La acidosis tubular renal distal (ATRD) o ATR tipo I se caracteriza por una disminución en la excreción urinaria de los hidrogeniones H+ y del amonio. En los niños afectados por ATRD hay retraso en el crecimiento, vómito, estreñimiento, falta de apetito, polidipsia y poliuria, nefrocalcinosis, debilidad y hasta parálisis muscular por la hipopotasemia. En este trabajo se resumen los avances en el estudio genético de la ATRD en las poblaciones hasta ahora estudiadas. La ATRD es heterogénea, por lo que también se analizan los transportadores y canales iónicos que se han identificado hasta ahora en las células intercaladas alfa del túbulo colector, y que podrían explicar los casos de ATRD que no se asocian con los genes hasta ahora estudiados. La ATRD puede ser autosómica dominante o autosómica recesiva. La ATRD autosómica recesiva se manifiesta en los primeros meses de vida, cursa con nefrocalcinosis y sordera temprana o tardía. La ATRD autosómica dominante es menos severa y aparece en la adolescencia o en la etapa adulta, y puede o no presentar nefrocalcinosis. En las células intercaladas alfa de los túbulos colectores se lleva a cabo la excreción urinaria de la carga ácida: los ácidos titulables (fosfatos) y el amonio. La ATRD autosómica recesiva se asocia con mutaciones en los genes ATP6V1B1, ATP6V0A4 y SLC4A1, los cuales codifican las subunidades a4 y B1 de la V-ATPasa y el intercambiador de bicarbonato/cloruro AE1, respectivamente. En contraste, la ATRD autosómica dominante se relaciona con mutaciones solo en AE1.
Distal renal tubular acidosis (dRTA) or RTA type I is characterised by reduced H+ hydrogen ions and ammonium urinary excretion. In children affected by dRTA there is stunted growth, vomiting, constipation, loss of appetite, polydipsia and polyuria, nephrocalcinosis, weakness and muscle paralysis due to hypokalaemia. This work summarises progress made in dRTA genetic studies in populations studied so far. DRTA is heterogeneous and as such, transporters and ion channels are analysed which have been identified in alpha-intercalated cells of the collecting duct, which could explain cases of dRTA not associated with the hitherto studied genes. DRTA can be autosomal dominant or autosomal recessive. Autosomal recessive dRTA appears in the first months of life and progresses with nephrocalcinosis and early or late hearing loss. Autosomal dominant dRTA is less severe and appears during adolescence or adulthood and may or may not develop nephrocalcinosis. In alpha-intercalated cells of the collecting duct, the acid load is deposited into the urine as titratable acids (phosphates) and ammonium. Autosomal recessive dRTA is associated with mutations in genes ATP6V1B1, ATP6V0A4 and SLC4A1, which encode subunits a4 and B1 of V-ATPase and the AE1 bicarbonate/chloride exchanger respectively. By contrast, autosomal dominant dRTA is only related to mutations in AE1.
In daily clinical practice, the diagnosis of a disease is usually based on the study of its clinical, biochemical, radiological and anatomical pathology manifestations. However, we depend on the development of basic research to identify and understand the cellular and molecular mechanisms involved in the aetiology of a disease.
Despite progress in the understanding of many diseases, the pathogenesis of many others has remained unknown. In many cases, although there are specific factors associated with the disease, we can not tell whether these are related to it by chance or whether they are actually a consequence of it.
Currently, genetic studies may reveal the genes involved in a disease whose origin is unknown. The importance of this strategy is that no hypothesis is necessary with regard to the pathogenesis of the disease, except the hypothesis that genetic variation contributes to the disease and that genes, which are related to the disease by chance, can be identified easily. During the past 20 years, the enormous power of this concept has been demonstrated with the identification of more than 2000 disease-related genes, which has revolutionised our perspective regarding their origin.
There are various genetic diseases in at least 10% of patients with renal failure1 and genetic factors that influence the progression of chronic damage in kidney diseases contracted.2-4 Hereditary kidney diseases have variable frequencies; for example, autosomal dominant polycystic kidney disease is the most common, affecting 1 in every 1000 people. By contrast, other hereditary kidney diseases are rare, which means that they only affect less than 5 people per 10 000.1
There is no doubt that hereditary kidney disease deteriorates the quality of life of patients. Unfortunately, our knowledge of most of these diseases is limited due to low incidence, phenotypic variability, lack of standardised diagnostic procedures and fragmentation of biological and clinical information obtained from studies with small groups. Moreover, the low prevalence of these diseases is not attracting the interest of the pharmaceutical industry and funding for research is scarce. However, the study of ‘rare’ diseases is a unique opportunity to shed light on their origin and understand the molecular scaffold complex that explains the functioning of an organ and the factors that causes deterioration.
