Background: Patients with renal graft dysfunction constitute an increasingly prevalent group of end-stage kidney disease (ESKD) patients that require dialysis therapy. These patients have special characteristics that set them apart from the ESKD general population. The aim of this study was to analyse the clinical condition and evolution of patients entering dialysis with a failed kidney graft at the time of restarting dialysis and over a year of therapy according to the K/DOQI guidelines, and to compare them with incidental patients with end-stage kidney disease. We also investigated whether the modality of kidney replacement therapy may determine the clinical improvement of transplant patients. Material and Method: This is a retrospective observational study of 106 patients with ESKD followed up in the Ramon y Cajal Hospital. They were classified in two groups. Group one was made up of 50 failed native kidney patients who started dialysis between 2000 and 2009. Group two was comprised of 56 transplant patients with graft dysfunction who returned to dialysis between 1997 and 2009. We studied parameters of kidney function, anaemia, calcium-phosphorus metabolism, cardiovascular risk factors and nutritional status at the time both groups started on dialysis and one year later. Results: Both groups had a similar clinical status at the time they started on dialysis in most of the parameters analysed with the exception of anaemia. This was more severe in transplant patients, despite the fact that transplant patients received a higher dose of erythropoietin than non-transplant patients. One year later the main difference between both groups was the residual kidney function rate, higher in non-transplant patients. There were no significant differences in the parameters analysed in patients with a failed graft according to the modality of kidney replacement therapy. Conclusion: Failed transplant patients start dialysis with more severe anaemia than patients entering dialysis for the first time. Twelve months later both groups present a similar clinical condition with the exception of residual kidney function, higher in failed native kidney patients. The method of dialysis treatment after kidney transplant failure did not have a bearing on the clinical improvement of our patients.
Introducción: Los pacientes trasplantados renales con disfunción crónica del injerto que precisan reinicio de diálisis constituyen un grupo cada día más prevalente, con características especiales que los diferencia de la población general con enfermedad renal crónica (ERC). El objetivo del presente estudio fue analizar y comparar la situación clínica al inicio de diálisis y su evolución en el primer año en dos grupos de enfermos, trasplantados y no trasplantados, de acuerdo con los criterios fijados en las guías K/DOQI. Asimismo, se estudia si la modalidad de terapia renal sustitutiva (TRS) escogida a su retorno a diálisis pudiera condicionar una mejoría clínica de los pacientes trasplantados. Material y métodos: Estudio retrospectivo y observacional en 106 enfermos con ERC estadio 5 seguidos en el Hospital Ramón y Cajal. Dichos enfermos fueron divididos en dos grupos. El primero estaba formado por 50 enfermos con ERC de riñones nativos que comenzaron tratamiento con diálisis entre los años 2000 y 2009. El segundo grupo estaba constituido por 56 enfermos trasplantados con disfunción del injerto que precisaron retorno a diálisis entre los años 1997 y 2009. Se recogieron parámetros de anemia, función renal, metabolismo calcio-fósforo, factores de riesgo cardiovascular y estado nutricional en el momento de comenzar tratamiento con diálisis y un año después. Resultados: Al inicio de diálisis ambos grupos presentan valores similares en los parámetros analizados con excepción de la anemia, mayor en los pacientes trasplantados, que además recibían de manera significativa una mayor dosis de darbepoetina. Un año después las diferencias se agrupan en la función renal residual, mayor en los pacientes no trasplantados, siendo los demás parámetros estudiados similares en ambos grupos. No se encontraron diferencias en cuanto a la modalidad de TRS elegida a su retorno a diálisis en los pacientes trasplantados. Conclusión: Los pacientes trasplantados presentan en el momento de reiniciar diálisis una anemia más grave que los pacientes no trasplantados. Doce meses después ambos grupos muestran una situación clínica similar salvo en la función renal residual, mayor en los enfermos con ERC nativa. La modalidad de diálisis tras la pérdida de un injerto no determina una mejoría clínica de nuestros pacientes.
INTRODUCTION
Within the group of patients who need to start kidney replacement therapy there is a subgroup of increasing significance: patients with kidney graft failure. This subgroup has special characteristics that distinguish it from the general population with native kidney failure, such as: follow-up during the transplant period by a specialist in nephrology and chronic immunosuppressive treatment. As they are followed up by very experienced doctors in managing and treating kidney disease, they would be expected to receive the best treatment. Nonetheless, some authors have stated that patients with kidney graft failure start dialysis in a worse clinical condition than those with end-stage chronic native kidney disease.1,2 Immunosuppressive treatment increases the risk of infection, cardiovascular diseases and neoplasms. Therefore, transplant patients could be more likely to suffer from these complications. Furthermore, if the failed kidney graft remains in the body, this may cause a persistent, chronic inflammatory state that can increase patients’ morbidity and mortality.
