Drug-induced nephrotoxicity is a frequent adverse event that can lead to acute or chronic kidney disease and increase the healthcare expenditure. It has high morbidity and mortality incidence in 40–70% of renal injuries and accounts for 66% cases of renal failure in elderly population.
ObjectiveAmelioration of drug-induced nephrotoxicity has been long soughed to improve the effectiveness of therapeutic drugs. This study was conducted to review the melatonin potential to prevent the pathogenesis of nephrotoxicity induced by important nephrotoxic drugs.
MethodsWe analyzed the relevant studies indexed in Pubmed, Medline, Scielo and Web of science to explain the molecular improvements following melatonin co-administration with special attention to oxidative stress, inflammation and apoptosis as key players of drug-induced nephrotoxicity.
ResultsA robust consensus among researchers of these studies suggested that melatonin efficiently eradicate the chain reaction of free radical production and induced the endogenous antioxidant enzymes which attenuate the lipid peroxidation of cellular membranes and subcellular oxidative stress in drug-induced nephrotoxicity. This agreement was further supported by the melatonin role in disintegration of inflammatory process through inhibition of principle pro-inflammatory or apoptotic cytokines such as TNF-α and NF-κB. These studies highlighted that alleviation of drug-induced renal toxicity is a function of melatonin potential to down regulate the cellular inflammatory and oxidative injury process and to stimulate the cellular repair or defensive mechanisms.
ConclusionThe comprehensive nephroprotection and safer profile suggests the melatonin to be a useful adjunct to improve the safety of nephrotoxic drugs.
La nefrotoxicidad inducida por medicamentos es un acontecimiento adverso frecuente que puede conducir a una nefropatía aguda o crónica, e incrementar los costes sanitarios. Presenta una incidencia elevada de morbimortalidad en el 40-70% de las lesiones renales y es responsable del 66% de los casos de insuficiencia renal entre la población de edad avanzada.
ObjetivoLa mejora de la nefrotoxicidad inducida por medicamentos es un objetivo anhelado desde hace mucho tiempo, para mejorar la eficacia de los fármacos terapéuticos. Este estudio se llevó a cabo con el propósito de revisar el potencial de la melatonina para prevenir la patogenia de la nefrotoxicidad inducida por medicamentos nefrotóxicos importantes.
MétodosAnalizamos los estudios relevantes indexados en Pubmed, Medline, Scielo y Web of Science, para explicar las mejoras moleculares posteriores a la administración concomitante de melatonina; prestando especial atención al estrés oxidativo, la inflamación y la apoptosis como actores fundamentales de la nefrotoxicidad inducida por medicamentos.
ResultadosUn sólido consenso entre los investigadores de estos estudios sugirió que la melatonina erradica de forma eficiente la reacción en cadena de producción de radicales libres e induce las enzimas antioxidantes endógenas que atenúan la peroxidación lipídica de las membranas celulares y el estrés oxidativo subcelular en la nefrotoxicidad inducida por medicamentos. Este consenso se vio respaldado por el papel de la melatonina en la desintegración del proceso inflamatorio a través de la inhibición de las principales citocinas proinflamatorias o apoptóticas, como el TNF-α y el NF-κB. Estos estudios subrayan que la mitigación de la nefrotoxicidad inducida por medicamentos se deriva del potencial de la melatonina para regular a la baja el proceso celular de lesión inflamatoria y oxidativa, y estimular la reparación celular o los mecanismos defensivos de las células.
ConclusiónLa nefroprotección exhaustiva y el perfil de seguridad más favorable sugieren que la melatonina es un complemento útil para mejorar la seguridad de los fármacos nefrotóxicos.
On the advent of modern medicine, number of drugs have been discovered to treat more advance and complex disorders. The efficacy and therapeutic potential of drugs have been exploited, in pre-clinical or clinical settings, by number of toxicities. Drug-induced nephrotoxicity has been remained one of the major complication in the treatment with the drugs. Different drugs have their own unique pattern and mechanisms to cause nephrotoxicity. Most commonly drug-induced nephrotoxicity is manifested by tubular necrosis, interstitial nephritis, crystal nephropathy, Angiopathy, alterations in intraglomerular hemodynamics, rhabdomylosis and fibrosis which may ultimately leads to renal failure.1–3 The drug-induced nephrotoxicity has been accounted for 66% renal failure cases of elderly population.4 Despite the advancements in medical research, drug-induced nephrotoxicity is continuously evolving into high morbidity and mortality in 40–70% of patient with acute renal injury.5 Moreover, it also limits the effectiveness of drugs and complicates the medical conditions in the treatment of serious malignancies, renal transplantation and several metabolic disorders.6–10
Melatonin, a pineal gland product, was first isolated in 1958 correlating its role with melanocytes.11 Melatonin (N-acetyl-5-methoxy tryptamine) was found to be present in all organisms and initially known for its physiological role in control of circadian rhythm. It was found to be biosynthesized as a metabolic end-product of tryptophan in pinealocytes with higher proportion in the night time and released easily into blood stream due to its higher lipophilic nature.12 Since its identification, melatonin has been well studied for its roles in complex physiological processes of controlling circadian rhythm to oncostatic function in cancer proliferation. Melatonin exerts its biological activity through melatonin receptors, i.e. MT1 and MT2, that are seven membranous G-Protein coupled receptors and modulate the intracellular concentrations of cyclic nucleotides (cAMP, cGMP) and their transduction pathways.13,14 However, studies have attributed the structural features of melatonin for its active role that endows it with direct free radical scavenging and augmentation of antioxidant enzymes in oxidative stress.15,16 Melatonin has been found to stimulate expression of antioxidant hemeoxygenase1 (HO-1) and anti-inflammatory cytokine (IL-10) in addition to mitigate the CD 19+ B cells concentration, pro-inflammatory cytokines and apoptotic process in idiopathic membranous nephropathy.17 Moreover, melatonin has been reported to effectively eradicate the renal oxidative stress induced by mercuric chloride, carbon tetrachloride, uranium, cadmium and chlorpryfos-ethyl.18–22 However, there are continuous efforts to investigate the therapeutic potential of melatonin in treatment of drug-induced toxicities.23–25
This review is aimed to explore the current state of the knowledge on melatonin's role and mechanisms in amelioration of drug-induced nephrotoxicity. This review will serve as concise literature to evaluate the trend and potential of melatonin as nephroprotective adjunct in drug-induced nephrotoxicity.