The rapid development of exome and genome sequencing technologies opens new perspectives for the diagnosis of more than 17 000 Mendelian or monogenic diseases. Moreover, a functional study of the mutant proteins in animal models and cell models reveals the aetiology of the disease and constitutes the reference framework for drug design and/or prevention of the toxic effects of some drugs.5, 6
METABOLIC ACIDOSIS
Metabolic acidosis is characterised by a decrease in blood pH with a drop in bicarbonate concentration in plasma. In individuals with normal respiratory response, metabolic acidosis causes compensatory hyperventilation which mitigates the fall in blood pH. Consequently, blood pH homeostasis is maintained as a result of the concentration quotient bicarbonate/CO2, which is the cell and blood buffer par excellence.
The extracellular pH must be maintained within a very narrow range of 7.38 to 7.42. pH homeostasis is critical for cell function and, therefore, of our organs. A more acidic blood pH than normal may be the result of: an accumulation of acids (lactic acidosis, ketoacidosis, renal failure), loss of bicarbonate via the gastrointestinal tract (as in the case of chronic diarrhoea or malabsorption7) and bicarbonate loss due to a defect in its renal reabsorption or due to its consumption as a result of a defect in the urinary excretion of hydrogen ions by the kidney (hyperchloraemic acidosis or distal renal tubular acidosis, dRTA).8-13
The catabolism of food ingested and that of the metabolites of our own cells produces two types of acids: volatile (CO2) and non-volatile (sulphuric, phosphoric and ammonium). Protein intake produces an acid load that results in H+ ions: from 1mEq/kg/day in adults and double this amount in children.12,14 The circulating nucleic acids consume the bicarbonate present in the plasma; however, the kidney can compensate this loss, since it produces and reabsorbs the bicarbonate.
ACID-BASE BALANCE IN THE KIDNEY
The kidney maintains and controls the acid-base balance of blood through three mechanisms: filtration and reabsorption of bicarbonate, acid (or alkali) excretion and synthesis of ammonium and bicarbonate. In the kidney, two connected biochemical processes take place: bicarbonate reabsorption and the synthesis, secretion, recycling and urinary excretion of ammonium.
The presence of multiple transport systems in the different segments of the nephron tubules makes it possible to recover all the bicarbonate (HCO3-) filtered (4320mmol/day) in the glomerulus.14 In the first tubular segments of the nephron, the proximal tubules reabsorb approximately 80% of bicarbonate. In this tubular segment, bicarbonate reabsorption occurs through the Na+/HCO3-(NBCe1) cotransporter; this absorption is connected the secretion of acid in urine by the Na+/H+ (NHE3) exchanger.15 In the proximal tubules, circulating glutamine is reabsorbed from which ammonium and bicarbonate are simultaneously synthesised.
The reabsorption of 15% of the bicarbonate occurs in the thick ascending loop of Henle and only about 5% of the bicarbonate is recovered in the distal tubules of the nephron.16,17 Lastly, kidney performs the excretion of the acid load in the urine: diacid phosphate H2PO4- (titratable acid) and ammonium sulphate.
THE IMPORTANCE OF URINARY ACIDIFICATION
Urinary acidification, together with citrate excretion, is essential in the removal of organic and inorganic salts in soluble form. The urinary buffers are phosphates, but ammonium/ammonia acts as a buffer to a greater extent.
