Acute post-infectious glomerulonephritis is an immune complex-mediated glomerulonephritis that classically occurs in children following streptococcal upper respiratory or skin infections. However, cases of IgA-dominant infection-related glomerulonephritis (IgA-IRGN), a diffuse endocapillary proliferative glomerulonephritis associated with intense IgA deposits following staphylococcus infection, have been increasingly reported in recent literature. In contrast to typical acute post-infectious glomerulonephritis, the causative infection is ongoing at the time of diagnosis.1 It often occurs in diabetic elderly patients and presents with acute kidney injury and heavy proteinuria. The prognosis is unfavourable. Here, we report an elderly-male patient with IgA-IGRN who demonstrated vasculitic skin rash.
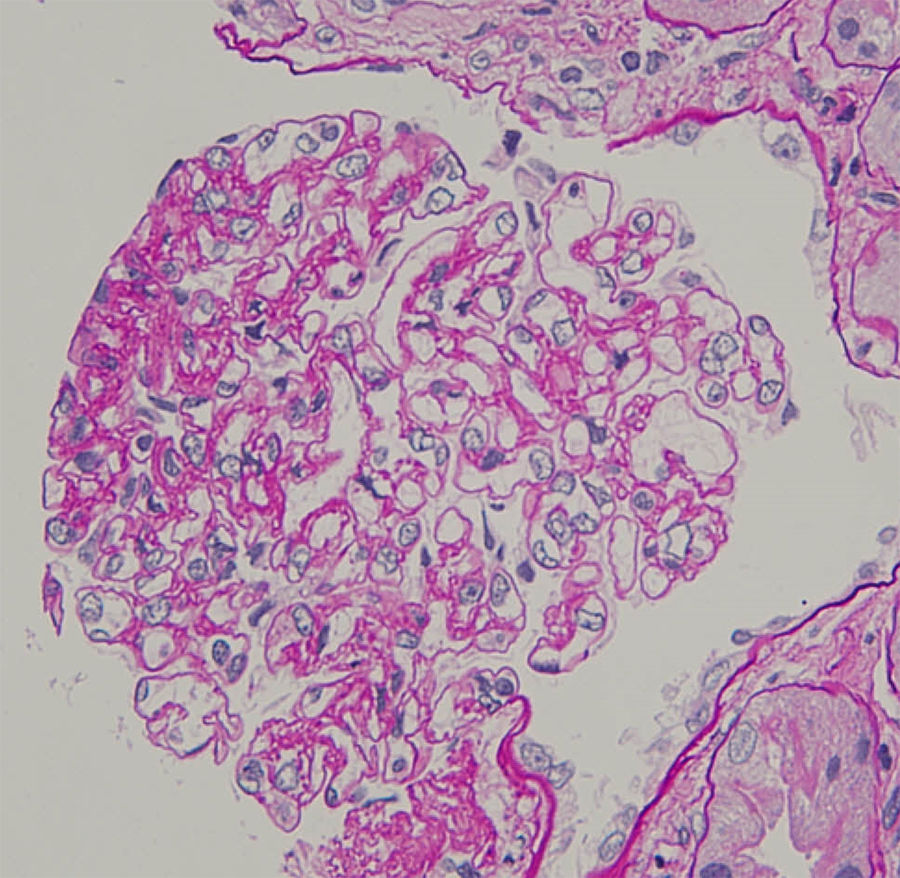
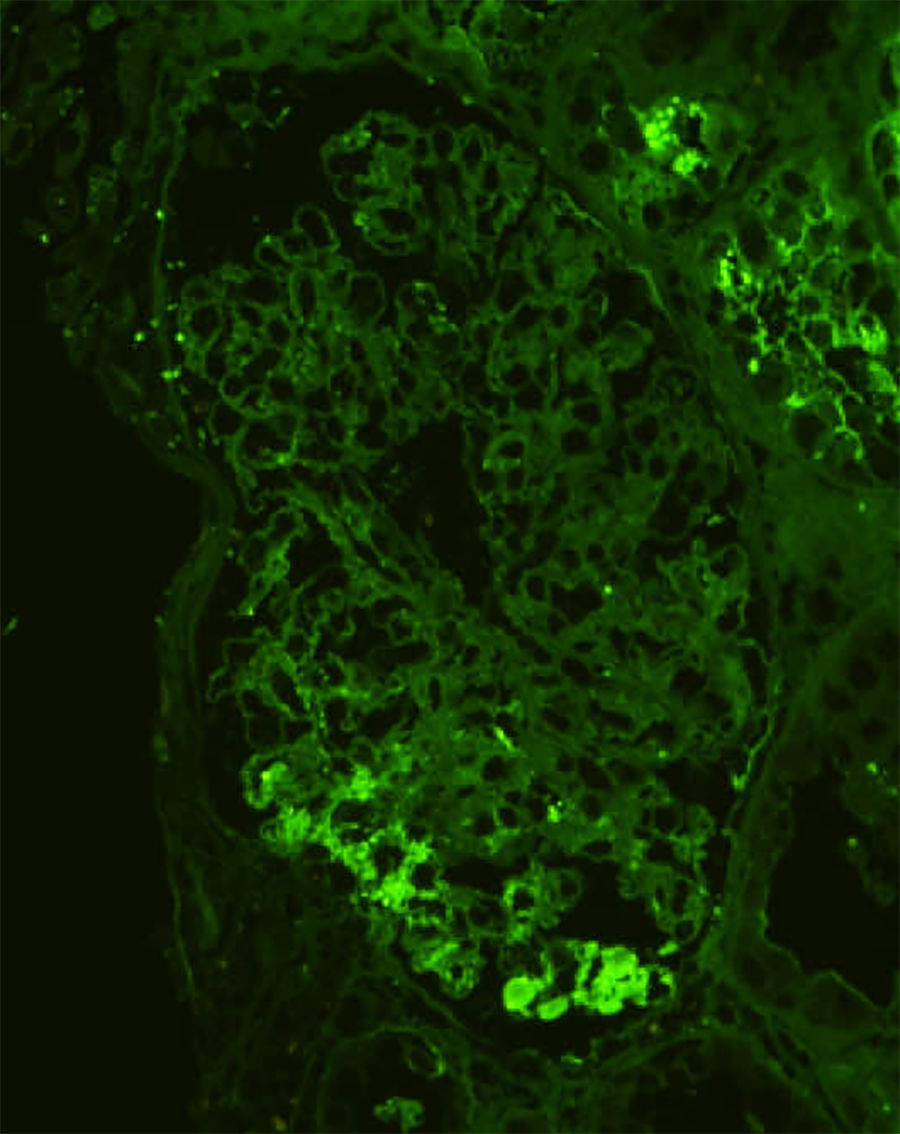
An 80-year-old Japanese man with stage G3a chronic kidney disease due to diabetes mellitus was admitted to the general medicine service for management of right haemopneumothorax after a motor vehicle accident and subsequent empyema caused by methicillin-sensitive Staphylococcus aureus treated with four weeks of intravenous cefazolin and drainage. The patient was seen in nephrology consultation for a rise in serum creatinine level from 1.4mg/dl upon admission to 5.3mg/dl (estimated glomerular filtration rate of 9ml/min/1.73m2) and purpuric lesions on the bilateral lower extremities. At the general medicine service, oral glucocorticoid was initiated for a presumed diagnosis of IgA vasculitis (Henoch-Schönlein purpura). However, the kidney function continued to worsen, and oliguria ensued despite infusion of lactated ringer's solution. Upon physical examination, he was not in distress. The blood pressure was 110/78mmHg. There were trace pretibial oedema and purpuric lesions on the bilateral legs. Urinalysis showed >100 red blood cells per high-power field (70% dysmorphic) and 5.2g/24h of proteinuria. Immunological analysis showed that increased immunoglobulins (IgG 1427mg/dl; IgA 1373mg/dl; IgM 48mg/dl) and decreased complement levels (C3 77mg/dl; C4 30mg/dl; CH50 44IU/ml). Antinuclear antibodies, antimyeloperoxidase antibodies, antiproteinase 3 antibodies, and a glomerular basement membrane antibody were negative. Renal biopsy showed 10 glomeruli, 4 of them sclerosed and 2 with endocapillary proliferation (Fig. 1). There was a mild interstitial infiltrate of polymorphonuclear cells, and