.0 | 34.5 | < 0.001 |
| Diabetes Mellitus (%) | 21.1 | 17.2 | 29.5 | < 0.001 |
| Charlson index (without age) | 3.3 (1.7) | 3.0 (1.6) | 3.8 (1.8) | 0.01 |
| Etiology CKD (%) | < 0.001 | |||
| Diabetes | 18.6 | 15.3 | 25.9 | |
| Vascular | 11.0 | 7.9 | 17.6 | |
| Glomerular | 24.3 | 28.3 | 15.7 | |
| Polycystic kidney disease | 11.1 | 13.8 | 5.3 | |
| Not affiliated / other | 35.0 | 34.7 | 35.5 | |
| Origin (%) | <0.001 | |||
| ESRD unit | 73.9 | 70.1 | 81.8 | |
| Hemodialysis | 18.1 | 19.5 | 15.1 | |
| Previous transplant | 8.1 | 10.4 | 3.1 | |
| RRF (ml / min) | 7.0 (4.4) | 7.0 (4.4) | 7.0 (1.0) | |
| APD at baseline (%) | 30.2 | 35.6 | 18.5 | <0.001 |
| Active on waiting list Tx (%) | 40.5 | 51.1 | 17.9 | <0.001 |
Values are expressed as mean and standard deviation or percentage.
CV: cardiovascular; APD: automated peritoneal dialysis; RRF: residual renal function; Tx: transplantation; ESRD: end stage renal disease.
The mean age of the older group was 72.9 (5.4), with a range of 65 to 91 years, and mean age of the younger patients was 47.9 (11.7), with a range of 18 to 65 years. Comparing the two groups, those over 65 years had a history of previous cardiovascular events, diabetes and Charlson comorbidity. A higher percentage of patients younger than 65 years were started with a cycler (Table 1).
Dialysis dose and technique survivalThe use of PD was increased significantly in both groups during follow-up; in group >65 65 years it increased from 18.5 to 29.4% (p = 0.001), while in the <65 years it increased from 35.6 to 48.0% (p = 0.001). The dose of dialysis in both groups was similar and remained within objectives at the start of PD and during the one-year follow-up. In those younger than 65 years old, the basal Kt / V was 2.5 (SD 0.7) and at one year it was 2.4 (SD 0.7) (p = 0.1). Similar results were obtained in the group over 65 years; the baseline Kt / V was 2.5 (SD 0.7) and at one year it was 2.4 (SD 0.7) (p = 0.3). There were no differences in the weekly creatinine clearance values.
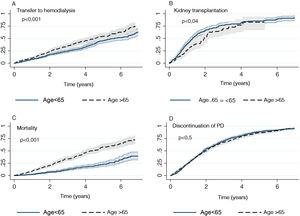
A greater failure of the technique and transition to HD was evidenced in the group > 65 years in the Kaplan-Meier analysis (median 5.6 years 95% CI [5.0-6.5] vs. 4.5 years [4.0 to 5.4]; p < 0.001) (Fig. 1). The reasons for the change in technique from PD to HD stratified by age groups are shown in Table 2. Social problems of self-care stand out in the group over 65 years. We found an independent association between the failure of the technique with transfer to HD and age > 65 year corrected for other comorbidities such as diabetes (Table 3).
Incidence of accumulated events for both groups (> 65 years and ≤ 65 years) of study. A) Transfer to hemodialysis; B) Kidney transplantation in those patients on the waiting list; C) Patient mortality, and D) Discontinuation of PD for any reason. Kaplan-Meier curve is presented with 95% confidence interval and p value.
Clinical outcomes of patients on peritoneal dialysis stratified by age.
| ≤65 years | > 65 years | p | |
|---|---|---|---|
| Exit causes (%) | <0 .001 | ||
| Permanence in PD | 17.3 | 20.3 | |
| Death | 9.3 | 28.5 | |
| Change to HD | 20.6 | 30 | |
| Transplantation | 48.2 | 17 | |
| Transplant of those on the waiting list | 95.4 | 95.8 | |
| Causes of failure of technique (%) | < 0.001 | ||
| Mechanical problems a | 18.7 | 12.0 | |
| Infectious | 27.2 | 26.6 | |
| Technique failure b | 16.7 | 7.3 | |
| Comorbidity | 9.6 | 13.3 | |
| Social / exhaustion c | 12.0 | 20.0 | |
| Others / unknown | 15.0 | 20.0 |
PD: peritoneal dialysis; HD: hemodialysis.