Nephroprotective mechanisms of melatoninMelatonin is well-reported anti-inflammatory and antioxidant agent that serves the basic mechanisms for protection against the drug-induced nephrotoxicity. In general, nephrotoxicity is mediated by stimulation of oxidative stress followed by acute or chronic inflammatory response in glomerulo-tubular cells that mediates the cellular necrosis.26 These pathways are interrelated as free radicals can also induce inflammatory cascade through enhanced mRNA expression of chemo-attractants and adhesions molecules, like ICAM-1, MCP-1 and CSF-1, which infiltrate tissue injury site with inflammatory cells.27 Moreover, free radicals also function as intermediary to induce the activation of transcription factor NF-κB.28 NF-κB translocate and bind to the DNA and up regulate the transcription of proinflammatory mediators such as iNOS, TNF-α, IFN-γ and IL-1.29 Melatonin has been suggested to restrict the translocation and binding of NF-κB to its active binding site P65 on DNA thereby hindering the subsequent inflammatory process.30–32
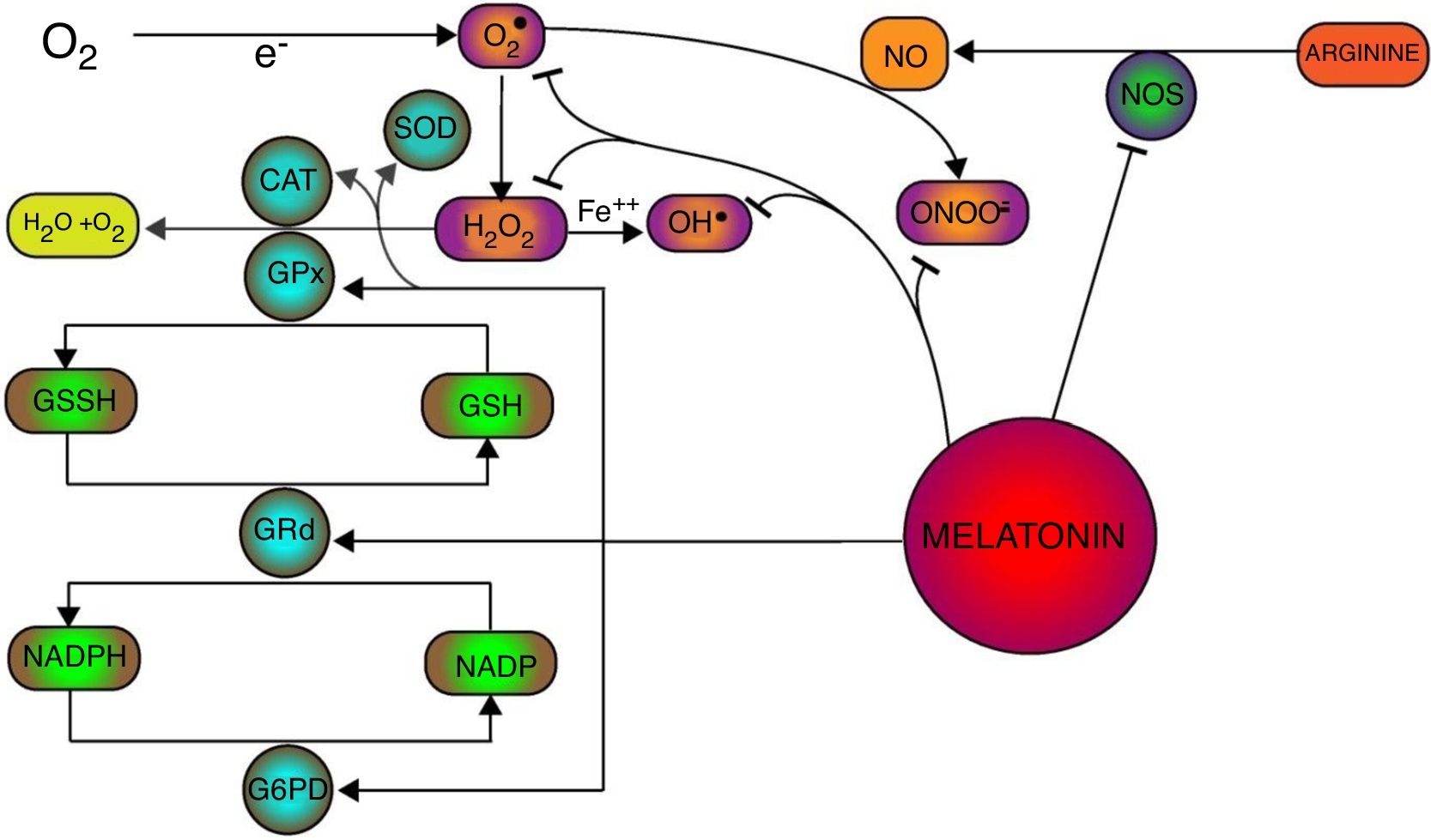
Oxidative stress has been manifested with saturation of antioxidant defense system which increases the production of free radicals and subsequent cellular damage and necrosis. Apart of its action on inflammatory process, Melatonin has been proved to be a potent scavenger of free radicals and inducer of antioxidant enzymatic machinery in oxidative stress (Fig. 1). Melatonin exerts its antioxidant action through both receptor-mediated and receptor-independent pathways.33,34 Receptor-mediated activity of melatonin is attributed to its ability to increase the stimulation and expression of variety of antioxidant enzymes. Melatonin's role in the stimulation of antioxidant enzymes was first identified where it was found to markedly increase the activity of glutathione peroxidase (GPx). It was suggested that increased glutathione peroxidase (GPx) activity is associated with the increased metabolism of H2O2 into water and oxygen instead of its conversion into potent free radical through Fenton-type reaction (i.e. OH().35–37 Primarily, GPx activity supplements the antioxidant process of superoxide dismutase (SOD) and catalase (CAT) induced by melatonin.38 However, increase activity of glutathione peroxidase (GPx) utilizes the reduced glutathione (GSH) and deposits glutathione disulphide (GSSH) as oxidized form. Glutathione disulphide (GSSH) is reduced into glutathione (GSH) through glutathione reductase (GRd), an enzyme which was also found to be stimulated by melatonin in physiological concentration, thereby recovering the glutathione (GSH) in the process of H2O2 metabolism.39,40 In this process, Glutathione reductase (GRd) uses NADPH as a co-factor which is to be recovered by the Glucose 6-phosphate dehydrogenase (G6PD). Melatonin has been reported to be involved in the induction of G6PD enzymatic activity to protect the cell from oxidative damage.41,42 Besides, melatonin also attenuates the enzymatic activity of nitric oxide synthetase (NOS) and production of NO that provides the further protection from NO mediated production of more potent oxidant peroxynitrite specie (ONOO−).43 Therefore, melatonin mediates the multifaceted stimulation of antioxidant enzymes and enhance the cellular protection against oxidative stress.
