Hepatitis C virus infection and chronic kidney disease are major public health issues all over the world. It has been suggested a role of HCV as a risk factor for the development and progression of chronic kidney disease (defined by reduced glomerular filtration rate and/or detectable proteinuria) in the general population but conflicting findings have been given.
Material and methodsA systematic review of the published medical literature was conducted to assess whether positive HCV serologic status is associated with greater rate of proteinuria in the adult general population. We used a random-effect model to generate a summary estimate of the relative risk of proteinuria with HCV across the published studies.
ResultsWe identified 23 studies (n=198,967 unique patients) and performed separate meta-analyses according to the study design. Overall effect estimate was significant in cross-sectional (OR, 1.47, 95%CI, 1.3; 1.66) (P<0.001) and obvious between-study heterogeneity was observed (Q value by Chi-squared [χ2] test 27.3, P=0.02). The risk of proteinuria after exposure to HCV was also consistent among longitudinal studies (HR, 1.79, 95% CI, 1.17; 2.74) (P<0.001) and between-study heterogeneity occurred (Q value, 27.82 by X2 test, P=0.0001). Stratified analysis did not report heterogeneity in several comparisons-pooling studies based on urine protein/creatinine ratio (UACR) showed that the adjusted OR with HCV was 1.64 (95% CI, 1.41; 1.91, P<0.001) without heterogeneity (Q value by Chi-squared [χ2] test 9.98, P=NS). Meta-regression recorded a link between greater prevalence of proteinuria in males with HCV exposure (P=0.03). Studies based on univariate analysis (n=6, n=72, 551 unique patients) gave similar results, pooled OR 1.54 (95% CI, 1.08; 2.19) (P=0.0001).
ConclusionsAn important relationship between HCV infection and higher risk of proteinuria in the general population exists. Research aimed to understand the biological mechanisms underlying such association is under way. We encourage to screen all patients with HCV exposure for proteinuria.
La infección por virus de hepatitis C y la enfermedad renal crónica son cuestiones de salud pública mayores a nivel mundial. Se ha sugerido el rol del VHC como factor de riesgo para el desarrollo y progresión de la enfermedad renal crónica (definida como reducción de la tasa de filtración glomerular y/o proteinuria detectable) en la población general, aunque se han aportado hallazgos contrapuestos.
Material y métodosSe realizó una revisión sistemática de la literatura médica publicada, para evaluar si el estatus serológico positivo de VHC está asociado a una mayor tasa de proteinuria en la población general adulta. Utilizamos un modelo de efecto aleatorio para generar una estimación resumida del riesgo relativo de proteinuria con VHC en los estudios publicados.
ResultadosIdentificamos 23 estudios (n = 198.967 pacientes únicos) y realizamos un metaanálisis separado, de acuerdo con el diseño del estudio. En general, la estimación del efecto fue significativa en los estudios transversales (OR, 1,47, 95% IC, 1,3; 1,66) (p < 0,001), observándose heterogeneidad obvia entre estudios (valor Q mediante la prueba χ2 27,3, p = 0,02). El riesgo de proteinuria tras la exposición a VHC fue también consistente entre los estudios longitudinales (HR, 1,79, 95% IC, 1,17; 2,74) (p < 0,001) produciéndose heterogeneidad entre estudios (valor Q, 27,82 mediante la prueba χ2, p = 0,0001). El análisis estratificado no reportó heterogeneidad en diversos estudios comparativos-agrupados basados en el ratio proteína/creatinina en orina (UACR), reflejando que el valor de OR ajustado con VHC fue de 1,64 (95% IC, 1,41; 1,91, p < 0,001) sin heterogeneidad (valor Q mediante la prueba χ2 9,98, p = NS). La metarregresión registró un vínculo entre la mayor prevalencia de proteinuria en varones con exposición a VHC (p = 0,03). Los estudios basados en análisis univariantes (n = 6, n = 72.551 pacientes únicos) arrojaron resultados similares, OR agrupado de 1,54 (95% IC, 1,08; 2,19) (p = 0,0001).
ConclusionesExiste una relación importante entre la infección por VHC y el mayor riesgo de proteinuria en la población general. La investigación dirigida a comprender los mecanismos biológicos subyacentes a dicha asociación está en curso. Animamos a cribar la proteinuria en todos los pacientes con exposición al VHC.