The aim of this study was to analyse and compare the clinical situation at the start of dialysis and the evolution of the patient during the first year in two groups: transplant patients and non-transplant patients, according to the criteria of the K/DOQI guidelines. We also studied whether the chosen KRT can have a bearing on the clinical evolution of these patients on return to dialysis.
MATERIAL AND METHOD
This retrospective study included 106 patients with stage 5 CKD that started dialysis in the Ramón y Cajal Hospital. The patients were divided into two groups. The first group was comprised of 50 patients with CKD in native kidneys who started dialysis between 2000 and 2009, 35 men and 15 women with a mean age of 56.9±17.4 years (Group 1). The kidney disease was caused by glomerulonephritis in 15 cases, diabetic nephropathy in 8 cases, nephroangiosclerosis in 10 cases, interstitial nephropathy in 5 cases, undetermined in 8 cases, polycystic disease in 2 cases and other hereditary nephropathies in 2 cases. The second subgroup was comprised of 56 patients with kidney graft failure who returned to dialysis between 1997 and 2009,
37 men and 19 women, with a mean age of 51.4±13.4 years (Group 2). The graft had been functioning correctly for at least one year in all of them. Two transplant patients died during the first year due to cardiovascular problems in one case and infection causes in the other. The underlying kidney disease of the patients of this group were glomerulonephritis in 11 cases, diabetic nephropathy in 4 cases, nephroangiosclerosis in 7 cases, interstitial nephropathy in 15 cases, undetermined in 10 cases, polycystic disease in 4 cases, hereditary nephropathies in 2 cases and other nephropathies in 3 cases. In this second group, 38 patients chose haemodialysis (HD) and 18 chose peritoneal dialysis (PD). Seven patients of the PD group had to start HD as no PD catheters were available. These patients were transferred from HD to PD during the first 3 months.