In addition to its receptor-mediated antioxidant pathways, receptor-independent antioxidant mechanisms provide lead towards melatonin's ability to neutralize free radicals which eventually provides the protection against cellular and sub-cellular oxidative damages. Direct free radical scavenging ability of melatonin is to be associated with the intrinsic capacity of melatonin's indole ring. It has been suggested that indole ring of melatonin undergoes the addition of free radical or donation of electron to neutralize the free radicals.44,45 Melatonin has been found to directly scavenge variety of free radicals such as O, OH, ONOO and H2O2.46 Overall oxidative stress, in aerobes, is initiated with the reduction of molecular or ground state oxygen into superoxide anion (O2−) free radical which further interact and transform into more toxic free radicals such as OH, ONOO and H2O2. Superoxide anion (O2−) is scavenged, naturally, by superoxide dismutase (SOD) into H2O2 which is further metabolized into the water and oxygen. Apart of the SOD stimulation, melatonin also directly scavenges the superoxide anion (O2−) thus hampers the generation of further toxic free radicals.47,48 Hydroxyl free radical (OH) is considered as most toxic and harmful electrophile due to its higher reactivity towards every cellular component which results into its insusceptibility toward antioxidant enzymes.49 Melatonin-mediated neutralization of OH radicals results into the formation of 3-hydroxymelatonin (3-OHM) metabolites.50 Nitric oxide (NO) free radical has been found to couple with O2−. free radical which result into more toxic peroxynitrite (ONOO−) free radicals. Melatonin has been reported to be the potent scavenger of peroxynitrite free radicals (ONOO−) and their interaction also result into the 3-OHM metabolites. Hydrogen peroxide (H2O2) is less toxic, as compare to OH, ONOO− and O2−, but it must be neutralized before it is converted into more potent specie, i.e.OH. Beside the amplification of GPx activity, melatonin also deactivates H2O2 free radicals in a dose-dependant manner which follows the formation of AFMK (N-acetyl-N-formyl-5-methoxykynuramine) metabolites.51 Interestingly, it was noticed that reaction products (of melatonin with free radicals), such as AFMK and 3-OHM, were also found to possess the strong free radical scavenging capacity which corroborate the cascade of free radicals scavenging by melatonin.52,53 Consequently, scavenging of all these potent free radicals, contributes into the melatonin's potential to offset the free radical mediated cellular toxicity in kidney.
Mitochondria is a key player in free radical generation consistent with impairment in electron transport chain (ETC) that, if endures, may lead to cellular redox imbalance and mitochondrial dysfunction in the pathogenesis of renal diseases.54 The amphiphilic nature of melatonin allows it to cross all morphological or physiological barriers to diffuse into the variety of sub-cellular compartments.55–57 However, melatonin has shown preferential deposition in membranes and mitochondria.58 The superior ability of melatonin to accumulate into the mitochondria makes it an attractive antioxidant to treat oxidative stress related pathologies as compare to conventional antioxidants with poor mitochondrial penetration such as co-enzyme Q10 and tocopherol.59 Conversely, melatonin has also been found to stimulate the generation of reactive oxygen species (ROS) at mitochondrial complex III (MC-III).60 Interestingly, the reduction in this pro-oxidant effect has been implicated in development of diabetic nephropathy due to impaired ETC at the MC-III level and its discordant regulation as compare to hyperactive mitochondrial complex I (MC-I).61 Overall, melatonin has been found to prevent the leakage of election or subsequent free radicals generation in mitochondrial respiration that makes it a mitochondrial stabilizer.62 Antioxidant property of melatonin counteract the membrane lipid peroxidation to stabilize the mitochondrial inner membrane potential and also protects the mitochondrial DNA (mtDNA) and ETC proteins from free radical induced structural or functional impairments to preserve the normal ATP generation or mitochondrial homeostasis.63–65 Melatonin has been found to protect the biological membranes from oxidative stress induced rigidity and preserve their optimal fluidity that may also corroborate the melatonin's ability to prevent the loss of mitochondrial inner membrane potential, opening of mitochondrial permeability transition pore (MPTP) and subsequent release of apoptotic mediators such as cytochrome C and caspases.64,66,67 Consistently, melatonin abrogates the mitochondria derived intrinsic apoptosis process by limiting the Bax activation, cytochrome C efflux and caspases expression to ameliorate the morphological damages in proximal tubules of kidneys.68 Therefore, due to its diversity in protective mechanisms, melatonin has been extensively studied and suggested for its ability to mitigate the oxidative stress and subsequent inflammatory process in nephrotoxicity of drugs discussed below.
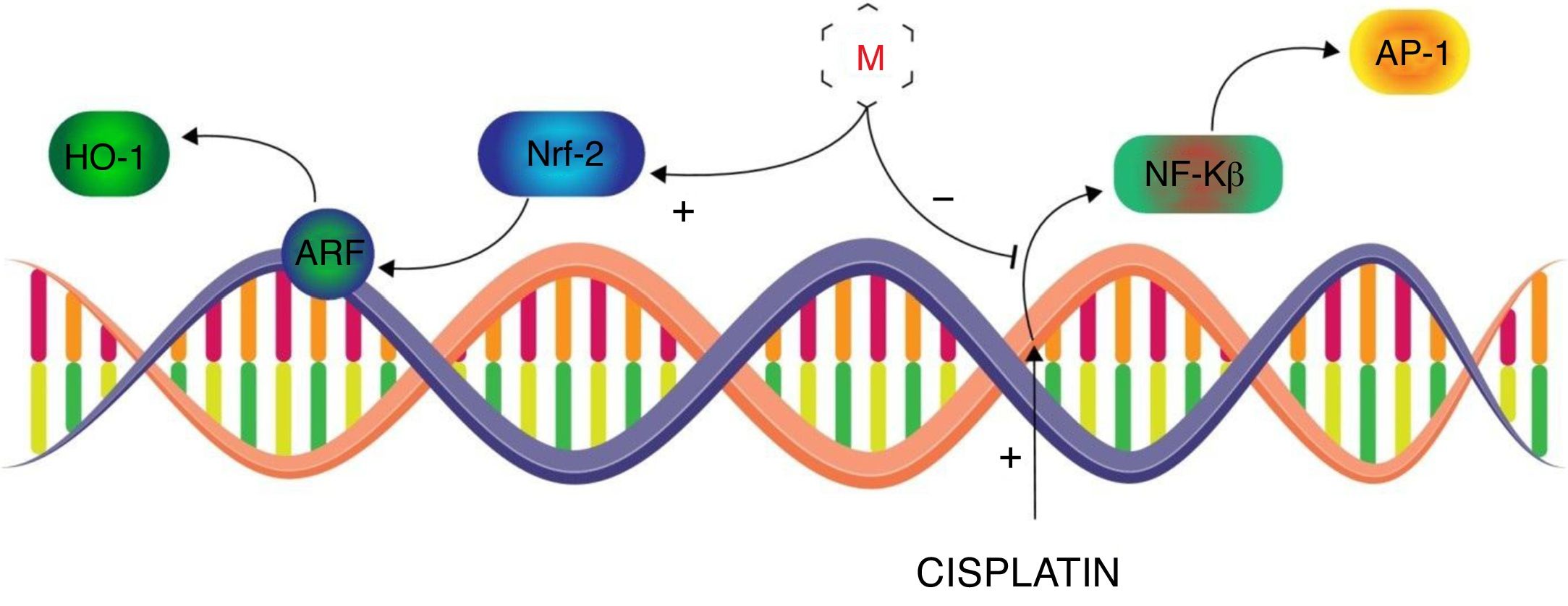
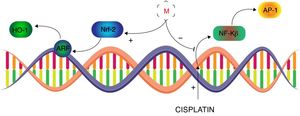