Hepatitis C virus infection is an important agent of liver disease worldwide. Recent information has been accumulated showing that chronic hepatitis C virus infection plays important activities in various tissues and organs other than the liver.1 It appears that the kidney is an important target of the extra-hepatic activity of HCV and a relationship between HCV and CKD has been recently mentioned. The relationship between CKD and HCV infection is bi-directional as HCV is both a cause and consequence of chronic kidney disease. HCV and CKD are major public health issues all over the world, the Global Burden of Disease (GBD) Chronic Kidney Disease Collaboration has reported that the global prevalence of CKD in 2017 was 9.1% (95% uncertainty interval [UI] 8.5–9.8), which is roughly 700 million cases. According to the 2023 report from the World Health Organization, an estimated 58 million people globally have chronic hepatitis C virus, with about 1.5 million new infections occurring per year. WHO in 2016 established the goal to eliminate HBV and HCV as a public health threat by 2030 and we are currently very far from this goal; in fact, only 21% of the 58 million with chronic HCV had been diagnosed in 2019, and 13% treated globally.2
The current prevalence of chronic kidney disease cannot fully explained by conventional risk factors such as comorbidities (arterial hypertension, diabetes and ageing, among others) or life-style factors (smoke, overweight, etc.); additional agents have been mentioned such as positive family history of CKD3 or viral hepatitis (mostly, HBV and HCV).1 Our meta-analysis of clinical observational studies (n=40; 4,072,867 patients) had revealed a relationship between anti-HCV positive serologic status and increased incidence of chronic kidney disease; the summary estimate for adjusted HR with HCV across the surveys, 1.54 (95% CI, 1.26; 1.87) (P<0.001).4 However, between-study heterogeneity was found (Q value by Chi-squared [χ2] test 500.3, P<0.0001).
The frequency of proteinuria according to anti-HCV positive serologic status was also assessed (n=10 studies). There was a consistent link between positive anti-HCV serologic status and increased frequency of proteinuria, adjusted risk of proteinuria associated with HCV across the surveys, 1.633 (95% CI, 1.29; 2.05) (P<0.001). These results were not conclusive for at least two reasons – large heterogeneity (Q value by Chi-square test [χ2]=37.47 (I2=75.9%) (P=0.0001), and limited number (n=10) of clinical studies.4 Several mechanisms have been advocated to explain the increased risk of proteinuria in HCV-infected individuals. Although chronic HCV infection is associated with tubulo-interstitial injury, the most common type of HCV-associated kidney damage is glomerular disease and an association between HCV infection and glomerular disease has been observed in native kidneys and after solid organ transplant.1 A variety of glomerular diseases, such as membrano-proliferative glomerulonephritis, membranous nephropathy, focal segmental glomerulosclerosis, IgA nephropathy, interstitial nephritis, renal thrombotic microangiopathy, and proliferative glomerulonephritis have been identified in patients with exposure to HCV. The most frequent HCV-related glomerular disease is immune complex-mediated membrano-proliferative glomerulonephritis, usually reflecting the presence of type II cryoglobulinemia. Cases of HCV-associated MPGN without cryoglobulinemia have not infrequently been reported.1
The recent publication of large and additional studies has led us to summarize again the scientific information concerning the relationship between proteinuria and exposure to HCV infection. We have performed a systematic review of the medical literature with a meta-analysis of clinical observational studies.
Material and methodsThis work is in agreement with the Preferred Reporting Items for Systematic reviews and Meta-analyses statement.5
Search strategy and data extractionCitations in English language from the national Library of Medicine's Medline database from 1989 to June 20, 2023 were evaluated by two authors (F.F., and C.M.A.). Initial testing for HCV was made in 1989 and data on HCV status are therefore not available for the time before 1989. The current research was performed by four Medline databases engines (Embase, Grateful Med, Ovid, and PubMed), and was limited to human studies.
The following algorithm in medical subject heading and in free text words was applied: (‘HCV’ or ‘HCV antibody positive serologic status’ or ‘HCV viremia’ or ‘Hepatitis C’ or ‘Hepatitis C Virus Infection’ or ‘HCV RNA’) AND (‘Albuminuria’ or ‘Macroalbuminuria’ or ‘Microalbuminuria’ or ‘Proteinuria’ or ‘Albumin/creatinine ratio’), AND (‘Incidence’ or ‘Prevalence’).
We performed an additional search with electronic searches of the Cochrane Library; manual searches of selected specialty journals were made to identify all pertinent literature. We also searched reference lists from qualitative topic reviews and published clinical studies. It was previously demonstrated that a Medline search alone might not be sensitive enough.6 Data on study design, study period, patient characteristics, HCV prevalence, antiviral therapy towards HCV, and kidney disease outcomes were abstracted. Authors of selected papers were contacted to obtain missing data and only information from patients with known HCV status were included in the meta-analysis. We achieved consensus for all data. We deleted duplicate reports for the same patients, which included contact with authors when necessary. We pre-specified eligibility and exclusion criteria. Our search was limited to human studies that were published in the English literature.