The intake of an acid load such as in a high-protein meal, causes the kidneys to produce a more acidic urine (pH<5.5); it also decreases the rate of bicarbonate excretion and increases phosphate and ammonium excretion.18-21
TRANSPORT MECHANISMS WHICH PARTICIPATE IN ACID-BASE HOMEOSTASIS IN ALPHA INTERCALATED CELLS
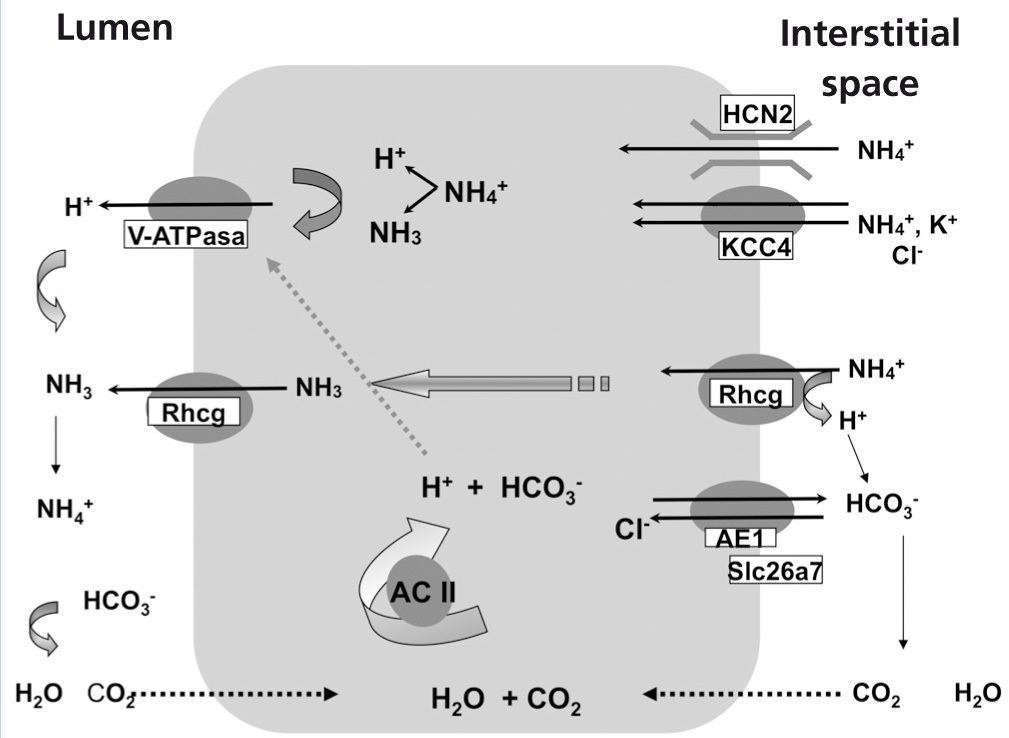
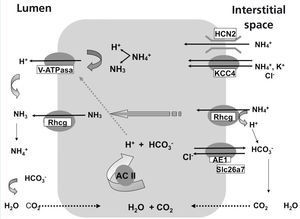
The secretion of H+ hydrogen ions in urine is carried out in the alpha-intercalated cells of cortical and medullary collecting ducts (Figure 1). H+ ATPase, V-ATPase, catalyses the passage of H+ from the cytoplasm to the tubular lumen. Anhydrase carbonic CA2 produces H+ hydrogen ions and simultaneously, bicarbonate is reabsorbed through the Cl-/HCO3- exchanger, corresponding to the AE1 isoform. The ammonium excretion mechanism takes place in two stages: firstly, there is uptake from the interstitium to the cytoplasm via HCN2 voltage-activated ammonium channels21 and Rhcg ammonia channels. 20 HCN2 channels are constitutive, they may uptake ammonium and/or sodium and are not regulated by metabolic acidosis.21 By contrast, Rhcg ammonia channels are located both in apical membranes and in basolateral membranes,22 and their destination to the membranes is regulated by metabolic acidosis.20
AE1 Cl-/HCO3- EXCHANGER
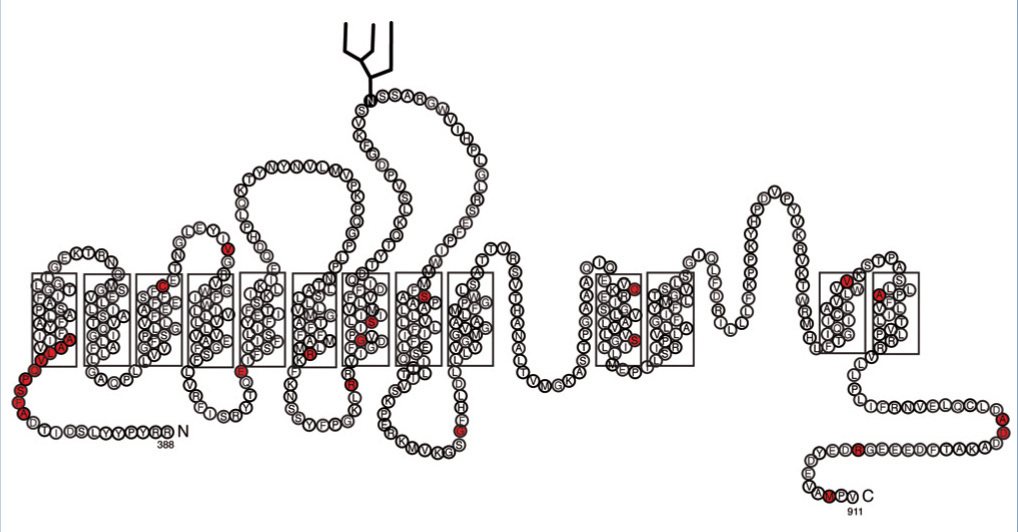
The SLC4A1 gene encodes the AE1 exchanger, a dimeric glycoprotein with 12-14 transmembrane domains23-25 (Figure 2). There are three genes in the AE1 family and in the tissues in which AE1 is expressed, AEI it participates in the regulation of pH, cell volume and the transcellular transport of acid and base in epithelial cells.26-28