Melatonin use to mitigate the nephrotoxicity of drugsAnticancerCisplatinCisplatin, cis-diamminedichloroplatinum II, is broadly and effectively used antineoplastic agent in chemotherapy of most solid tumors. The therapeutic effectiveness, of Cisplatin, has been compromised by its dose limiting toxicities, mainly nephrotoxicity, which leads to the discontinuation of therapy.69 Despite its severe toxicities, it has been proven to be effective in variety of tumors and has not lost its use in clinical therapeutics. Cisplatin-induced nephrotoxicity is majorly associated with its metabolites to cause intracellular oxidative stress and subsequent vigorous inflammatory pathways in proximal tubular cells.70 Free radicals are believed to induce the intracellular signaling, of NF-κB and TNF-α, through P38 MAPK pathway and formation of DNA adducts which ultimately leads to pro-inflammatory cytokines release and apoptosis.71,72 In this regard, Sener et al. performed an animal study to evaluate whether melatonin can attenuate the cisplatin-induced nephrotoxicity.73 It was observed that cisplatin significantly depletes the renal GSH content while enhanced the malondialdehyde (MDA – lipid peroxidation), creatinine level, blood urea nitrogen (BUN) suggesting renal injury. The histological examination of kidney revealed the glomeruli shrinkage, vacuolization in proximal and distal tubular cells, disrupted microvili and leukocytes infiltration in parenchyma. Melatonin co-administration presented comparatively moderate cortical damage and leucocytes infiltration. However, with melatonin pre- and post-treatment in cisplatin administration, histological appearance was overall normal with fewer vacuolization and minor degeneration at the proximal and tubular cells. The biochemical profile of this group revealed the significant improvement in GSH levels and marked reduction in the MDA, creatinine and BUN as compare to cisplatin-only treated group. In this way Sener et al. documented the melatonin potential, which was more prominent when it was given before and after cisplatin, to ameliorate cisplatin induced nephrotoxicity. It is noteworthy that severity of cisplatin-induced nephrotoxicity is influenced by the physiological concentration of circulatory melatonin that in lower levels (e.g. pineal insufficiency or aging) may result into the more pronounced cisplatin-induced acute tubular necrosis (ATN). However, pre-treatment with exogenous melatonin has been found to significantly alleviate the cisplatin-induced nephrotoxicity even at suboptimal physiological concentration of circulatory melatonin.74 Hara et al. further investigated the comparative nephroprotective potential of melatonin and its metabolite, 6-hydroxymelatonin (6-OHM), that suggested the superior capacity of melatonin to recover the GSH/GSSH ratio and induction of GPx in cisplatin-induced nephrotoxicity.75 Electron spin resonance study revealed that OH. Free radical to be the main offender in cisplatin-induced oxidative insult and melatonin significantly suppressed the formation of OH adducts as compare to 6-OHM, mannitol and glutathione.76 Furthermore, in vivo study elucidated the more detailed underlying nephroprotective mechanism of melatonin against cisplatin. Melatonin was found to enhance the nuclear concentrations of activated nuclear factor-E2-related factor (Nrf2) which amplified the expression of antioxidant enzymes, i.e. heme oxygenase (HO-1) through nuclear activation of antioxidant response element (ARE) (Fig. 2). It was suggested that melatonin ameliorates the cisplatin-induced nephrotoxicity by inhibiting the nuclear-factor κB (NF-κB) and activator proteins (AP-1) along with augmentation of Nrf2/HO-1 signaling in the kidneys of cisplatin treated rats.77 Therefore, melatonin tends to improve the nephroprotective pathways which serves to counterbalance the cisplatin dose-related nephrotoxicity and extends the therapeutic index of cisplatin.
Transcriptional control of Melatonin (M) to alleviate cisplatin-induced nephrotoxicity. Melatonin inhibits the cisplatin-induced production of NF-κB and AP-1 while induces the expression of Nrf-2 to mediate the ARF derived activation of HO-1 in alleviation of cisplatin-induced nephrotoxicity.
Ifosfamide is an structural analogue of cyclophosphamide that is widely used antineoplastic agent in various malignancies such as breast cancer, non-small cell lung cancer, soft tissue sarcomas but its effectiveness has been compromised by its nephrotoxicity.78–81 It is a prodrug that is metabolized into active but toxic metabolite chloracetaldehyde (CAA) in order to produce its pharmacological antitumor effect.82 Ifosfamide conversion into nephrotoxic chloracetaldehyde (CAA), mainly in renal proximal tubules, results into renal dysfunction that is associated with oxidative stress and impaired tubular transporters functions which is most commonly presented as Fanconi's syndrome.83–85 Sener et al. investigated putative role of melatonin in alleviation of ifosfamide induced nephrotoxicity in an animal study that involved the biochemical and histological considerations.86 Ifosfamide 50mg/kg for 5 days induced the GSH depletion and severe lipid peroxidation, myeloperoxidase activity, fibrosis which implied the marked oxidative stress and neutrophil infiltration. Moreover, ifosfamide was also found to severely impaired the tubular reabsorption by diminishing the activity of Na+-K+ ATPase which results into defective and enhanced urinary excretion of glucose, calcium, phosphate and plasma proteins, i.e. Fanconi's syndrome. Increase in oxidative stress and inflammatory process resulted in renal morphological degeneration such as vascular congestion, glomerular degeneration and tubular interstitial oedema which were alleviated with melatonin (10mg/kg) co-administration. Melatonin co-administration reversed the GSH depletion and significantly reduced MDA, myeloperoxidation, abnormal collagen accumulation thereby attenuating oxidative stress, inflammation and tissue fibrosis. Apart of this, melatonin concomitant administration was also shown to restore the Na+-K+ ATPase activity which improved the tubular re-absorption process and subsequently attenuated the Fanconi's syndrome. These finding suggested that melatonin can be effectively use as nephroprotective adjunct to ameliorate the oxidative stress, inflammatory process and renal metabolic impairments in ifosfamide nephrotoxicity.