Inclusion criteriaWe retrieved studies if they met numerous inclusion criteria as follows:
- •
They presented original data from cohort and longitudinal studies;
- •
The outcome of interest was clearly defined as frequency of detectable proteinuria in the adult general population according to HCV status;
- •
The studies provided adjusted risk estimates and their confidence intervals. Both case–control and cohort studies were considered as eligible for inclusion in the analysis.
If data on the same population were duplicated in more than one report, we enrolled the most recent study in the analysis. Information on HCV status was identified at the time of enrolment. We included reports where the diagnosis of HCV infection was conducted by testing for anti-HCV antibody in serum and/or detection of HCV RNA by nucleic acid testing. Surveys based on administrative codes (ICD-9) were also addressed.
Ineligible studiesWe have excluded studies if they reported inadequate information or data on the link between proteinuria and HCV positive status. We have excluded unpublished studies and reports that were published in abstract form only. Letters and reviews (narrative or systematic reviews, meta-analyses) were not considered.
Quality assessmentThe quality of the 23 studies was evaluated using a scale from the ‘Newcastle–Ottawa Scale (NOS)’.7 The Newcastle–Ottawa Scale is a scoring system that assesses all aspects of an observational epidemiologic survey from a methodological point of view. When a study included relevant information that could be associated with the NOS, one point was added. Seven items in cross-sectional studies and eight items in cohort and case–control studies that could be related to the NOS were retrieved. Therefore, cross-sectional studies assigned 8–10, 6–7, 4–5, or 0–3 points (stars) were assessed as very good, good, satisfactory or unsatisfactory studies, respectively. Similarly, cohort and case–control studies with 7–9, 5–6, 4 and 0–3 points (stars) were retrieved as very good, good, satisfactory or unsatisfactory studies, respectively. We carried out subgroup analyses based on those reports provided with very good quality. Data extraction and quality scoring were performed independently by two reviewers (F.F., and F.M.D.) and the results were merged by consensus. The complete protocol for quality scoring is available on-line.
Outcomes measuresWe made separate meta-analyses according to the outcome. The current meta-analysis included cross-sectional studies and longitudinal studies addressing the prevalence and incidence of proteinuria, respectively. The primary end-point was to provide adjusted estimates of the risk (and 95% CIs) of incidence (or prevalence) of proteinuria according to HCV infection. Multivariate analysis was conducted in each study to evaluate the independent impact of HCV positive status on the frequency of proteinuria after adjustment for potential confounders (covariates) (e.g., age, gender, diabetes mellitus, and others). Cox proportional hazard regression analysis and logistic regression analysis were carried out in longitudinal and cross-sectional studies, respectively.
An additional outcome was the occurrence of albuminuria according to HCV infection in studies without adjustment for covariates (univariate analysis only).
Data synthesis and analysisWe weighted study-specific log hazard ratios (relative risks or odds ratios for cohort studies) by the inverse of their variances to obtain a pooled effect estimate and its 95% confidence intervals. We used the assumption that odds ratios provide a reasonable approximation of the RR in case control studies and in cohort studies in which the outcome occurs unfrequently (around 10%) in the unexposed population. In addition, we assumed that hazards can be thought as incidence rates (dichotomous, non-time-varying exposures) and thus the HRs can be roughly interpreted as incidence rate ratios. We combined cohort studies and case–control studies in the absence of statistical heterogeneity. For each study, we adopted the estimate of the effect measure that was adjusted for the largest number of confounders. When results from the fixed and random effects models were different, we presented the second as it represents a more conservative approach. We adopted the random-effects model, as reported by DerSimonian and Laird,8 and the heterogeneity was evaluated by Ri (i.e., the proportion of total variance due to between-study variance). Heterogeneity was considered consistent if Ri was ≥0.75. The origin of the heterogeneity was addressed adopting stratified analysis: we identified subgroups of studies defined by study characteristics such as country of origin, reference year, or size. Sensitivity analysis adopting a fixed-effect model was also conducted to evaluate the consistency of results. We assessed publication bias graphically by using a funnel plot as well as quantitatively with Egger's regression. Multivariate meta-regression analysis was performed with the aim to observe the impact of continuous covariates on the outcome (prevalence or incidence of proteinuria among HCV-infected patients). Statistical analysis was performed with the software HEpiMA (version 2.1.3)9 and Comprehensive Meta-analysis (Biostat Inc., USA, 2005). A 5% significance was adopted for the alpha risk. Every estimate was given with its 95% CI.