Parameters for kidney function were collected from all patients on starting dialysis and after one year (creatinine, urea, and creatinine clearance according to the Cockcroft-Gault formula), as well as for anaemia (haemoglobin, haematocrit, ferritin, and treatment with erythropoietin), calcium-phosphorus metabolism (calcium, phosphorus, iPTH, chronic treatment with calcium, chronic treatment with vitamin D), cardiovascular risk factors (systolic blood pressure [SBP], diastolic blood pressure [DBP], cholesterol and triglycerides), and nutritional status (weight, total proteins, and albumin).
Statistical Analysis
The results were expressed as mean±standard deviation for numerical variables, and as absolute and relative frequencies for the categorical variables. The comparison between the two groups was carried out using Student’s t-test for numerical variables, the chi-square test for categorical variables and the Mann-Whitney test was used where necessary.
RESULTS
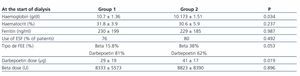
At the start of dialysis (Tables 1 and 2)
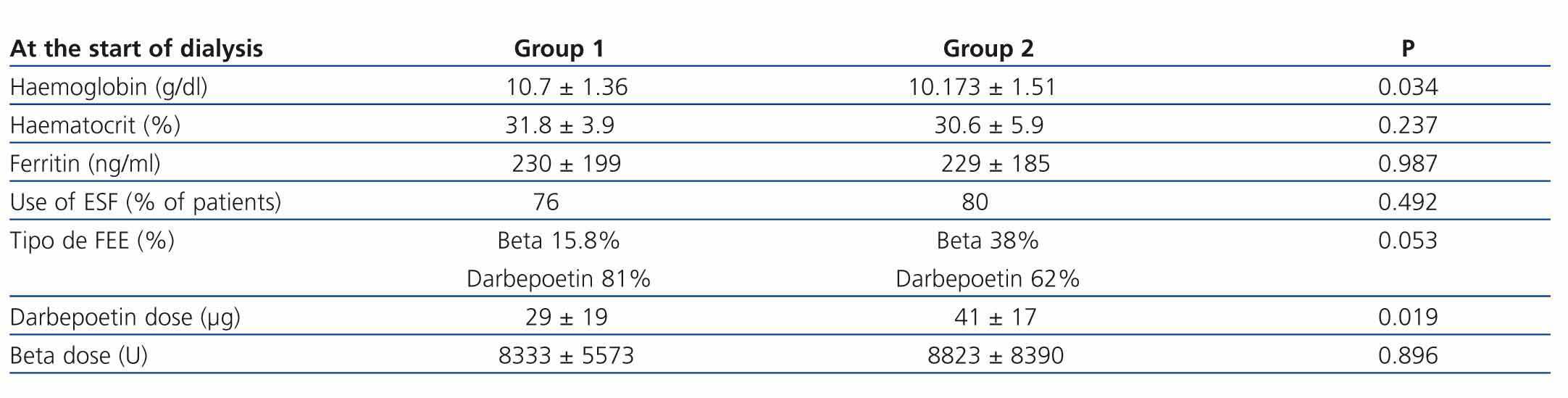
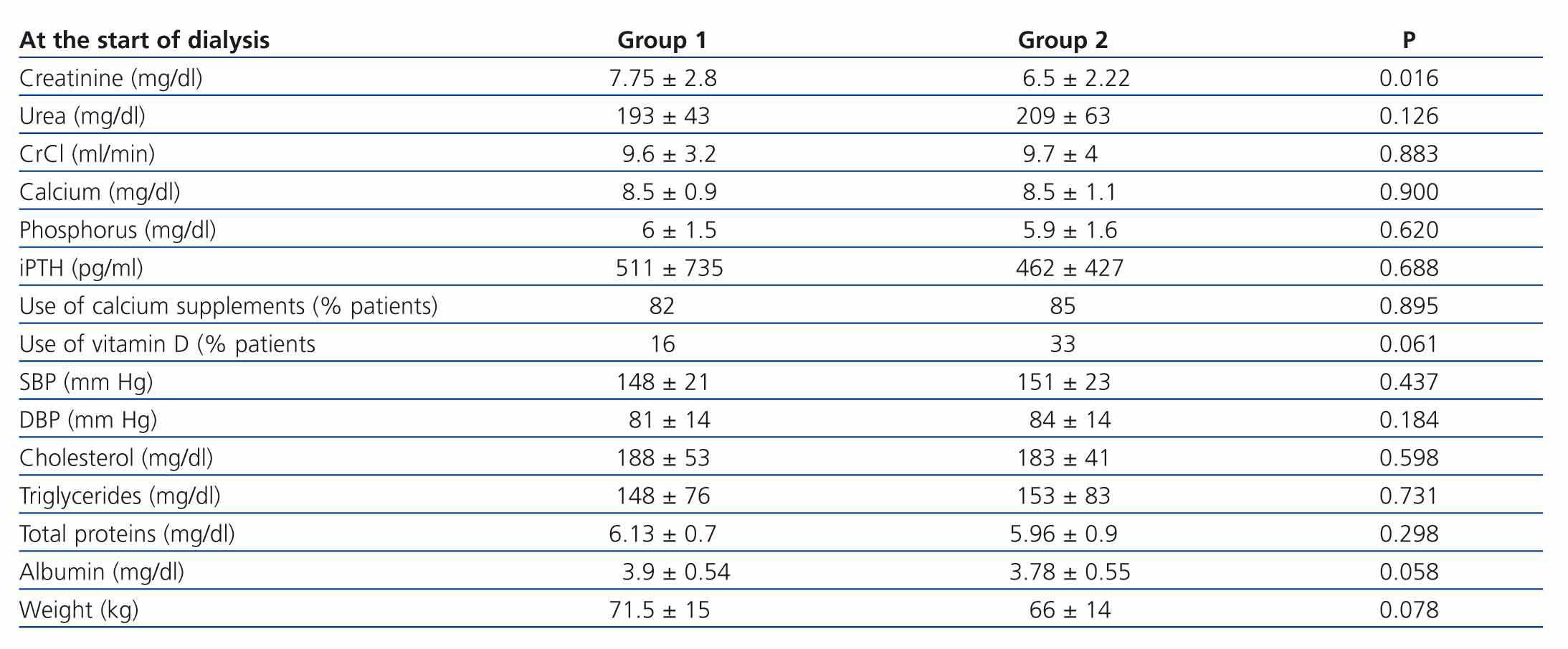
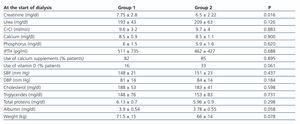
No differences were found in haematocrit and ferritin levels, or the percentage of patients treated with erythropoiesis-stimulating agents. Nevertheless, transplant patients treated with darbepoetin received significantly higher doses than non-transplant patients. Both groups had a similar kidney function, which was assessed based on urea levels and estimated creatinine clearance according to the Cockcroft-Gault equation, although creatinine levels were higher in the group of non-transplant patients. Haemoglobin was significantly lower in transplant patients. The serum calcium, phosphorus, and iPTH levels and the percentage of patients with calcium supplements were similar in both groups. A higher percentage of transplant patients received vitamin D supplements, but the difference was not statistically significant. With regard to cardiovascular risk factors, SBP and DBP values, and the total serum cholesterol and triglyceride levels were similar in both groups. With regard to the parameters concerning the patients’ nutritional status, no significant differences were found in weight, total proteins or albumin between the two groups.