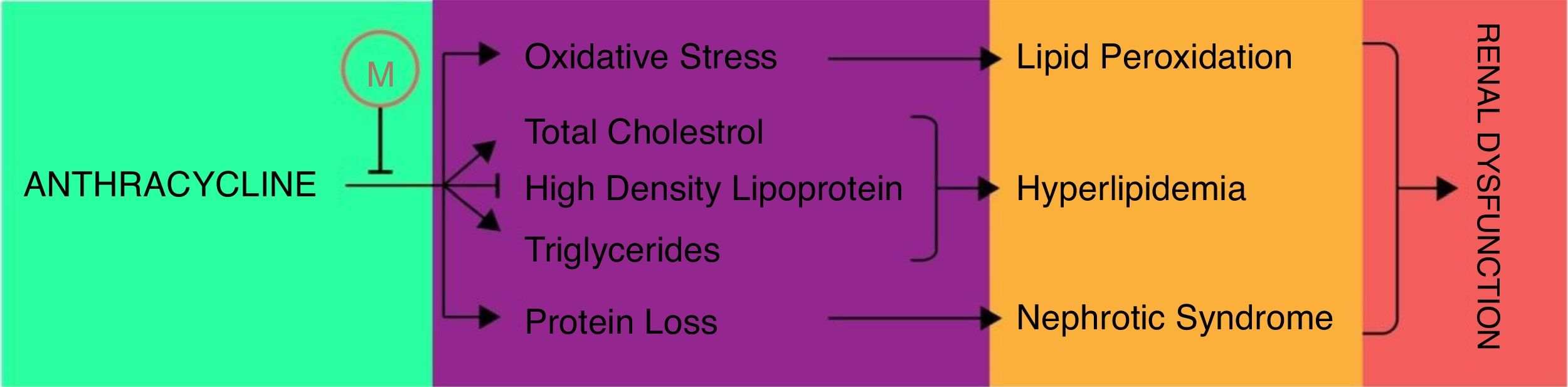
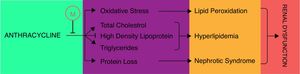
AnthracyclinesAnthracycline are broadly used antineoplastic agents that are effectively used to treat variety of solid and hematological malignancies. The therapeutic vitality of these antineoplastic agents, such as daunorubicin and doxorubicin, has been compromised by increased resistance and dose-dependent toxicities including severe nephrotoxicity.87 The mechanism of anthracycline associated nephrotoxicity is not clearly understood but it has been proposed that anthracycline metabolism may induce the mitochondrial toxicities such as membrane disruption, impairment of calcium homeostasis, induction of free radical, lipid peroxidation and release of cytochrome C that may contribute toward apoptosis or necrosis of glomerular and tubular cells.88–90 Montilla et al. first time reported the melatonin potential to ameliorate the doxorubicin (adriamycin) induced nephrotoxicity in terms of oxidative stress and hyperlipidemia.91 In this study melatonin co-administration was found to attenuate the adriamycin-induced oxidative stress by reducing the lipid peroxides and recovering the depleted GSH and CAT activity. Moreover, melatonin co-administration also efficiently recovered the depleted high density lipoprotein-cholesterol (HDL-C) and condensed the higher levels of total cholesterol (TC) and triglycerides (TG) except phospholipids (PL) which were induced in adriamycin nephropathy (Fig. 3). In addition to this, simultaneous melatonin administration also restored the normal serum creatinine, urea, total proteins and urinary protein in adriamycin treatment thereby limits the adriamycin induced nephrotic syndrome. Together these findings demonstrated that melatonin may impart protection against oxidative stress, nephrotic syndrome and hyperlipidemic nephropathies of adriamycin. In addition to doxorubicin, Dziegiel et al. also studied the daunorubicin and the putative nephroprotective role of melatonin in their toxicities.92 Biochemical investigations revealed that melatonin prevented the amplification of malondialdehyde (MDA) and 4-hydroxyalkenals (4-HDA) which were increased due to lipid peroxidation by sub-chronic administration of daunoruicin and doxorubicin. This protection was further supplemented with histological data which revealed the melatonin co-administration alleviate the protein deposits in lumen of tubules and glomerular vacuolization in nephrotoxicity of daunoruicin and doxorubicin. Hrenak et al. further substantiated the nephroprotection of melatonin, against doxorubicin, and revealed that melatonin offer the nephroprotection comparable to other reno-protective drugs such as captoprils, olmesartan and compound 21.93 Taking all these findings into considerations, authors suggested that melatonin addition into the daunorubicin and doxorubicin treatment may provide excellent protection to avoid their renal toxicities.
MethotrexateMethotrexate is, a dihydrofolate reductase inhibitor, extensively studied and clinically useful cytotoxic drug in multiple cancers (such as breast cancer, acute leukemia, head and neck carcinoma) and autoimmune diseases. Most of the cancers required higher doses of methotrexate which may induce the renal dysfunction and subsequently decreases its renal clearance. Reduced renal clearance of methotrexate has been associated to increase its half-life and making leucovorin ineffective which may induce the further toxicities such as myelosuppression, mucositis and more pronounced nephrotoxicity.94 Higher plasma concentration of methotrexate significantly decreases the NADPH production and subsequent depletion of GSH that ultimately results into impaired antioxidant defense system and cellular oxidative stress.95 Jahovic et al. suggested the melatonin (10mg/kg) potential to ameliorate the methotrexate associated GSH depletion, lipid peroxidation and myeloperoxidation.96 However, a more extensive study was conducted by Abraham et al. to investigate the melatonin ability to regulate the antioxidant defense system and evaluate its nephroprotective role in higher doses of methotrexate.97 In addition to previously studied biochemical parameter, they found that methotrexate severely reduced the cellular content of antioxidant enzymes such as SOD, CAT, GPx, GRd and GST. Apart of these biochemical impairments, methotrexate also caused the severe cortical glomerulo-tubular nephritis and medullary tubular edema and distention. Interestingly, when melatonin was co-administered, it significantly recovered the enzymes of cellular antioxidant defense system and alleviated the histopathological complications of methotrexate nephrotoxicity. It was found that melatonin does not only recover the antioxidant enzymes but also stimulate them to eradicate the cellular oxidative stress in methotrexate nephrotoxicity. Therefore, authors concluded that co-administration of melatonin in high dose methotrexate treatment might attenuate the nephrotoxicity and enhance the safety of methotrexate.
MechlorethamineMechlorethamine is a chemical warfare and strong DNA alkylating antineoplastic agent that has limited effectiveness due to its toxicity in healthy tissue owing to its vesicant property.98,99 Kunak et al. performed a study which not only highlighted the nephrotoxic behavior of mechlorethamine, but also investigated the protective role of melatonin and S-methylisothiouria (potent iNOS inhibitor) by taking biochemical and histological data into consideration.100 Transdermal administration of mechlorethamine 3.5mg/kg demonstrated the severe induction in TNF-α, IL-1β and NO and morphological damages such as glomerulo-peritubular congestion and mild mesangiolysis. In the present study, melatonin co-administration after 30min of mechlorethamine showed significant reductions in TNF-α to a level even lower than S-methylisothiourea. Moreover, simultaneous administration of melatonin also decreased the IL-1β levels comparable to the S-methylisothiourea co-administration in mechlorethamine nephrotoxicity. Although melatonin also significantly limits the NO levels but this rescue was comparatively lower than S-methylisothiourea. In addition to these biochemical parameters, melatonin and S-mehtylisothiourea were also found to mitigate the morphological abnormalities which suggested that melatonin does not only attenuate the oxidative stress but also alleviate the inflammatory injuries in mechlorethamine nephrotoxicity.