ResultsLiterature reviewAs listed in Fig. 1, we identified 452 articles and 68 were evaluated for eligibility. The list of the 68 full-text papers is reported available on request. Twenty-three studies met our inclusion criteria and were published in 22 papers10–31 and carried out in 3 continents (Europe, Asia and America); a total of 198,675 unique patients were retrieved. Thus, one study contributed data on more than one kidney disease outcome; however, the cohort was considered once in our meta-analysis. There was a 100% concordance between reviewers with regard to final inclusion and exclusion of studies reviewed based on the predefined inclusion and exclusion criteria.
Information on HCV status was collected at the time of study inclusion. We pooled studies where the diagnosis of HCV infection and chronic kidney disease were done by administrative codes (ICD-9-CM codes).
Patient characteristicsTables 1 and 2 report some salient demographic and clinical characteristics of subjects enrolled in the included studies. The mean age of subject cohorts ranged from 38.5 to 60.8 years. The gender distribution ranged from 0 to 100% male. The average follow-up was between 15 and 132 months (longitudinal studies) and the pooled prevalence of HCV positive individuals was 0.19 (95% CI, 0.17; 0.21). Nine studies were from the USA, seven from Asia, five from Europe, and one from southern America. The quality of study scores ranged between 5 and 7 (longitudinal studies) and 6 and 9 (cross-sectional studies) (data not shown). The great majority of studies made diagnosis of HCV infection by detecting anti-HCV antibody in serum; one study assessed the presence of HCV antigen14 or HCV RNA24 in serum. Information on diagnosis of HCV was missed in one survey.23
Cross-sectional studies on the relationship between HCV and proteinuria (outcome: prevalence of proteinuria).
| Author (ref. number) [ref. year] | Country | Proteinuria, n | Age, yrs | Study size | Anti-HCV pos. pts, n |
|---|---|---|---|---|---|
| Szczech L, et al.10 [2002] | USA | 671/2057 (32%) | 38.5±7.7 | 2059 | 854 (41.5%) |
| Liangpunsakul S and Chalasani N11 [2005] | USA | 108/1357 (7.9%) | 47.6±19 | 15,336 | 368 (2.4%) |
| Tsui J, et al.12 [2006] | USA | NS | NA | 15,029 | 366 (2.4%) |
| Huang J, et al.13 [2006] | Taiwan | 707/9934 (7.1%) | 55.2±6 | 9934 | 642 (6.4%) |
| Ishizaka N, et al.14 [2008] | Japan | 1179/12,535 (9.4%) | 53±10.6 | 12,535 | 72 (0.6%) |
| Yanik E, et al.15 [2010] | USA | 76/902 (8.4%) | 49.5 (21; 80) | 902 | 772 (85.7%) |
| Derbala M, et al.16 [2010] | Qatar | 45/300 (15%) | 46 (41; 53) | 300 | 233 (77.6%) |
| Lee J, et al.17 [2010] | Taiwan | 2140/54,966 (3.9%) | 60.8±11.5 | 54,966 | 5189 (9.4%) |
| Yanagisawa N, et al.18 [2011] | Japan | 36/725 (4.9%) | 46.6±11.7 | 732 | 24 (3.3%) |
| Aoufi Rabih S, et al.19 [2012] | Spain | 38/225 (16.8%) | 55.6±16.1 | 265 | 120 (45.3%) |
| Reynes J, et al.20 [2013] | France | 104/1210 (8.5%) | 48 (41; 54) | 1210 | 220 (18.1%) |
| Zeng Q, et al.21 [2014] | China | 938/15,549 (6.0%) | 49.2±9.3 | 15,449 | 94 (0.06%) |
| Kurbanova N, et al.22 [2015] | USA | 4295/33,729 (12.7%) | 49.8±18.7 | 33,729 | 659 (1.9%) |
| Antonello V, et al.23 [2015] | Brazil | 133/666 (19.9%) | NA | 666 | 80 (12%) |
| Lucas G, et al.24 [2016] | USA | 43/292 (14.7%) | 49 (45; 54) | 292 | 115 (39.4%) |
| Zeder A, et al.25 [2016] | Germany | 60/393 (15.2%) | 45 (38; 53) | 393 | 46 (11.7%) |
Longitudinal studies on the relationship between HCV and proteinuria (outcome: incidence of proteinuria).