Factors associated with failure of the technique.
| Univariate | Multivariate | |
|---|---|---|
| HR [95% CI] | HR [95% CI] | |
| Previous CV disease | 1.4 [1.1-1.6] | |
| Diabetes Mellitus | 1.7 [1.4-2.0] | 1.6 [1.3-1.9] |
| Age >65 years | 1.4 [1.2-1.7] | 1.4 [1.2-1.7] |
| Charlson index | 1.1 [1.0-1.2] | |
| Peritonitis | 1.2 [1.0-1.4] | |
| Previous treatment | Previous HD: 1.1 [0.9-1.4] | Previous HD 1.2 [0.9-1.4] |
| Ref. ESRD | Previous Tx: 1.3 [1.1-1.9] | Previous Tx: 1.6 [1.2-2.0] |
CV: cardiovascular; HD: hemodialysis; HR: hazard ratio; CI: confidence interval; Tx: transplantation; ESRD: end stage renal disease units.
There are no differences in the time spent in PD treatment (median follow-up of 2.1 years, Fig. 1), but there are differences in the causes of exit. The main reason in ≤ 65 years was the transplant, and in the group > 65 years the causes for discharge are more diverse (Table 2). After 4 year follow up the percentage of patients on the waiting list who agreed to a effective kidney transplant was the same in both groups, but the waiting time was slightly shorter in younger than in older (median probability kidney transplantation: 1.3 years 95% CI [1.2-1.5] vs. 1.9 years [1.5-2.2], respectively, Fig. 1).
Intermediate endpoints and incidence of peritonitisNo differences were found in the correction anemia in both groups (hemoglobin > 11.5 g / dl: 62.6% vs. 60.9%; p = 0.5) or in the control of hypertension (BP< 140/90 mmHg: 46.9% vs. 45.2%; p = 0.4, Table 4). Some episodes of peritoneal infection was recorded in 1,077 patients, it was more frequent in the elderly than in the young (54.6% vs. 39.4%; p < 0.001). The rate of peritonitis per year at risk was greater in the group > 65 years (rate in < 65 years: 0.45; and in > 65 years: 0.65; overall rate: 0.51 episodes / patient -year at risk; p = 0.001). There were no differences between the two groups in the evolution of peritonitis (Table 4) or in the type of causative germ, a 57% were gram-positive infections in the elderly compared to 56% in the younger ones. There were also no differences with the percentage of exit site infection: 9.7% versus 9.5%, respectively (p = 0.5).
Compliance with intermediate objectives stratified by age.
| ≤ 65 years | > 65 years | p | |
|---|---|---|---|
| Peritonitis rate / patient-year at risk | 0.45 [0.42-0.47] | 0.65 [0.62-0.70] | < 0.001 |
| Healing (%) | 80.5 | 83.4 | |
| Recurrence (%) | 8.3 | 6.8 | 0.5 |
| Catheter removal (%) | 10.9 | 9.3 | |
| Death (%) | 0.2 | 0.5 | |
| Gram positive (%) | 55.9 | 57.6 | 0.87 |
| Gram negative (%) | 21.1 | 20.7 | |
| Fungi / polymicrobial (%) | 2.1 / 3.1 | 2.4 / 3.2 | |
| Sterile (%) | 17.8 | 16.1 | |
| Exit site infection (%) | 9.4 | 9.7 | 0.5 |
| Efficacy targets (basal / year) | |||
| Target Kt / V (%) | 82.6 / 83.6 | 85.9 / 86.6 | 0.1 / 0.2 |
| Mean Kt / V (SD) | 2.5 (0.7) / 2.4 (0.7) | 2.5 (0.7) / 2.4 (07) | 0.9 / 0.3 |
| Anemia targets (basal / year) | |||
| Hb> 11,5 g / dl (%) | 62.6 / 63.8 | 60.9 / 69.1 | 0.5 / 0.05 |
| Mean Hb, g / dl (SD) | 12.0 (1.5) / 12.1 (1.5) | 11.9 (1.4) / 12.1 (1.5) | 0.7 / 0.4 |
| Targets BP (basal) | |||
| BP < 140 / 90 (%) | 46.9 | 45.2 | 0.4 |
| SBP mmHg (SD) | 135.4 (19.2) | 139.6 (21.3) | <0.001 |
| DBP mmHg (SD) | 83.3 (12.1) | 75.6 (11.3) | <0.001 |
SD: standard deviation; Hb: hemoglobin; BP: blood pressure; DBP diastolic blood pressure; SBP: systolic blood pressure.