After one Year on Dialysis (Table 3)
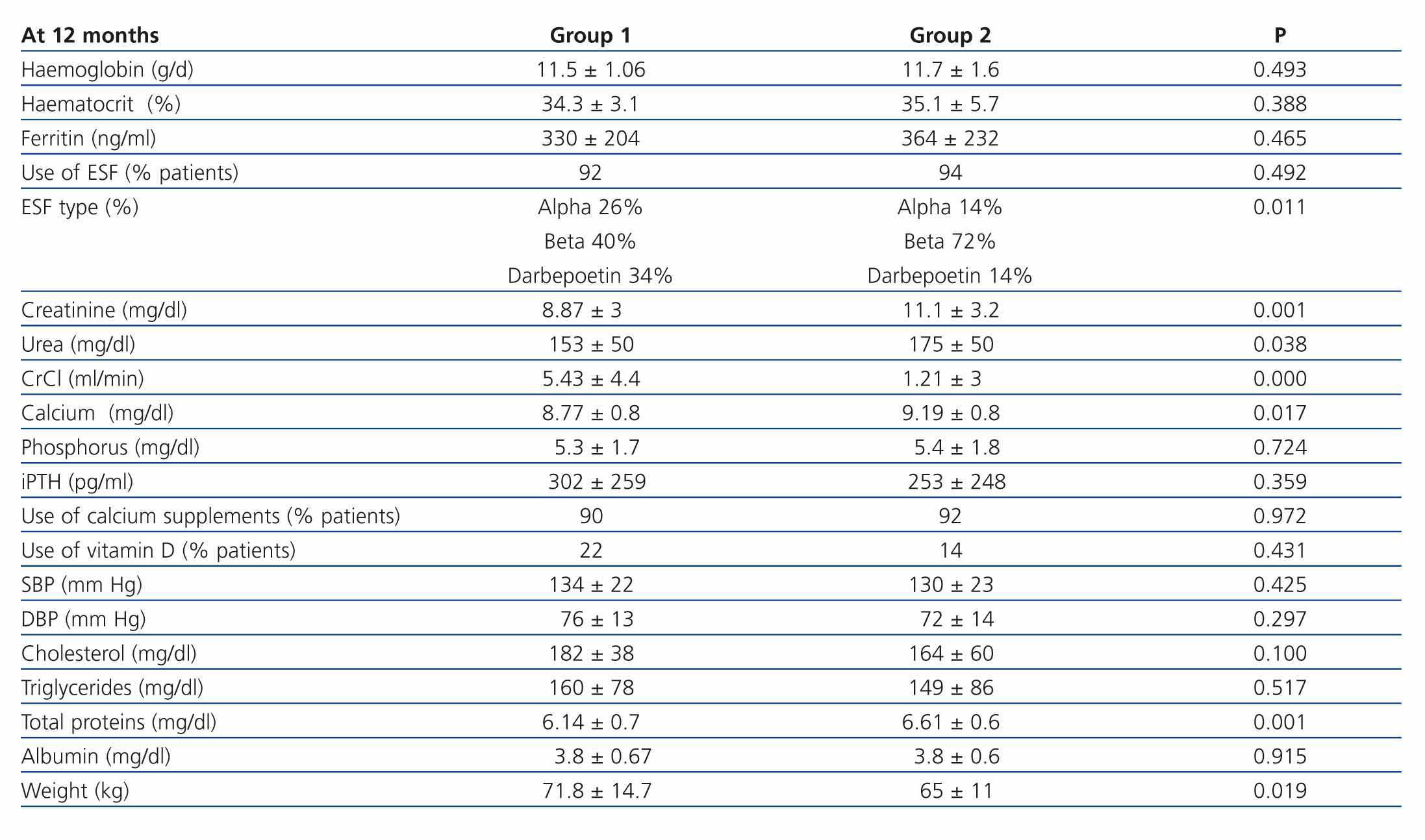
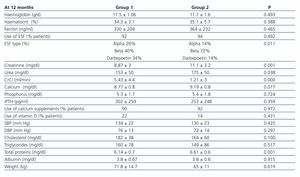
When the two groups were compared one year after starting dialysis, differences were found only in kidney function. Transplant patients had a significantly lower residual kidney function, measured by creatinine clearance, and higher urea and creatinine levels. The rest of the parameters analysed after a year were similar in both groups, except for a lower weight in transplant patients. Both groups had improved compliance with the recommendations of the K-DOQI guidelines.
