Acute interstitial nephritis (AIN) which is characterized as inflammation and oedema around renal tubuli mostly due to hypersensitivity to drugs, has been reported to be underlying aetiology among 5–27% of acute kidney injury cases in hospitalized patients.1 Chronic idiopathic urticaria (CIU), is an intrinsic immunological abnormality independent of external causes without a triggering stimulus like stress, physical factor, drug and food. It usually affects females with a prevalence of 1% and mostly begins at the age of 30–50 years.2 Diagnosis mostly depend on chronic recurrent itching persisting at least six weeks with exclusion of diseases like infections, malignities, auto-immune and auto-inflammatory diseases.2,3 Treatment is needed for cumbersome symptom of pruritis and life-threatening medical emergency of angioedema. Omalizumab, which is recombinant humanized monoclonal antibody for immunoglobulin (Ig) E is advised for treatment of CIU as the third or fourth line of therapy after histamine receptor type 1 (H1) antihistamines and leukotriene receptor antagonists are unsuccessful.3,4
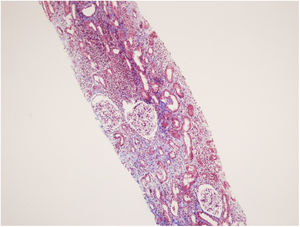
A 53-year-old female patient with diabetes mellitus, hysterectomy and CIU had symptoms of nausea and fatigue. In her medical history, she has been taking bilastine (20mg/day) for 1 year, methylprednisolone was prescribed to the patient due to angioedema which affected tongue and larynx for 30 days until 5 months ago, diclofenac was given for pain relief after hysterectomy, she had taken dexketoprofen (25mg/every 3 day) for 3 months until 2 months ago and she was injected the fifth monthly dose of 150mg omalizumab 15 days ago. Laboratory evaluation revealed increased serum creatinine level (1.64mg/dL) with respect to basal levels (0.65mg/dL) of 6 months earlier, decreased glomerular filtration rate (GFR) (estimated GFR using Chronic Kidney Disease Epidemiology Collaboration, CKD-EPI equation was 34mL/min/1.73m2, creatinine clearance using 24-h urine collection was 37.4mL/min), microalbuminuria (98mg/d), increased proteinuria (1.3g/d), and increased beta-2 microglobulinuria (21,300μg/L, normal value <300μg/L5). Her urinary beta-2 microglobulin: creatinine ratio was 76071 (μg/g) (normal<300μg/g6). Biochemically her serum creatinine level was increased to 2.21mg/dL within 8 days (Table 1). Presence of high amount of low molecular weight proteins (beta-2 microglobulin) in her urine analysis indicated tubular type consisting majority of proteinuria. Kidney biopsy showed acute interstitial nephritis accompanied with chronic interstitial injury related findings without immune deposition (Fig. 1). In addition, early stage of diabetic nephropathy was observed due to diffuse basal membrane thickening. Oral methylprednisolone was given to the patient at the dose of 1mg/kg/d for treatment of AIN.7 Dose was slowly tapered to 5mg/day after a month and prescribed for another 3 months. Her serum creatinine level was decreased from 2.24mg/dL to 1.58mg/dL in 10 days of treatment, and after 4 months her serum creatinine level was 0.82mg/dL.
Clinical and laboratory findings of the patient.
| Parameter | Patient |
|---|---|
| Age (years) | 53 |
| Gender | Female |
| Complain | Nausea |
| Blood pressure (mmHg) | 120/80 |
| Serum creatinine level (mg/dL)6 months earlier/at admission/during AKI/last visit | 0.65/1.64/2.21/0.82 |
| Proteinuria level (g/d)At admission | 1.3 |
| Albuminuria (mg/d)At admission | 98 |
| Beta-2 microglobulinuria level (μg/L)At admission | 21,300 |
| Past drug history for the last 6 months | Methylprednisolone, diclofenac, dexketoprofen, and omalizumab |
| Serum ANA, anti dsDNA, and ANCA test results | Negative |
| Present drug history | Gliclazide, metformin, bilastine, and omalizumab |
| Diagnosis based on | Clinical and histopathological data |
| Comorbid diseases | Diabetes mellitus, chronic idiopathic urticaria |
| Renal pathology | Acute interstitial nephritis accompanied with chronic interstitial injury and early stage of diabetic nephropathy |
The cause of AIN in majority of the patients was known as hypersensitivity towards drugs (75% of the cases).8 Practically, every drug may have a potential for causing AIN, yet mostly reported drugs are antibiotics, NSAID, proton pump inhibitors and allupurinol.9 The time interval between drug intake and onset of AIN can vary from 1 week to 9 months (10–11 weeks being the commonest) among the patients.9 The most possible offending agent for AIN of the patient was thought as diclofenac since absence of any report regarding the other medications that our patient had taken (methylprednisolone, dexketoprofen, gliclazide, metformin, bilastine, and omalizumab). It is known that NSAID induced AIN does not generally response to glucocorticoid therapy.7 Conflicting with this information, our patient responded well to corticosteroid treatment which made us suspect some other drug she used might have led to AIN other than NSAID. The association of CIU with AIN has not been reported yet. It might be possible but needs to be clarified that AIN of our patient may result from one of the hypersensitivity reaction and manifestation of CIU. However, her itching symptoms completely abolished by omalizumab and this AIN developed during omalizumab treatment of 5 months duration. Omalizumab has been shown to decrease serum eosinophil levels and effective in treatment of CIU through its functions against eosinophils.10 In theory, omalizumab might have been protective for development of AIN via these eosinophil depleting effects. However, even though 5 months of omalizumab treatment, our patient had AIN which was treated successfully with methylprednisolone.
In conclusion, AIN of our case most probably was secondary to diclofenac intake 5 months earlier even though presence of response to glucocorticoids. Whether presence of CIU increase the tendency of patients to development of AIN secondary to offending drugs has not been known yet. Renal functions especially tubular proteinuria should be evaluated and searched thoroughly in order to diagnose early and treat appropriately for AIN among individuals with CIU before and during prescription of any drug.
Authors’ contributionsKubra Kaynar: Conception, design, drafting the article.
Nejla Küçük: Drafting the article.
Sevdegül Mungan: Drafting the pathological data of the case.
Beyhan Güvercin: Drafting the article.
Şükrü Ulusoy: Drafting the article.
Conflicts of interestNone declared.