| Author (ref. number) [ref. year] | Country | Proteinuria, n | Study design | Study size | Anti-HCV pos. pts, n |
|---|---|---|---|---|---|
| Hestin D, et al.26 [1998] | France | 44/322 (13.7%) | Retrospective | 322 | 31 (9.6%) |
| Virgilio B, et al.27 [2001] | Italy | 22/187 (12%) | Retrospective | 187 | 51 (27.2%) |
| Ando M, et al.28 [2011] | Japan | 36/429 (8.3%) | Prospective | 429 | 9 (2.0%) |
| Banerjee T, et al.29 [2014] | USA | 7031/21,129 (33.3%) | Retrospective | 21,129 | 5069 (24%) |
| Lucas G, et al.24 [2016] | USA | 43/292 (14.7%) | Prospective | 292 | 115 (39.3%) |
| Palella F, et al.30 [2018] | USA | 1613/22,177 (7%) | Prospective | 2381 | 169 (7.0%) |
| Wong E, et al.31 [2021] | USA | 157/10,430 (1.5%) | Prospective | 10,430 | 112 (1.07%) |
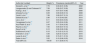
Sixteen studies (n=163,797 unique patients; n=9854 HCV-positive and n=153,943 HCV-negative patients) evaluated the prevalence of proteinuria among HCV-infected patients.10–25 The pooled prevalence of proteinuria was 0.11 (95% CI, 0.09–0.14) (Table 3). We found a significant relationship between positive HCV status and increased prevalence of proteinuria, aOR 1.47 (95% CI, 1.3; 1.66) (P<0.001). The funnel plot concerning the publication bias is reported in Fig. 2; the Egger test reported significant publication bias (P=0.003). Tests for homogeneity of the aOR across the studies gave a Q value (by χ2 test) of 27.3 (P=0.026). Fig. 3 reports the aOR and 95% confidence intervals for each cross-sectional study.
Prevalence of proteinuria (n=15 cross-sectional studies).
| Author [ref. number] | Weight, % | Prevalence (random)95% CI | Year |
|---|---|---|---|
| Szczech L, et al.10 | 7.70 | 0.33 (0.31; 0.35) | 2002 |
| Liangpunsakul S and Chalasani N11 | 7.89 | 0.08 (0.07; 0.09) | 2005 |
| Huang J, et al.13 | 8.09 | 0.07 (0.07; 0.08) | 2006 |
| Ishizaka N, et al.14 | 8.09 | 0.09 (0.09; 0.10) | 2008 |
| Yanik E, et al.15 | 7.89 | 0.08 (0.07; 0.10) | 2010 |
| Derbala M, et al.16 | 2.96 | 0.15 (0.04;0.26) | 2010 |
| Lee J, et al.17 | 8.11 | 0.04 (0.04; 0.04) | 2010 |
| Yanagisawa N, et al.18 | 8.07 | 0.05 (0.04; 0.06) | 2011 |
| Aoufi Rabih S, et al.19 | 2.43 | 0.17 (0.04; 0.30) | 2012 |
| Reynes J, et al.20 | 7.84 | 0.09 (0.07; 0.10) | 2013 |
| Zeng Q, et al.21 | 8.10 | 0.06 (0.06; 0.06) | 2014 |
| Kurbanova N, et al.22 | 8.10 | 0.13 (0.12; 0.13) | 2015 |
| Antonello V, et al.23 | 4.55 | 0.20 (0.13; 0.27) | 2015 |
| Lucas G, et al.24 | 6.66 | 0.15 (0.11; 0.19) | 2016 |
| Zeder A, et al.25 | 3.52 | 0.15 (0.06; 0.25) | 2016 |
| Total (95% CI) | 100.00 | 0.11 (0.09; 0.14) |
Test for heterogeneity: Chi2=3274, 62, df=14 (P<00001), I2=99.6%.
Test for overall effect: Z=9.29 (P<0.0001).
aOR and 95% confidence intervals for each study (n=16 cross-sectional studies; n=163,979 unique patients) (outcome: prevalence of proteinuria). aOR of proteinuria associated with exposure to HCV: 1.47 (95% CI, 1.3; 1.66) (P<0.001) Ri=0.61. Q value by χ2 test 27.3 (P=0.026) (asymptotic tests).