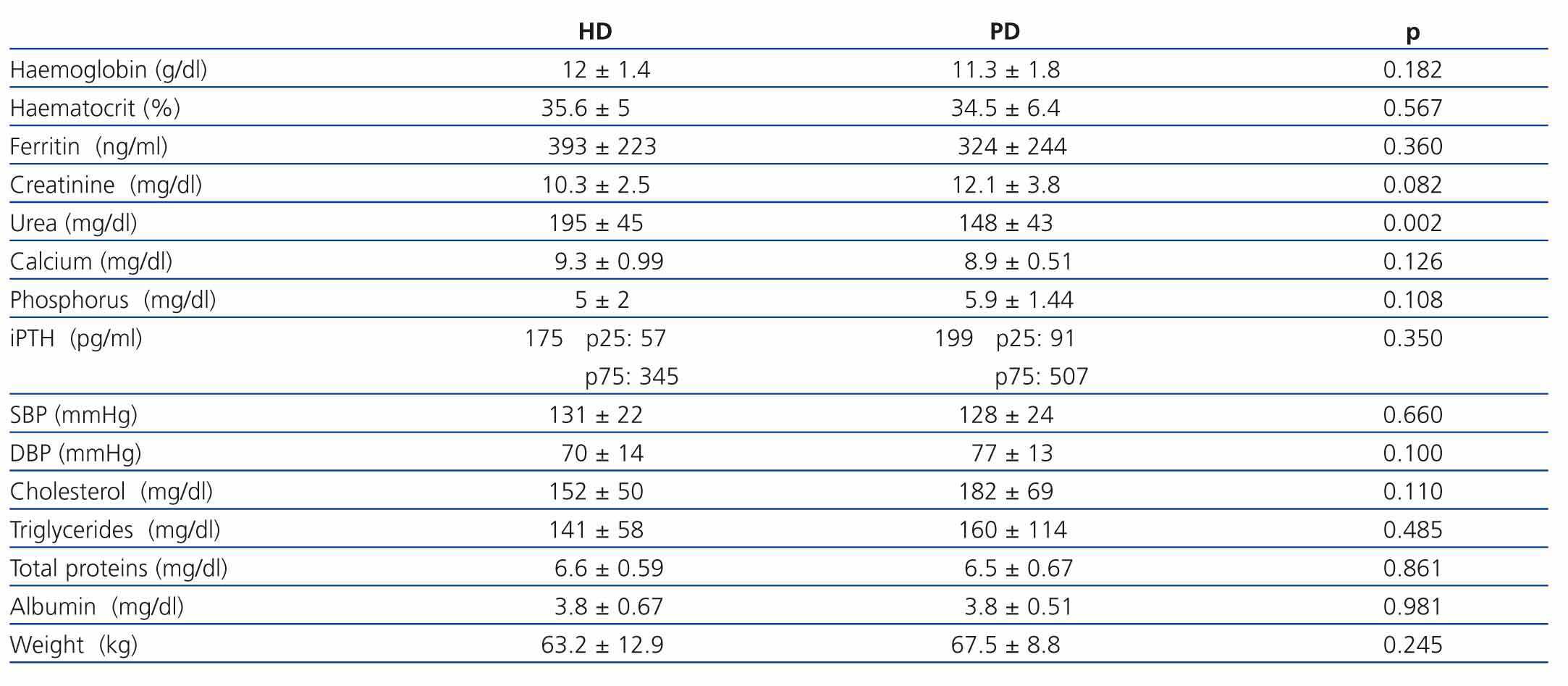
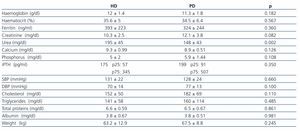
After analysing the clinical situation of the transplant patients according to the KRT method chosen, we only found significant differences in urea levels. These were higher in the patients that opted for haemodialysis. The rest of the parameters: anaemia, calcium-phosphorus metabolism, cardiovascular risk factors and nutritional state were similar in both groups (Table 4).