AE1 presents a specific isoform of erythrocytes and a specific short isoform of the kidney.29 In erythrocytes, AE1, in addition to exchanging chloride for the bicarbonate of the plasma, has a structural role in interacting with cytoskeletal proteins that contribute to AE1 traffic and its stability in the plasma membrane.23 As such, AE1 plays a central role in respiration by transporting and removing CO2 via the lungs and in acid-base homeostasis in the kidney.30 In the kidney, AE1 performs bicarbonate reabsorption into the interstitial space and blood vessels.31 There is a group of mutations in AE1 that cause deformations in the erythrocyte and whose inheritance is autosomal dominant: inherited spherocytic anaemia, Southeast Asian ovalocytosis and other stomatocytosis with normal kidney function.32 There are other series of AE1 mutations that generate dRTA associated with erythrocyte problems.10,33,34 AE1 mutations can be consulted at: www.ensembl.org and www.hgmd.org
V-ATPase
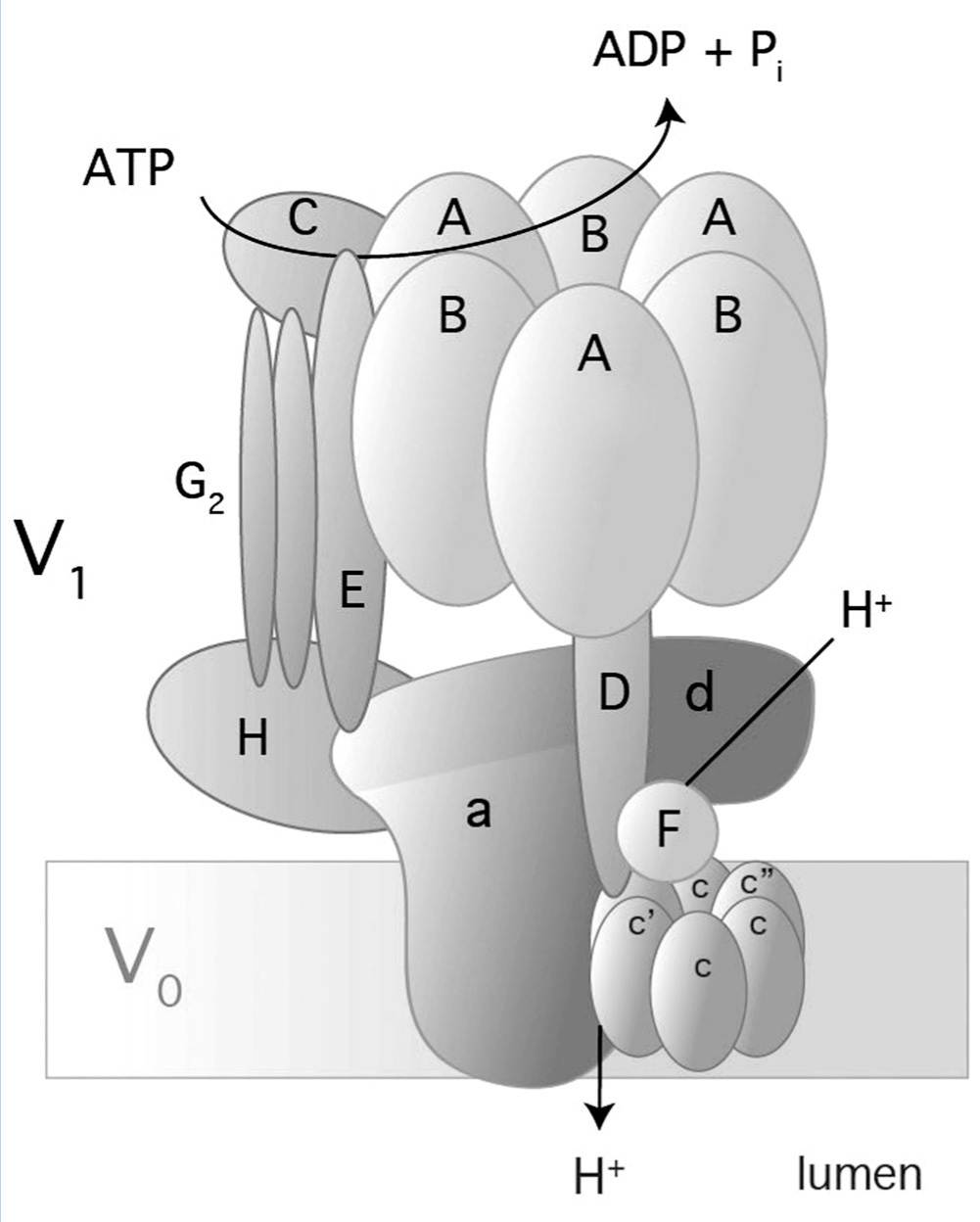
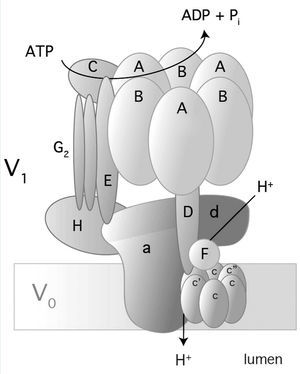
Vacuolar H- ATPase (V-ATPase) belongs to an H+ hydrogen-ion pump family and is located in a variety of membranes: endosomes, lysosomes, secretory vesicles and in the plasma membranes of eukaryotes.35-37 V-ATPase is a multimeric enzyme complex that consists of 14 subunits (Figure 3); it has two domains: one in the cytoplasm (V1) and the other in the membrane (V0). V1 is the catalytic domain and it has 8 subunits (A-H). Domain V0 comprises 6 subunits (a, c, c”, d, e, and Ac45 in mammals) and translocates H+ through the membrane.35-37 There are three copies of subunits A and B that alternate in a ring-shaped arrangement (Figure 3). The catalytic sites are