Mortality in the elderly group was higher, with a median time to event of almost 3 years less (4.5 years 95% CI [3.9-4.9] vs. 8.4 years [7.0-11.4]) (Fig. 1). The causes of mortality in the group over 65 years were in order of frequency were cardiovascular (36.6%), unknown (36.0%), infectious (23.8%) and tumor (3.7%). Cause mortality did not differ from those of the group ≤ 65 years (Table 5).
Univariate and multivariate analysis of risk factors of mortality.
| Univariate | Multivariate 1 | Multivariate 2 | |
|---|---|---|---|
| HR [95% CI] | HR [95% CI] | HR [95% CI] | |
| Previous CV disease | 3.2 [2.6 - 3.9] | 2.2 [1.7 - 2.7] | 2.1 [1.7 - 2.7] |
| Diabetes | 2.4 [2.0 - 3.0] | 1.6 [1.3 - 2.1] | 1.7 [1.4- 2.2] |
| Age > 65 years | 3.1 [2.5 - 3.8] | 2.5 [2.0 - 3.1] | |
| Charlson index | 1.3 [1.3 - 1.4] | ||
| Peritonitis | 1.0 [0.8 - 1.2] | ||
| Previous treatment (Ref. ESRD) | Previous HD: 1.5 [1.2-1.9] | 1.6 [1.2 - 2.0] | 1.5 [1.2 - 2.0] |
| Previous Tx: 0.8 [0.5-1.2] | 1.2 [0.8 -1.8] | 1.3 [0.9 - 2.0] | |
| Age > 70 | 3.0 [2.5 - 3.7] | ||
| Age 65-70 ; Age >70 (Ref. Age <65) | 2.2 [1.6 - 3.0] | 1.7 [1.2 - 2.3] | |
| 3.5 [2.8 - 4.4] | 2.9 [2.3 - 3.6] |
CV: cardiovascular; HD: hemodialysis; HR: hazard ratio; CI: confidence interval; Tx: transplantation; ESRD: end stage renal disease units.
In the univariate Cox model, we found an association between mortality and: the elderly group (> 65 years), previous CV disease, the presence of diabetes, the elevated Charlson index, the modality of initiation with CAPD, and the presence of peritonitis. However, in the multivariate Cox model it was found an independent association between mortality and edad > 65 years (HR 2.4, 95% CI [1.9-3.0], previous cardiovascular events (2, 2 [1.7-2.7]) and diabetes mellitus (1.6 [1.3-2.1]). There was no independent association between mortality and the development of peritonitis or the type of automatic or manual technique.
DiscussionOur study is the first specifically dedicated to describing the results of PD on a large contemporary database that includes almost 800 patients older than 65 years, distributed in 5 regional communities of our country. As a whole, we found that the permanency in the technique in older patients is similar to that of younger patients with intermediate results of similar quality and a good access to transplantation for those included in the waiting list. Although, as expected, mortality was higher in the group that was older than 65, we did not find any results that prevent us from continuing to consider PD as an adequate and effective technique in patients older above 65years of age.
We consider that these results should be sufficient to continue recommending the free choice of dialysis from patient from the ESRD units without establishing a strict age limit, but rather using individualized evaluation based on the overall characteristics of the patient, their comorbidity and frailty.8,9 Among elderly patients there is a wide range of profiles, from the frail patient who needs help for their care to the patient without associated pathology or social problems who is defined as a healthy elderly person.10,11 Based on these characteristics, it is our task to adapt the learning process and type of technique to each situation.12 During the last century, life expectancy and quality of life have increased progressively and in fact the retirement age has been progressively postponed. Today, a 65- year- old person can remain active with a good quality of life and a life expectancy of more than 20 years, although this is not always the case in CKD. We have maintained this cut-off point in 65 years old because it is commonly see in publications and constitutes the limit established by the definition of age for action by the Spanish Society of Geriatrics and Gerontology. The estimated risk of death for people over 65 years old allows for comparisons with other registries, and in the sub-study by age group we observe how the risk increases progressively in the groups 65-70 and older than 70 compared to the younger.