Seven longitudinal studies (n=35,170 unique patients; n=5556 anti-HCV positive and n=29,614 anti-HCV negative) gave information on the incidence of proteinuria. The pooled incidence of proteinuria was 0.14 (95% CI, 0.04; 0.23). The summary estimate of adjusted HR with HCV across the surveys, 1.79 (95%CI, 1.17; 2.74) (P<0.001).24,26–31 Significant publication bias occurred (P<0.04), according to the Egger test. Tests for homogeneity of the aHR across the studies (n=7) gave a Q value of 27.82 by χ2 test (P=0.0001).
Stratified analysis (and meta-regression)Stratified analyses were carried out and did not show consistent differences in pooled aOR across various subgroups (Tables 4 and 5) but the homogeneity assumption was rejected in numerous subsets (Tables 4 and 5). As listed in Table 6, we carried out multivariate meta-regression analysis and found a significant link between percentage of HCV-infected males and adjusted OR (cross-sectional studies). The limited number of studies with longitudinal design (n=7) and the incomplete information on many covariates precluded the conduction of meta-regression in this setting. We collected incomplete information on many clinical or biochemical parameters and this hampered the completion of many comparisons.
Summary estimates for adjusted OR of detectable proteinuria in HCV-exposed individuals (cross-sectional studies).
| Study, n | Fixed-effects aOR (95% CI) | Random-effects aOR (95% CI) | Ri | P-value (by the Q-test) | |
|---|---|---|---|---|---|
| All studies | 16 | 1.3 (1.23; 1.37) | 1.47 (1.3; 1.66) | 0.61 | 0.026 |
| Studies from USA | 6 | 1.3 (1.23; 1.38) | 1.44 (1.22; 1.71) | 0.75 | NS |
| Studies from Asia | 6 | 1.75 (1.38; 2.23) | 1.75 (1.38; 2.23) | 0 | NS |
| Studies from Europe | 3 | 1.7 (1.18; 2.44) | 1.82 (1.07; 3.1) | 0.51 | NS |
| Studies based on urine protein/creatinine ratio | 12 | 1.64 (1.41; 1.91) | 1.64 (1.41; 1.91) | 0 | NS |
| Studies with quality score ≥7 | 11 | 1.29 (1.22; 1.36) | 1.48 (1.27; 1.71) | 0.75 | 0.007 |
| Studies with quality score <7 | 5 | 1.52 (1.2: 1.91) | 1.52 (1.2: 1.91) | 0 | NS |
| Studies based on urine dipstick proteinuria | 3 | 1.26 (1.19; 1.33) | 1.27 (1.12; 1.45) | 0.78 | 0.06 |
Szczech L, et al. OR adjusted for age, serum creatinine, serum albumin, hepatitis B surface antigen, systolic/diastolic blood pressure, risk behaviour for HIV infection, and antiretroviral medication use
Liangpunsakul S and Chalasani N, et al. OR adjusted for age, race, arterial hypertension, gender, body mass index
Tsui J,et al. OR adjusted for age, gender, race/ethnicity, educational status, smoking status, diabetes and hypertension
Huang J, et al. OR adjusted for age, gender, body mass index, alanine aminotransferase, total cholesterol, tryglicerides, diabetes, hypertension, HBsAg status
Ishizaka N, et al. OR adjusted for age, gender, systolic blood pressure, and fasting plasma glucose
Yanik E, et al. OR adjusted for race, gender, age, work status, health insurance, injection drug use in prior 6 months, crack use in prior 6 months, HIV status, hepatitis B status, diabetes, blood pressure
Derbala M, et al. OR adjusted for age, gender, diabetes, cryoglobulinemia, serum urea, creatinine
Lee J, et al. OR adjusted for age, gender, educational status, body mass index, haemoglobin level, albumin level, cholesterol level, uric acid, hypertension, diabetes mellitus
Yanagisawa N, et al. OR adjusted for serum creatinine, diabetes, hypertension, cholesterol
Aoufi Rabih S, et al. OR adjusted for gender, age, hypertension, diabetes, obesity, rheumatologic comorbidities (rheumatoid arthritis, lupus erythematosus, etc.)