in subunit A1 and the interface between subunits A-B regulates the activity of the enzyme.38-39 Subunit 'a' in V0 allows access to hemichannels through which H+ hydrogen ions are exported to the luminal space.37 There are four isoforms of subunit 'a' (a1-a4) and they have a 47-61% identity in humans.35 Subunit 'a' also participates in traffiking of V-ATPase in mammal cells.37
In alpha-intercalated cells of the collecting duct, V-ATPase is located on the apical membranes and secretes H+ in urine (Figure 1).35 Subunits B1 and a4 of V-ATPase are specific alpha-intercalated cells of the collecting duct. Defects in these subunits lead to “distal renal tubular acidosis” or dRTA.9,40 As the B1 subunit is also expressed in the ciliary cells of the inner ear,9 mutations in subunit B1 produce dRTA with deafness.
The gene ATP6V1B1 encodes B1 subunit and comprises 14 exons, which produce a protein consisting of 513 amino acids. The gene ATP6V0A4 has 24 exons of which 20 encode the 840 amino acids of a4 subunit.35
There are other transport systems in alpha-intercalated cells of the distal nephron which are also involved in acid-base homeostasis, such as carbonic anhydrase II,41 the KCC4 potassium/chloride cotransporter42, 43, Rhcg20,44 and the HCN2 ammonium channel (Figure 1).21 H-K-ATPase present in the apical membrane of alpha-intercalated cells does not seem to participate in secretion, but rather in reabsorption of K+ in hypokalemia.40.45
Figure 1 illustrates transporters, ion channels and V-ATPase in alpha-intercalated cells of the collecting duct. It is important to highlight that, traduction, and destination to the membrane of many transporters and ion channels depend on metabolic conditions.