AntibioticsAminoglycosidesAminoglycosides are widely used antibiotics against most gram negative bacterial infections due their low bacterial resistance and potent bactericidal properties. Therapeutic benefits associated with the use of aminoglycoside have been restricted due to their high propensity to cause serious nephrotoxicity.101 Gentamicin, a strong cationic moiety, is mostly considered as prototype drug in studies of aminoglycosides associated nephrotoxicity as it is most nephrotoxic among this group.102 Gentamicin is believed to cause nephrotoxicity by its accumulation mainly in renal proximal tubular cells which results into lysosomal phospholipidosis mediated mitochondrial oxidative/nitrosative stress and subsequent inflammatory cascade and apoptosis.103 Shiffow and co-workers found that gentamicin cause severe degenerative granulation of renal cortex with marked lipid peroxidation. Biochemical analysis of rats, treated with gentamicin alone, revealed the significant elevation of malondialdehyde (MDA), reduction in creatinine clearance, increased blood urea and enhanced N-acetyl-β-D-glucosaminidase (β-NAG) renal excretion. However, these biochemical changes were reversed or significantly prevented when gentamicin was co-administered with melatonin. Simultaneous administration of melatonin and gentamicin revealed the marked reduction in the cortical granular degeneration and necrosis.104 Moreover, melatonin was also found to restore the enzymatic activity of antioxidant enzymes, i.e. SOD, CAT and GPx which were compromised in gentamicin induced oxidative stress.105 Ozbek et al. also found the melatonin role to moderate serum γ-glutamyltranspeptidase levels in gentamicin induce nephropathy that may suggest the possible mechanism to restore the GSH levels. Taking these findings into consideration, Sener et al. investigated the modulation of biochemical parameters to further elucidate the nephroprotective mechanism of melatonin against gentamicin. In addition to protection against lipid peroxidation, their investigation revealed that melatonin pretreatment demolishes the up regulated activity of myeloperoxidase (MPO) which is correlated to neutrophil infiltration in gentamicin nephropathy. In addition, Protein carbonyl concentration (indicator of protein oxidation – PO), was also decreased with melatonin pretreatment which suggests the melatonin potential to protect proteins from gentamicin induced oxidative damage.106 These finding substantiated the hypothesis of these researchers and suggested that melatonin may provide nephroprotection against gentamicin. Nephropathic pattern of amikacin is related to that of gentamicin but, comparatively, with lower nephrotoxicity.107 Parlakipnar et al. performed an animal study to investigate the melatonin physiological and pharmacological concentration relationship in attenuation of amikacin induced nephrotoxicity.108 It was found that amikacin treated rats with lower physiological concentration of melatonin, when given exogenous melatonin, showed significant improvements in GSH levels and reductions in lipid peroxidation. On the other hand, these improvements were not seen, when exogenous melatonin was given, in the amikacin treated rats with normal melatonin physiological concentration. This interplay of pharmacological and physiological concentration of melatonin has indicated the researchers to further investigate the role of exogenous melatonin to attenuate the amikacin induced nephrotoxicity in normal physiological concentration of melatonin.
ColistinColistin, aka polymixin E, is a peptide antibiotic that is used as last resort in infections of multidrug resistant gram negative microbes such as Pseudomonas aurigenosa, Klebsiella pneumonia and Acinetobacter baumannii. It has been suggested, with emergence of resistance against colistin suboptimal plasma concentration following current recommended regimen, to increase the recommended dose which is, however, limited due to its severe nephrotoxicity.109,110 Mechanism of colistin nephrotoxicity involves the increase cellular membrane permeability in tubular epithelium, increase in nitric oxide synthase (NOS) and increase caspase activity which results into cellular swelling, lysis, oxidative stress and subsequent apoptosis or necrosis of renal tubules.111 Yousef et al. evaluated the protective role of melatonin against colistin-induced nephrotoxicity through biochemical, histopathological and pharmacokinetic studies in an animal model.112 In preliminary studies, they determined the nephrotoxic potential of four different regimens of colistin to benchmark the regimen for reproducible nephrotoxicity. Accordingly, the increasing twice daily dose with cumulative 36mg/kg dose of colistin for 7 days caused the significant increase in serum creatinine and 2.4-fold elevation in N-acetyl-β-D-glucosamindase (NAG) renal excretion which were further substantiated with cortico-tubular necrosis in histopathological studies. In pharmacokinetic studies, significant urinary excretion of colistin was observed when it was administered alone. More interestingly, plasma concentration vs. time studies revealed the 2.5-fold increase in colistin plasma concentrations with 10mg/kg melatonin co-administration which suggested the increase in half life and decreased clearance of colistin. In addition to this, biochemical and histopathological parameters were also significantly improved with melatonin co-administration. Therefore, these finding proposed that melatonin co-administration may reduce the nephrotoxicity of colistin through pharmacokinetic modification. However, the mechanism by which melatonin alter the pharmacokinetics of colistin is still poorly known and needs further investigations into this domain. Moreover, the melatonin mediated scavenging of free radicals, in colistin induced oxidative stress, also require further studies in order to further explore its protective behavior in colistin nephrotoxicity.
VancomycinVancomycin is a glycopeptide antibiotic reserved for serious resistant gram positive infection such as methicillin-resistant staphylococcus aureus, enterococcus faecium and methicillin-resistant coagulase negative staphylococci. Vancomycin use has been reported to cause severe but reversible dose and duration limiting nephrotoxicity which compromise its therapeutic potential.113 Nephrotoxicity mechanism of vancomycin is suggested to be associated with its increase uptake in renal proximal tubular cells which induces the oxidative phosphorylation and generation of free radicals. Free radicals mediate the mitochondrial membrane depolarization and release of cytochrome C which in turn activate and induce the apoptosis process.114,115 A single study has reported the protective role of melatonin against vancomycin-induced nephrotoxicity in comparison with other important antioxidants.116 Intraperitoneal administration of vancomycin 10mg/kg, for 7 days, severely enhanced the serum creatinine, urea and malondialdehyde (MDA) levels which indicated the impaired renal function and cellular injury. In addition to this, vancomycin was also found to reduce cellular superoxide dismutase and glutathione peroxidase levels that implied the weaken antioxidant system. Co-administration of melatonin 10mg/kg resulted in restoration of creatinine excretion, MDA levels, SOD and GPx activity comparable to control group. Melatonin was found to be most effective in restoring the creatinine and glutathione (GSH) levels as compare to ginkgo biloba, α-lipoic acid and amrinone. However, melatonin mediated restoration of MDA, urea, superoxide dismutase (SOD) was comparable to ginkgo biloba and amrinone but more than α-lipoic acid. Therefore, this study provided some key findings to evaluate the nephroprotective effect of melatonin as compare to other important antioxidants and suggested that melatonin co-administration can be effective adjunct to compensate the vancomycin nephrotoxicity and improve the effectiveness of vancomycin.