We must personalized the modality and pattern of PD, especially in older patients, seeking a balance between dose of dialysis, the ability to maintain self - care and quality of life.13 Cyclers seem to be the technique of choice for young people in our environment, while starting with the manual technique is preferred for older patients; perhaps this is due to its simplicity and more time available without work obligations. The vast majority of patients over 65 achieve a Kt / V greater than 2, similar to that of younger patients, and this is maintain during the follow-up. This observation had been highlighted previously by other groups, with a more limited sample size.3 In older patients, we did not find difficulties in achieving other intermediate goals, such as adequate control of blood pressure or anemia. We have not found publications of specific studies on the control of anemia in patients over 65 on PD, but according to the 2015 USDRD report, 80% of PD patients received erythropoietic agents (ESA) and had a mean hemoglobin of 10.9 g / dl, 55% of them 10-12 g / dl, 21% > 12 g / dl and 23% < 10 g / dl.14 In the same registry, 92% of the HD patients required ESA and had lower hemoglobin levels.
Regarding peritonitis some results are controversial, several studies report a high rate of peritonitis in the elderly population.15 In the work by Wu et al, not only the risk of infection is increased four times, but also the mortality secondary peritonitis is significantly increased.15 However, although our older patients had a higher incidence of peritonitis than our younger ones, the episodes were not more severe. In fact, they did not have an appreciable impact on technique failure or mortality, and therefore did not appear as a risk factor in any of the multivariate regression models. An individual evaluation of the risk of peritonitis in elderly patients would allow us to anticipate the need for re-training plans or support measures to reduce infections.
In our experience, training older people implies more time and dedication, and the application of adapted protocols with more clear and simple instructions, and fostering empowerment. Retraining is also desirable, either after peritonitis or periodically, depending on the protocols of the dialysis unit. All these measures can help to reduce the rate of peritonitis and its impact on the technique and quality of life.
PD promotes self-care and autonomy in the elderly patient, and the results in terms of doses of dialysis support the notion that this therapy is also recommended in older patients.6 If the clinical situation of the older patient is not adequate, the correct approach would be to propose other techniques; however, we found no reason to discourage PD in patients just by virtue of being older than 65 years.
In our study, the probability of failing PD is higher in the elderly, diabetics and those who have had a previous failed transplant. Diabetes is a known to be poor prognostic factor and failed transplantation is usually associated with early loss of residual kidney function and other complications.16,17 Despite all this, the effective time that our patients spend on PD is around 2 years, very similar to that of young people. One of the most interesting findings, shown in the literature, has been to observe that technique survival in the older age group may be similar to that of younger patients.18 We have found that a significant percentage of patients abandon the technique due to a social problem, or exhaustion of themselves or the caregiver while they have no technical reasons such as insufficient dose, filtration failure, leaks or infections. For various reasons, assisted PD has not been developed in Spain. However, other countries have been able to find models based on the support of caregivers who are trained to perform the complete technique, or to perform the connection and disconnection, and that are financed by different systems.19,20 If we had this model, we could reduce dropouts due to social causes or incapacity for self-care. Our objective should be that a change from PD to HD would take place only because there are unsolvable technical failures or the express wish of the patient and not as the result of lack of home support or social resources.21 In a recent European study, the availability of assisted PD with the support of caregivers increases the use of the technique (HR 1.78 [1.21-2.61]).22
It is true that, in previous studies, the elderly patient that is dependent of some support that received assisted PD remains on PD for less time than those that are autonomous and use conventional PD.23 But this may be due to a negative selection bias of assisted PD with greater comorbidity and frailty. In fact, in our health care model, most of these dependent patients would be on HD at a center or hospital.
In any case, it is acceptable to consider that in patients, due to the mere fact of aging and despite having assistance, there is a greater risk of failure of the technique,24 which is estimated to be 6% per 10 years in the study by Kumar et al. including 1,378 patients.25 However, in another study of nearly 13,000 patients, Lim et al. found that those over 65 had better survival of the technique after an event of peritonitis than the younger population.26 The authors point out that patients above 65 years have the advantage of more time available for recovery and retraining as compared to younger patients, who are sometimes limited by work and/or family load that makes difficult retraining after an event of infectious peritonitis. From this perspective, the older patient would have an advantage. We can interpret that the elderly patients have a certain advantage, however the very old, frail or needing assistance could be at a more disadvantageous position.
The ERA-EDTA registry found an increase in mortality and failure of the technique due to social difficulties in patients older than 65 years. The authors conclude that it is necessary to deepen the analysis of social factors to improve integrated support as early as possible so it could be reversible.27 While assisted dialysis is not a generalized practice in Spain, it is necessary to perform a careful and more frequent evaluation of the efficacy of this therapy in this group of patients, in order to identify failures that can be solved if detected promptly.