Reynes J, et al. OR adjusted for age, gender, diabetes, hypertension, eGFR, AIDS stage, current or previous tenofovir use, CD4 cell count
Zeng Q, et al. OR adjusted for age, gender, body mass index, albumin, hypertension, diabetes, tryglicerides, total and low-density lipoprotein cholesterol
Kurbanova N, et al. OR adjusted for age, gender, race, hypertension, diabetes mellitus, and body mass index
Antonello V, et al. OR adjusted for age, alcohol abuse, smoke, diabetes, hypertension, HBV, CD4 cells, tenofovir current use, HIV RNA load
Lucas G, et al. OR adjusted for cocaine use, HIV status, cardiovascular risk score
Zeder A, et al. OR adjusted for age, CD4 nadir, exposure to TDF, diabetes mellitus, arterial hypertension, baseline eGFR, ethnicity, current use of atazanavir (ATV), body mass index
Summary estimates for adjusted HR of detectable proteinuria in HCV-exposed individuals (longitudinal studies).
| Study, n | Fixed-effects aOR (95% CI) | Random-effects aOR (95% CI) | Ri | P-value (by the Q-test) | |
|---|---|---|---|---|---|
| All studies | 7 | 1.24 (1.18; 1.30) | 1.86 (1.27; 2.73) | 0.98 | 0.0000 |
| Population-based studies | 3 | 1.22 (1.16; 1.28) | 1.37 (0.9–2.09) | 0.98 | 0.034 |
| Single-centre studies | 4 | 2.06 (1.57; 2.71) | 2.67 (1.39; 5.14) | 0.80 | 0.0086 |
| Prospective studies | 4 | 1.74 (1.27; 2.38) | 1.79 (0.99; 3.21) | 0.83 | 0.039 |
| Studies from US | 4 | 1.22 (1.16; 1.28) | 1.37 (1.01; 1.87) | 0.96 | NS |
| HIV studies | 3 | 1.21 (1.16; 1.27) | 1.63 (0.89; 2.98) | 0.99 | 0.033 |
| Quality score≥7 | 6 | 1.24 (1.18; 1.3) | 2.06 (1.35; 3.15) | 0.98 | 0.0001 |
| Studies based on urine protein/creatinine ratio | 4 | 1.74 (1.27; 2.38) | 1.79 (0.99; 3.21) | 0.68 | 0.039 |
Hestin D, et al. HR adjusted for age, time on dialysis, first/second graft, HLA mismatch number.
Virgilio B, et al. HR adjusted for age, gender, time on dialysis, ALG/ATG use, hypertension, primary nephropathy, acute rejections (first year), creatinine clearance (year 5).
Ando M, et al. HR adjusted for age, gender, body mass index, smoke, hypertension, diabetes, HIV RNA level, CD4 cell count, eGFR, haemoglobin, serum cholesterol and tryglicerides, and use of TDF-boosted protease inhibitors.
Banerjee T, et al. HR adjusted for age, gender, race, diabetes, hypertension, cardiovascular disease, dyslipidemia, eGFR, illicit drug use, CD4 cell count, HIV RNA, HBV status, body mass index, low density/high density lipoprotein, total cholesterol and serum glucose.
Lucas G, et al. HR adjusted for cocaine use, HIV status, cardiovascular risk score.
Palella F, et al. HR adjusted for baseline eGFR, race, gender, diabetes, hypertension, smoking status, HBV infection, CD4 cell count, HIV RNA, history of clinical AIDS diagnosis.
Wong E, et al. HR adjusted for age, centre, gender, education status, Caribbean origin, comorbidities (diabetes, hypertension, alcohol, smoke, body mass index).
Multivariate meta-regression analysis (random effects model): impact of continuous variables on proteinuria (prevalence) (n=16 cross-sectional studies).
| Covariate | Regression coefficient | Standard error | 95% CI | Z-value | P-value |
|---|---|---|---|---|---|
| Intercept | 35,740 | 3829 | −39.3; 111.8 | 0.93 | 0.35 |
| Reference year | −0.0177 | 0.019 | −0.05; 0.01 | −0.92 | 0.35 |
| Study size | −0.0001 | 0.000 | −0.00; 0.000 | −1.72 | 0.08 |
| Male | 0.0071 | 0.0033 | 0.00; 0.013 | 2.13 | 0.033 |
| HCV positive status | −0.0019 | 0.0030 | −0.007; 0.003 | −0.64 | 0.52 |
Test of the model: simultaneous test that all coefficients are 0 (excluding intercept): Q=18.28, df=4, P=0.001.
Goodness of fit: test that unexplained variance is 0: Tau2=0.000, Tau=0.000, I2=0.00%, Q=9.13, P=0.60, df=11.
We recorded studies (n=6; n=72,551 unique patients) addressing the occurrence of proteinuria according to HCV status (no adjustment for covariates made).32–37 There was an increased frequency of proteinuria among HCV-exposed individuals and this reached statistical significance, pooled OR 1.54 (95% CI, 1.08; 2.19). The homogeneity assumption was not rejected, Ri=0.88, Q value of 36.1 by χ2 test (P=0.001).