CiprofloxacinCiprofloxacin is broad spectrum fluoroquinolone antibiotic, effective against gram negative and gram positive micro-organisms. Acute renal failure is, a dose limiting adverse effect, considered one of most common toxicity that complicates the treatment with ciprofloxacin. Mechanism of ciprofloxacin toxicity is not clearly understood but is manifested as crystal nephropathy, interstitial nephritis and renal failure.117–120. Shaki et al. conducted a study to elucidate the mechanism of ciprofloxacin nephrotoxicity and its attenuation by melatonin co-administration.121 They found that ciprofloxacin 100mg/kg/day, for 8 days, significantly increase the serum creatinine and BUN levels which were effectively restored with melatonin 10mg/kg co-administration. Amplification of lipid peroxidation, protein carbonyl concentration, reactive oxygen species and depletion of GSH were also observed with ciprofloxacin administration and these were also alleviated with melatonin co-administration. Besides melatonin co-administration, vitamin E co-administration was also investigated to compare the effectiveness of melatonin. Although Vitamin E, comparable to melatonin, lowered the lipid peroxidation, reactive oxygen species and protein carbonyl concentrations but it was ineffective to restore the creatinine, BUN and GSH. Moreover, tissue necrosis factor alpha (TNF-α) and nitric oxide were also significantly increase in ciprofloxacin administration which were effectively prevented with melatonin co-administration. These findings were further endorsed with significant reduction in severity of tubular dilation, epithelial degeneration, hyaline granules and necrosis by melatonin co-administration. Therefore, together all these findings proposed that ciprofloxacin may induced nephrotoxicity through increased generation of free radicals, nitric oxide, TNF-α and stimulate the inflammatory process which can be effectively attenuate by melatonin co-administration owing to melatonin's antioxidant effect.
TenofovirTenofovir, a nucleoside reverse transcriptase inhibitor (NRTI), is effectively used in HIV and Hepatitis B viral infections. The therapeutic efficacy of tenofovir has been limited by its dose and time related nephrotoxicity which is manifested by Fanconi's syndrome, nephrogenic diabetes inspidus and acute tubular necrosis.122 Nephrotoxicity of tenofovir implies the reduced efflux transport activity of multidrug resistant proteins (i.e. MRP2, MRP-4) and enhanced activity of organic anion transporter (hOAT-1) in proximal tubules which results into the accumulation of tenofovir in tubular cells. Sub-cellularly, tenofovir inhibits the mitochondrial DNA polymerase-γ mediated replication of mitochondrial DNA (mtDNA). Subsequently it reduces the mitochondrial enzymatic content for oxidative phosphorylation and electron transport chain which ultimately leads to the impaired mitochondrial function and oxidative cellular injuries.123 Ramamoorthy et al. found that 600mg/kg oral tenofovir was able to induce the Fanconi syndrome, acute tubular nephrotoxicity and renal dysfunction and these abnormalities were effectively countered by melatonin pretreatment.124 Biochemical data revealed that tenofovir induce the urinary potassium, phosphate, bicarbonates, urea and serum uric acid which were restored, except phosphate, with melatonin (20mg/kg) pretreatment. Melatonin also completely restored the tenofovir associated twofold increased protein carbonylation and nine fold increased myeloperoxidase activity that explained the melatonin's ability to attenuate the tenofovir induced oxidative stress and inflammation. Following tenofovir administration GSH levels and activity of major antioxidant enzymes, such as SOD, GPx, GRd and GST, were reduced to about half of their physiological activity. Melatonin pre-treatment significantly improved the activity of GST and GRd whereas it was found ineffective against tenofovir induced reductions in the SOD, GPx activity and GSH levels. Apart of its effect on oxidative stress, melatonin also strongly inhibits the 50% rise of nitrosative stress in tenofovir nephrotoxicity. Histopathological data also revealed the melatonin ability to alleviate the severity of cytosolic vacuolization, mitochondrial swelling, cristae fragmentation, epithelial disruptions and interstitial oedema in tenofovir induced nephrotoxicity. Therefore, together, these investigations suggested the melatonin potential to ameliorate the oxidative, nitrosative and inflammatory damages of tenofovir nephrotoxicity.
ImmunosuppresantsCyclosporinCyclosporin, a calcineurin inhibitor, is effectively used immunosuppressant in prevention of allograft rejection of most solid organs and autoimmune aggravations. Cyclosporin has proven its therapeutic efficacy in prevention of transplantation rejection for short period which is limited by its severe dose-dependent deleterious nephrotoxicity in long term use.125–127 Mechanism underlying the cyclosporine induced nephrotoxicity involves the reversible hemodynamic impairments and irreversible tubule-interstitial chronic inflammation and fibrosis. Renal hemodynamic damages are associated with the marked vasoconstrictions mediated by provoked renin–angiotension–aldosterone system (RAS), cyclooxygenase pathway and reduced nitric oxide (NO) which further induced the ischemic oxidative damage, macrophage infiltration and acute inflammatory cascades. However, long-term use of cyclosporine may impart the chronic inflammation which may further induce irreversible tubulo-interstitial fibrosis and scarring in cortico-medullary area by profibrogenic mediators such as TGF-β.128 Kumar et al. first time investigated the nephroprotective capacity of melatonin in cyclosporine induced nephrotoxicity.129 In this animal study, melatonin was found to attenuate the cyclosporine-induced lipid peroxidation and cortico-medullary microcalcification. In addition to this, cyclosporine was also found to impair the renal function implied by elevated blood urea and reduction in creatinine and lithium clearance which were improved by the melatonin co-administration. Subsequently, another study also reported the melatonin ability to reduce the lipid peroxidation and improve the renal function during cyclosporine administration without compromising the effectiveness of cyclosporine.130 Stacchiotti conducted the electron microscopic analysis to reveal the effectiveness of melatonin to attenuate the cyclosporine-induced nephropathies at ultrastructural level.131 This morphometric study provided that melatonin alleviates the apoptotic nuclei, distended mitochondria, microvilli disruption, fused podocytes and glomerular basement membrane destructions in cyclosporine nephrotoxicity. Moreover, quantitative data suggested that melatonin may attenuate the cyclosporine induced tubule-interstitial fibrosis and atrophy. Longoni et al. further supported previous studies in an isolated and perfused rat kidney model to investigate the melatonin effect without any metabolic regulation.132 Lipid peroxidation was found to be the primary outcome of cyclosporine-induced nephrotoxicity which was manifested by elevated malondialdehyde (MDA) and 4-hydroxyalkenals (4-HDA). Moreover, morphological abnormalities, such as glomerular collapse and tubular damage, and NO metabolites were also fostered as part of nephrotoxicity progression. Pre-treatment with melatonin was found to significantly reduce the lipid peroxidation and morphological damages which they endorsed to the antioxidant property of melatonin. Besides, melatonin did not reveal any important reduction in reactive metabolites of NO. Therefore, authors concluded that melatonin might attenuate the oxidative damaged in cyclosporine nephrotoxicity. However, studies are needed to further investigate the possible mechanism of melatonin to attenuate the cyclosporine induced nephrotoxicity.