Collecting duct microperfusion trials and knockout mouse models have helped to elucidate transport pathways involved in acid-base homeostasis in alpha-intercalated cells. For example, the mouse not expressing KCC4 develops sensorineural deafness, as well as dRTA.42 There is another Cl/bicarbonate exchanger which also operates as a Cl- channel, Slc26a7, activated by hypertonicity.46 Mouse Slc26a7 -/- develops dRTA.47 It is noteworthy that mice that do not express the ammonia channel (Rhcg -/-) have problems in excreting only in metabolic acidosis, suchas in incomplete dRTA.44 The ammonium channel HCN2 is a constitutive ion channel involved in baseline ammonium excretion but it does not appear to be regulated by metabolic acidosis. 21
DISTAL RENAL TUBULAR ACIDOSIS
dRTA belongs to the group of renal diseases with a very low incidence in any population.
In dRTA, the ability to acidify urine is lost due to a defect in the excretion of acid load (H+ and ammonium ions) in alpha-intercalated cells of the collecting duct. The acid load accumulation in the distal nephron results in consumption and reduction of the bicarbonate/CO2 buffer in blood. The symptoms accompanying dRTA include stunted growth, vomiting, diarrhoea and/or constipation, loss of appetite, polydipsia and polyuria, nephrocalcinosis and it may also present weakness and muscle paralysis due to loss of potassium (hypokalemia).
To diagnose dRTA in the clinic, it is necessary to determine plasma creatinine and fractional sodium, potassium and chloride excretion, calciuria and citraturia. Acidosis is generally observed in blood (pH<7.35) as well as a marked decrease in the concentration of bicarbonate and CO2 (<15mEq/l). In dRTA, urine pH is higher than 6 in the presence of systemic metabolic acidosis.
For cases in which diagnosis is uncertain, as in incomplete dRTA, it is advisable to perform acidification tests. These tests involve the administration of NH4Cl to determine pH, titratable acidity and urinary ammonium excretion.48 Due to complications of this test in children, acidification capacity can be evaluated by determining the maximum urinary pCO2 (UpCO2) with the intake of sodium bicarbonate (4mEq/kg).49 The pCO2 urinary test can be performed with sodium bicarbonate or acetazolamide stimuli or both, in this case administered at half the usual dose. Another test is furosemide with fludrocortisone.49-52
Diagnostic tests confirm the inability to excrete acid loads by observing a urinary pH higher than 5.5.
Ultrasound studies in patients with dRTA show the presence of calcium deposits in the renal tissue (nephrocalcinosis) and/or urinary tract stones (nephrolithiasis).
Chronic acidosis and intercurrent secondary problems (vomiting, polyuria, dehydration, rejection of dose, etc..) affect growth and, consequently, there is a decrease in the size and weight of the patients.
dRTA is accompanied by hyperchloraemia as a result of decreased HCO3- in blood. In dRTA, hypokalemia is observed ([K]<3.5mEq/l), along with hypercalciuria and hypocitraturia. Hypercalciuria occurs when there is urinary calcium excretion greater than 4mg/kg/day in both adults and in children. It is necessary to consider that the urinary calcium/creatinine quotient in infants varies with age. Normal values according to age are: 0-6 months <0.8 mg/mg, 6 to 12 months <0.6 mg/mg, 1 to 2 years <0.5 mg/mg.53 In adults, hypocitraturia is considered a value below 300 mg/day for both sexes, and/or a citrate/creatinine rate value below 250 mg/g. In children hypocitraturia is considered a value below 8 mg/kg/day and/or a citrate/creatinine quotient below 400 mg/g.53
It is important to highlight that calcium excretion in urine is high in infants and decreases progressively with age. As such, hypocitraturia is most relevant in the development of nephrocalcinosis and urolithiasis, in which primarily calcium phosphate salts are deposited. It is also noteworthy that dRTA cases have been found without no hipercalciuria.54
Patients with dRTA display a positive urine anion gap and a normal plasma anion gap ([Na+] + [K+] - [Cl]), distinguishing it from other types of acidosis, such as ketoacidosis , lactic acidosis and acidosis due to poisoning with solvents or drugs, with a higher plasma anion gap than normal.7
Untreated, dRTA causes stunted growth, rickets in children and osteomalacia in adults, and deterioration of renal function over the years.
Fortunately, dRTA has good prognosis if it is diagnosed at an early age and alkaline treatment is continued, which consists of periodic doses of sodium bicarbonate and/or potassium citrate during the day.