DISCUSSION
In spite of improved survival of kidney grafts thanks to new immunosuppressive therapies, graft dysfunction and the need to restart dialysis is a reality and there is no clear consensus on how it should be approached. This study, which analysed the situation of kidney transplant patients who needed to restart KRT, showed that these patients and the non-transplant population were in a similar situation except for anaemia. These results coincide with Gill et al.1’s study on 4643 transplant patients with
originals
chronic graft dysfunction who started dialysis compared with 233 patients with stage 5 CKD in native kidneys. They found that the transplant patients had a clinical situation which was a long way from the recommendations of the guidelines and similar to the non-transplant population. They concluded that a sub-optimal care of this type of patient could explain the results.1 However, other authors have reported different results; in this way, Arias et al. analysed levels of creatinine, urea, creatinine clearance, urea clearance, weekly Kt/V at the start of dialysis and during the previous year in 192 patients (70 transplant patients with chronic graft nephropathy who needed to restart dialysis and 122 that were starting dialysis for the first time). They observed that transplant patients starting dialysis had a worse clinical situation, lower kidney function and more severe anaemia than non-transplant patients. Furthermore, these values were a long way from the K-DOQI guidelines. These authors suggested that this situation could be due to the fact that the patients were not referred to a dialysis unit in time.2
Furthermore, the start of KRT in patients with kidney disease is determined by the clinical situation and analytical parameters, especially creatinine clearance.3 The American and European guidelines are general guidelines for patients with stage 5 chronic kidney disease.4,5 They do not set out clear recommendations on when patients with kidney graft failure have to restart dialysis and there is no global consensus on its use in these patients. We currently have the recommendations of the Spanish Society of Nephrology (S.E.N.) on the transplant patient’s return to dialyses, which were recently drawn up.6,7 These recommendations state that dialysis should be started when the glomerular filtration rate is below 15ml/min1.73m2, but there is no evidence that this improves the patient’s evolution. In our study both groups started dialysis with the same clearance rate, although transplant patients had higher urea levels and lower creatinine levels. As other studies have suggested, treatment with steroids in transplant patients probably contributes to an increased catabolism and leads to a certain level of malnutrition. This causes muscle mass to diminish and serum creatinine levels to drop. As the majority of the formulae for estimating kidney function are based on creatinine blood levels, this parameter may not be the most appropriate for deciding when to start KRT in this group of patients.8 Patients with chronic graft dysfunction would thus start dialysis later and with greater complications caused by uraemia.
It must be highlighted that transplant patients lost residual kidney function faster. Stopping immunosuppressive treatment just after starting dialysis in order to reduce infections and associated morbidity, could explain these findings, which are consistent with those of previous studies.9,10
The anaemic situation was also analysed in our study. We observed in our series that transplant patients started KRT with more severe anaemia than patients with CKD in native kidneys, although the percentage of patients treated with erythropoiesis-stimulating factors (ESF) was similar in both groups. This difference was even greater when taking into account that the darbepoetin dose received by transplant patients was significantly higher than in the group with native CKD. These results coincide with other studies that state that the chronic inflammatory state of these patients may explain why it is harder to treat the anaemia. Furthermore, graft dysfunction and immunosuppressive treatment leads to a greater resistance to erythropoietin.11 These differences disappeared a year after starting KRT, both groups having analytical parameters that coincided with the recommendations of the K-DOQI guidelines.12
With regards to calcium-phosphorus metabolism, it has been described in medical literature that a significant reduction in PTH can be seen 3 months after a kidney transplant, as well as an increase in calcaemia and a decrease in phosphataemia, and sometimes transient hypophosphataemia.13 Later studies have confirmed that the main factor for the rise in PTH and the secondary hyperparathyroidism must have been the progressive decrease in the glomerular filtration rate.14 In our study we were able to observe the presence of secondary hyperparathyroidism in the two groups before starting dialysis, with parameters a long way from those recommended by the K/DOQI guidelines.5 Both groups had a greater control of bone metabolism one year after KRT. No significant differences were found between the two groups.