TacrolimusTacrolimus is a calcineurin inhibitor which is broadly used in the heart, liver and kidney allograft transplantation. Tacrolimus associated immunosuppression involves the inhibition of calcium-calmodulin dependent phosphatase activity of calcineurin which ultimately modulate the intracellular calcium concentration, NF-κB translocation, IL-2 production and activation of T lymphocytes. Tacrolimus-induced nephropathy follows the similar molecular mechanism to cyclosporine but with lower nephrotoxic potential and less severe pathological manifestations.133 Ara et al. reported the intraperitoneal 5mg/kg tacrolimus was found to induce the MDA, IL-6, NO and TNF-α concentrations which were correlated to acute and chronic nephrotoxicity of tacrolimus.134 Whereas melatonin (4mg/kg) co-administration was shown to significantly reduce the NO, TNF-α and IL-6 concentrations owing to its nephroprotective potential against tacrolimus induced renal inflammation. But MDA levels, in tacrolimus co-administered with melatonin, were not significantly different from that of tacrolimus alone which implied that further investigations are needed to elucidate the melatonin ability to attenuate the tacrolimus induced lipid peroxidation or oxidative stress and its correlation to endogenous antioxidant defense.
Anti pyreticsAcetaminophenAcetaminophen, N-acetyl-P-aminophenol, is widely and most commonly used antipyretic and analgesic drug. It has been reported to be the most common toxic ingestion and purported to be nephrotoxic in overall 1–2% cases of overdose. Nephrotoxic mechanisms of acetaminophen have been proposed to be associated with its renal CYP450 metabolism, activation through prostaglandin endoperoxidase synthetase and de-acetylation through N-deacetylase into reactive metabolites or free radicals.135 Sener et al. evaluated the protective role of melatonin in acetaminophen nephrotoxicity in an animal study while comparing its protective efficacy against vitamin E and N-acetyl cysteine.136 A single intraperitoneal 900mg/kg injection of acetaminophen was shown to significantly enhance the plasma MDA, tissue myeloperoxidase (MPO) activity and protein oxidation (PO) after 4h of administration while such damage was more evident after 24h. These enhanced biochemical parameters were accompanied with marked reduction in the GSH levels which implied the oxidative stress or renal damage. When 10mg/kg melatonin was given 30min before acetaminophen, it significantly restored the GSH and reduced the oxidative damage by lowering MDA, MPO activity and PO comparable to the N-acetylcysteine pretreatment while it was shown more effective than vitamin E pretreatment. Protective role of melatonin was further investigated with respect to its potential to induce the antioxidant enzyme system. Melatonin co-administration, with acetaminophen, was found to markedly stimulate the activities of superoxide dismutase (SOD), glutathione peroxidase (GPx) and catalase (CAT) which were compromised in the acetaminophen administration only. Histopathological data of melatonin co-administration revealed the marked reduction in the acetaminophen induced vacuolization, degeneration, cellular desquamation and necrosis in proximal tubules. These studies suggested the nephroprotective activities of melatonin might be associated with its potent antioxidant properties which implied the melatonin to be a nephroprotective adjunct in the treatment of acetaminophen overdose.
Melatonin as nephroprotective adjunct in clinical studiesThe safety and efficacy of melatonin have been extensively investigated in several clinical trials that documented its potential therapeutic usefulness in variety of disease conditions.137 There has been a major drive in patent applications claiming the therapeutic exploitation of melatonin and its analogues with pre-dominant focus on cancer, neurological, immunological and metabolic diseases.138 Interestingly, the proven therapeutic potential of melatonin or its use as a therapeutic adjunct resulted into the successful development of its hybrid ligands such as with tacrine in Alzheimer's.139 There are some clinical interventions that highlight the relationship between nocturnal secretion of melatonin and renal physiology and suggest the role of melatonin to reduce the renal blood flow and urine concentration of sodium, potassium, urate and chloride ions.140,141 These temporal changes in renal function may suggest the role of melatonin to influence the development of renal colic.142 Moreover, the allelic variation of Melatonin receptor 1A (MTNR1A) gene is also associated with pre-disposition to calcium nephrolithiasis.143 Interestingly, the development or severity of chronic kidney disease (CKD) accompany significant impairments in endogenous melatonin secretions that may warrant its exogenous administration.144 Recently, the effect of exogenous melatonin supplementation has been assessed for its therapeutic benefits in CKD associated risk factors and sleep disorders.145,146 Melatonin co-administration has been found to enhance the survival rate and efficacy of IL-2 immunotherapy by attenuating the immunosuppressive effect of opioids without compromising its analgesic effect in palliative care of metastatic renal cancer patients.147 Pre-treatment of melatonin has also been found to ameliorate the iron and erythropoietin-induced oxidative stress in anemic patients with end-stage renal failure.148 A double-blind placebo controlled clinical trial has revealed that concomitant administration of melatonin and zinc acetate ameliorate the poorly controlled microalbuminuria induced renal failure in type 2 diabetes mellitus patients treated with biguanides.149 Moreover, there is an ongoing randomized, double-blind, placebo controlled clinical trial to address the nephroprotective potential of melatonin in polymixin B induced nephrotoxicity.150 Despite the impressive pre-clinical data, there are still limited clinical interventions that address the putative role of melatonin in amelioration of drug-induced nephrotoxicity.
ConclusionTherapeutic doses of several important drugs have become suboptimal due to the emergence of drug resistance that necessitates the use of toxic higher doses. These higher doses are, however, associated with severe toxicities that need therapeutic adjuncts to compensate the toxicities. Favorable safe nature and nephroprotective property of melatonin has suggested it to be a pharmacological adjunct to increase the therapeutic window of important nephrotoxic drugs in clinical trials. Mitigation of drug-induced nephrotoxicity by melatonin involves the complex series of biochemical improvements at cellular levels. Melatonin attenuates the nephrotoxicity of most drugs through its potent antioxidant action as it is believed to reinforce the antioxidant enzymes and direct free radical scavenging at subcellular levels. Apart of its antioxidant actions, modulation of various inflammatory cytokines was also associated with its ability to restore the balance of apoptosis and cell survival. However, previous studies have only endorsed the melatonin direct antioxidant and anti-inflammatory action without any interventions of melatonin receptors (MT1, MT2), found on kidney, in culmination of drug induced nephrotoxicity. Therefore, investigation regarding melatonin receptors involvement in mitigation of drug induced nephrotoxicity may further justify the melatonin induced moderation of inflammatory cytokines and stimulation of antioxidant enzymes.
Conflict of interestNone.
We are thankful to Dr. Zaki Hussain Rizvi for proofreading the manuscript.