It should be noted that the mean age of our elderly population is close to 73 years and that at the time of initiating PD could be over 90 years. Older people with limited life expectancy could benefit from PD on a palliative basis; this has been shown in previous studies have shown.9 It is an easy technique to implement, carried out safely at home, and with good results, especially in the initial phases, so the benefit is greater than the assumable risk. An example that has become established is that of patients with advanced heart failure treated with PD improve congestion thanks to ultrafiltration.28
It is possible that we may have concerns of having patients receiving treatment at home and sometimes we prefer a face-to-face control in our HD centers. With proper training and gaining self-confidence, we will be able to give our patients the best possible quality of life; it depends, once again, on our proactive attitude.29
We have to point out certain limitations in this study; the results have been obtained from the GCDP database launched in 2003, so there are potentially relevant parameters that were not available. Among them, the following stand out: indicators of nutrition or inflammation together with elements of geriatric evaluation such as functional, social, quality of life scales, sarcopenia30 or frailty, which are not used in our daily practice.31 Also, we do not have clinical follow-up after the change to HD or transplantation. However, it is a methodologically rigorous study, with a broad territorial implementation, monitoring and timeliness, which is why it is highly representative of routine clinical practice that confers external validity.
Given all these considerations, we propose three mainstays as potential areas for improvement in our PD programs. First, adaptive training and home retraining to reduce peritoneal infections. Second, strategies of comprehensive support for patients over 65 years of age and their caregivers with a multidisciplinary team that allow us to adapt the technique to the patient and prevent the exhaustion of the caregiver and/or the patient.32 Finally, models of assisted PD in homes or residences should be explored for the subgroup of dependent patients or those who show signs of exhaustion.
We conclude that patients over 65 on PD meet adequate intermediate objectives and maintain themselves in the technique, with good quality of dialysis, long enough to continue offering PD. We have identified as opportunities for improvement the preventive management of infections and the anticipation of the exhaustion of the patient / caregiver with adequate support to avoid the discontinuation of PD for reasons unrelated to the complications of the technique. Successful models in other countries such as assisted PD could be established in our system
FinancingResearch groups integrated in the public research network REDInREN RETIC ISCIII 016/009. Co-financed by Baxter (2003-2017), Fresenius (2005-2017) and Vifor (2018-2020) through the Madrid Nephrology Foundation.
Conflict of interestsThe authors have no conflict of interest in the area of knowledge of this work.
AcknowledgementThe authors thank all the GCDP nurses and physicians who have been involved in the daily follow-up of our patients during these 15 years.
The Peritoneal Dialysis Center Group (GCDP) is made up of the following members: 1.- HUF Alcorcón: Tato A MD, Bernal Molano, MD. 2.- F. Jiménez Díaz: Martín Cleary C MD; Escribano A. 3.- H Clínico San Carlos: Vian J, MD. 4.- Hospital Clinic U de Valladolid: Sanz Ballesteros S. MD. 5.- HGU Gregorio Marañón: Vega A MD. 6.- HU Puerta de Hierro: Martínez A, Campos J. 7. HU La Paz: Bajo MA. MD, Del Peso G MD. 8.- HGU of Guadalajara: Rodríguez Palomares JR. MD. 9.- HG Ciudad Real: Carreño A MD. 10.- HNS de Sonsoles, Ávila: Felipe C MD. 11.- HU De La Princesa: Perpén A MD, Hernández MJ. 12.- HU. Prince of Asturias: Moreno F MD. 13.-HU Río Hortega de Valladolid: Sánchez García, L MD. 14.- HU Ramón y Cajal: Rivera Gorrín M. MD Burguera V. MD 15.- HG Segovia: Fernández-Reyes MJ MD, Rodríguez A, MD. 16.- H Infanta Sofía: Hevia C MD, Lorenzo M. 17.-H del Henares: Paraíso, V MD. 18.- .HU San Pedro de Alcántara: González Sanchidrián S MD. 19.-H del Sureste: Tornero F MD. 20.-H. Río Carrión de Palencia. Hernández E MD. 21.- H. Infanta Leonor: Ortega M MD. 22.-H. Miguel Servet: Campos Gutiérrez, B MD. 23.- HU Infanta Cristina: Benito J MD. 24.- H Severo Ochoa (Leganés). Ortega O MD, Herrero JC MD. 25. H Rey Juan Carlos. Pizarro MS, MD.
Please cite this article as: Portolés J, Vega A, Lacoba E, López-Sánchez P, Botella M, Yuste C, et al. ¿Es adecuada la diálisis peritoneal para pacientes mayores de 65 años? Estudio multicéntrico prospectivo. Nefrologia. 2021;41:529–538.