the arteries had no significant lesions. Immunofluorescence microscopy showed diffuse depositions of IgA (Fig. 2) and C3. Electron microscopy could not be performed because of insufficient specimens. Based on clinical features and pathologic findings, IgA-IRGN was diagnosed. Intravenous cefazolin was administered for another four weeks, and oral glucocorticoid was tapered and stopped gradually without relapse of infection. However, the patient progressed to end-stage kidney disease even after 20mg of olmesartan was initiated.
IgA-IRGN is a morphologic variant of acute postinfectious glomerulonephritis mainly associated with staphylococcal infection, low serum complement, and underlying diabetic nephropathy.2 At least three of the following criteria are required for diagnosis: clinical or laboratory evidence of infection preceding or at the onset of glomerulonephritis; depressed serum complement; endocapillary proliferative and exudative glomerulonephritis; C3-dominant or co-dominant glomerular immunofluorescence staining; and hump-shaped subepithelial deposits on electron microscopy.3 The patient fulfilled four of the criteria. The mainstays of treatment are eradication of infection and management of nephritis, but the role of steroids is controversial. The prognosis is unfavourable, and 23% of patients progressed to dialysis-dependent end-stage kidney disease.4
IgA vasculitis should be differentiated from IgA-IRGN, but it may be difficult on morphologic grounds because endocapillary hypercellularity is common in IgA vasculitis. Clinical features that favour IgA vasculitis over IgA-IRGN include purpura, arthritis, abdominal pain and gastrointestinal bleeding, and the presence of normal serum complement levels.5 However, vasculitic skin rash can be observed in patients with IgA-IRGN. One research reported that 18% of patients with IgA-IRGN had purpura,6 and another reported that IgA-IRGN may have IgA vasculitis-like lower extremity purpuric lesion.7 Our patient was diagnosed with IgA-IRGN because of the absence of arthritis, abdominal pain, gastrointestinal bleeding, and decreased complement levels.
Ethical approvalAll procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee at which the studies were conducted (IRB approval number: 2017-107) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consentInformed consent was obtained from all individual participants included in the study.
FundingThis research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.