HEREDITARY FORMS OF RENAL TUBULAR ACIDOSIS
Mutations in genes produce varying effects: premature stop codons, shift of the open reading frame, alternate RNA processing and changes in the chemical nature of more than one amino acid. Mutations cause structural alterations in proteins which may lead to loss of function.
In autosomal dominant dRTA, one parent suffers and is the carrier of the disease. The groups of Michael Tanner of the University of Bristol and Fiona Karet of the Medical Research Institute of Cambridge in the United Kingdom were the first to identify mutations in the SLC4A1 gene in families with autosomal dominant dRTA.55,56 AE1 mutations in autosomal dominant dRTA are always heterozygous. The loss of AE1 function in the erythrocyte causes hereditary spherocytosis and ovalocytosis.55
The AE1 kidney-specific isoform is shorter: it does not have the first 65 amino acids of the NH2-terminus of the AE1 erythrocyte. As such, autosomal dominant dRTA only affects renal function in patients when AE1 mutations are located in the transmembrane domain and in the carboxyl-terminus (Figure 2).23,25,32,57-59
Autosomal dominant dRTA appears in later childhood or in adulthood.
AUTOSOMAL RECESSIVE DISTAL RENAL TUBULAR ACIDOSIS IS HETEROGENEOUS
In autosomal recessive dRTA, parents do not suffer from the disease. Symptoms occur during the first months of life. Autosomal recessive dRTA is associated with mutations in any of the following genes: SLC4A160, ATP6V0A4 and ATP6V1B1.10,54,61
Fiona Karet’s group pioneered lineage studies in families with autosomal recessive dRTA.9 The families that were analysed were of Turkish origin, mostly from consanguineous marriages. The age of children with autosomal recessive dRTA who participated in the study ranged from 1 month to 3 years; all had nephrocalcinosis and hypercalciuria, and more than 50% displayed sensorineural deafness. So far, around 20 mutations of the gene ATP6V1B1 have been identified.9,54,61,62 Most mutations are homozygous and exceptionally, compound heterozygous. These findings also revealed that the H-K-ATPase enzyme, present in the alpha-intercalated cells, can not compensate for the lack of function of V-ATPase.9
There was an interesting case of dizygotic twins with deafness, only one of whom developed autosomal recessive dRTA.63
dRTA in which deafness appears from the second decade of life is associated with mutations in the gene ATP6V0A440,61,64. So far, more than 20 mutations in ATP6V0A4 are known (www.ensembl.org and www.hgmd.org).
Approximately 20% of cases with dRTA are not associated with mutations in any of these genes: there are dRTA patients with deafness who have no ATP6V1B1 gene mutations and others with normal hearing who do not have ATP6V0A4 gene mutations. These findings suggest that other transporters or channels (Figure 1) would be mutated. Therefore, recessive dRTA is heterogeneous because it may occur by mutations in more than one gene.
Mutations have been identified in genes ATP6V1B1 and ATP6V0A4 in groups of Arab10,61 and Italian65 origin; and only mutations in ATP6V1B1 have been discovered in those of Spanish54, Greek66, Iranian67 and Serbian68 origin.
Mutations in the SLC4A1 gene, which encodes the AE1 Cl-/HCO3- exchanger, also produce autosomal recessive dRTA. To date, 11 mutations of SLC4A1 are known that produce dRTA, in addition to ovalocytosis or spherocytosis13 (Figure 3). AE1 mutations have been found mostly in the Asian population. dRTA cases in Asia are an example of natural selection, since they are resistant to malaria.
In conclusion, genetic studies have contributed to the identification of three genes affected in dRTA. Genetic studies of new molecular markers, such as Slc26a7 and KCC4 transporters or Rhcg and HCN2 channels in patients with dRTA are a challenge in the identification of new molecular targets that help the understanding of the disease and, consequently to the acid-base homeostasis in the kidney.
Conflicts of interest
The authors declare that they have no conflicts of interest related to the contents of this article.
Laura Escobar receives an allowance and funding from the Dirección General de Asuntos del Personal Académico (DGAPA) de la Universidad Nacional Autónoma de México and the Fundación Carolina.
Figure 2. Topology of the erythrocyte AE1 Cl-/HCO3- exchanger
Figure 3. Structure and composition of human V-ATPase
Figure 1. Model of an acid-secreting alpha-intercalated cell