DiscussionThe occurrence of CKD in the world is rising and gives additional burden on both the healthcare and economy in the years to come. Previous studies have reported that patients with CKD are at a higher risk of progression to end-stage kidney disease (ESRD) which needs expensive renal replacement programmes. Progression to kidney failure and other adverse outcomes can be prevented or at least delayed by early detection and treatment – proteinuria and glycosuria are early biochemical markers for kidney disease which can be used as a proxy measure to give clue to clinicians to make diagnosis on the occurrence of kidney disease at an early stage. Among the various predictors of progression of CKD to ESRD, proteinuria appears to be an important one.
The findings of the current meta-analysis reinforce the hypothesis that HCV infection is an important risk factor for the development of proteinuria in the adult general population. This meta-analysis (n=22 studies; n=198,967 unique patients) includes a number of studies greater than that reported in the previous one (n=10) and confirms the higher risk of proteinuria among HCV-infected individuals, overall estimate for adjusted OR with HCV across the cross-sectional surveys, 1.47 (95%CI, 1.3; 1.66) (P<0.001). Of note, we noted in the current meta-analysis small between-study heterogeneity, particularly among cross-sectional studies (Q value by Chi-squared [χ2] test 27.3, P=0.02) and stratified analysis reported no heterogeneity in several comparisons (Tables 4 and 5). The results obtained with univariate analyses were in keeping with those obtained with multivariate regressions.
HCV has been implicated in derangements of multiple organ systems including cardiovascular, muscular, nervous, ocular, respiratory, skeletal, cutaneous, and urinary systems. Lymphoproliferative disorders such as lymphoma and mixed cryoglobulinemia are associated with HCV infection. Moreover, HCV has a deleterious impact on psychosocial status. Kidney involvement in HCV-infected patients was first observed more than 2 decades ago38; but the relationship between HCV and CKD (reduced GFR or detectable proteinuria) in the adult general population had remained controversial until a few years ago. The KDIGO HCV Work Study Group1 recommended the screening for proteinuria in HCV-infected individuals and the findings obtained in this study reinforce such a recommendation.
The relationship between detectable proteinuria and HCV infection has been explained with various underlying mechanisms; HCV can give proteinuria by immunological and non-immunological pathways (i.e., oxidative stress, pro-inflammatory cytokines, and others). The pathogenesis of glomerular disease associated with HCV infection involves immune-mediated damage (including the activity from cryoglobulins); HCV-associated membranous nephropathy is thought to be related to the deposition of immune complexes containing HCV proteins in the glomerular basement membrane.1 Direct effects of virus on renal tissue have been cited; as an example, it has been mentioned the binding of HCV to CD81 and SR-B1 receptors in order to penetrate into the renal parenchymal cells.39 HCV RNA has been recorded in mesangial cells, tubular epithelial cells, and endothelial cells (glomerular and tubular capillaries).40 Viral HCV antigens have been demonstrated by immunohistochemistry, in situ hybridization, immuno-gold labelling, and laser capture microdissection.40,41 On the other hand, HCV-infected patients are at risk for kidney damage due to hereditariness, cirrhosis, illicit drug use, and co-infection (HBV and/or HIV). It has been recently mentioned that an accelerated atherosclerosis also contributes to the increased risk of proteinuria among patents with HCV.4
The findings from the current meta-analysis show several limitations. First, the majority of reports have retrospective and cohort design and it is well known that studies with this design cannot give excellent evidence. Secondly, the current availability of safe and effective drugs for the treatment of HCV clearly hampers the conduction of longitudinal studies on the incidence of proteinuria among HCV-infected patients. Thirdly, each study retrieved in the current meta-analysis was performed with multivariate analysis in order to evaluate the independent and significant impact of clinical or demographic parameters (confounders) on the occurrence of proteinuria; residual confounding cannot be excluded as complete information on various confounders was missing. Finally, the link between HCV infection and proteinuria was less evident in longitudinal studies; the discrepancy between cross-sectional and longitudinal studies could be explained by the need of longer follow-ups.
The results of the current meta-analysis support the notion that a strong link between detectable proteinuria and HCV positive status exists. The current availability of effective and safe drugs (DAAs) for the early treatment of HCV gives us the opportunity to improve the management of CKD in the adult general population. We suggest screening all patients for proteinuria at the time of HCV infection diagnosis.
Institutional review board statementNot applicable.
Informed consent statementNot applicable.
Data availability statementNot applicable.
FundingNo sources of funding were used for the preparation of this manuscript.
Conflict of interestThe authors declare no conflict of interest.