As regards cardiovascular risk, both groups had blood pressure rates higher than those recommended by the guidelines, with cholesterol and triglyceride levels on the upper limit when starting dialysis. Many studies state that cardiovascular disease is the main cause of death in transplant patients and non-transplant patients with kidney failure. It has a much higher incidence in this group than in the general population,15 meaning that kidney failure itself is a cardiovascular risk factor. The elevated incidence of traditional cardiovascular and transplant-related risk factors could contribute to the high cardiovascular morbidity and mortality of these patients, which is lower than those on the waiting list. The transplant patient’s early-onset high cardiovascular morbidity and mortality prompted recommendations to act early on these factors to prevent and reduce the incidence of cardiovascular events.16 Dialysis makes it easier to control blood pressure, such that one year after starting KRT both groups had values in accordance with the recommendations of K/DOQI guidelines.17 Nevertheless, we did not include the treatment for hypertension or hyperlipidaemia used by the patients in our study.
We found albumin levels on the lower limit in both groups when starting dialysis and after one year of treatment. It is has been shown that hypoalbuminaemia is a factor associated with increased mortality after kidney transplant. Different factors may cause this hypoalbuminaemia, such as malnutrition and chronic use of steroids.18
Although other studies analysed mortality and the number of hospitalisations instead of analytical parameters one year after restarting dialysis, all of them agree that the transplant population has a higher morbidity and mortality.19,20 Our study reached the same conclusions as previous studies, which found that the clinical situation of transplant patients on restarting dialysis was similar to the non-transplant population and that the analytical parameters were inadequate. These results are surprising, considering that both groups were followed up by doctors with large experience treating CKD. However, we must point out that it is very difficult to achieve the objectives of the K-DOQI guidelines due to the many complications that patients with end-stage kidney failure have and the many different medications that they have to take to control these complications. Furthermore, the objectives of the guidelines are mostly derived from expert opinion and optimal values are recommended without prior studies to evaluate how to difficult it is to attain them.
Lastly, the dialysis method chosen after the loss of a kidney graft is a question that still has to be answered. The few studies available (with small patient series) that have analysed the survival of patients after starting HD or PD have not found significant differences in favour of either of these two techniques.21 Davies et al. compared 45 patients with kidney graft failure, 28 patients restarted on PD and 17 on HD. The survival rates of both techniques were similar, although PD was seen to be more beneficial. They did not find any differences when comparing the population that started PD after losing the graft and the general population starting PD for the first time.10 De Jonge et al. compared 60 patients with kidney transplant failure, 21 patients chose PD and 39 HD. They found that the survival rate for both techniques was similar, although HD was found to be more beneficial.22 We did not find any differences in our series between the two KRT methods in the parameters for anaemia, kidney function, bone metabolism, cardiovascular risk factors or nutritional state. Only urea levels were higher for HD, probably due to the continuity of PD treatment and the analytical samples being taken after a much longer interdialysis period in HD. The current recommendations would a prioribe the same as those for patients starting dialysis for the first time. Providing sufficient information to the patient and choosing the method early would be essential to carry out an appropriate vascular access and start the chosen technique according to the schedule.
CONCLUSION
To summarise, transplant patients that restart KRT have a similar clinical situation to patients with CKD in native kidneys, except for anaemia, which is more severe in transplant patients. Twelve months later differences were only found in residual kidney function, which was higher in patients with native CKD.
The dialysis method after graft failure is a question that still needs to be answered. Nevertheless, although larger studies are needed to determine the best option, there are currently no evident differences between HD and PD for this group of patients restarting KRT. Given that the results obtained in different studies are disparate, the recommendations of the guidelines could be the key to obtaining better results in this special group of patients.
Table 1. Comparing the anaemia parameters of non-transplant patients (group 1) and transplant patients starting dialysis (group 2)
Table 2. Comparing the parameters of kidney function, calcium-phosphorus metabolism, cardiovascular risk and nutrition of non-transplant patients and transplant patients starting diálisis
Table 3. Clinical situation of non-transplant patients and transplant patients 12 months after starting dialysis
Table 4. Clinical situation of non-transplant patients and transplant patients 12 months after starting dialysis according to the type of KRT chosen