Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has negatively impacted on patients of the whole CKD spectrum, causing high rates of morbi-mortality. SARS-CoV-2 vaccines opened a new era, but patients with CKD (including kidney transplant, hemodialysis and peritoneal dialysis) were systematically excluded from pivotal clinical trials. The Spanish Society of Nephrology promoted the multicentric national SENCOVAC study aimed at assessing immunological responses after vaccination in patients with CKD. During the first year after vaccination, patients with non-dialysis CKD and those on hemodialysis and peritoneal dialysis presented good anti-Spike antibody responses to vaccination, especially after receiving the third and fourth doses. However, kidney transplant recipients presented suboptimal responses after any vaccination schedule (initial, third and fourth dose). Especially worrisome is the situation of a patients with a persistently negative humoral response that do not seroconvert after boosters. In this regard, monoclonal antibodies targeting SARS-CoV-2 have been approved for high-risk patients, although they may become obsolete as the viral genome evolves. The present report reviews the current status of SARS-CoV-2 vaccination in the CKD spectrum with emphasis on lessons learned from the SENCOVAC study. Predictors of humoral response, including vaccination schedules and types of vaccines, as well as the integration of vaccines, monoclonal antibodies and antiviral agents are discussed.
Síndrome agudo respiratorio severo coronavirus 2 (SARS-CoV-2) ha impactado negativamente en todos los pacientes con enfermedad renal crónica (ERC), causando elevadas tasas de morbimortalidad. La vacunación frente a SARS-CoV-2 han abierto una nueva era, aunque precisamente los pacientes con ERC (incluyendo los portadores de un injerto renal y aquellos en programas de hemodiálisis y diálisis peritoneal) han sido sistemáticamente excluidos de los ensayos clínicos. La Sociedad Española de Nefrología (S.E.N.) promovió el estudio multicéntrico SENCOVAC para evaluar la respuesta inmunológica tras la vacunación en todo el espectro de la ERC. Un año después de haber recibido la pauta inicial de vacunación, los pacientes con ERC sin necesidad de diálisis, y aquellos en hemodiálisis y diálisis peritoneal, han presenado una adecuada respuesta humoral (monitorizada con el desarrollo de anticuerpos frente a la proteína Spike), especialmente después de recibir la tercera y la cuarta dosis. Sin embargo, los portadores de un injerto renal han presentado una constante respuesta subóptima en cualquier momento de la vacunación (dosis inicial, tercera y cuarta dosis). Especialmente preocupante es la situación de los pacientes con respuesta humoral persistentemente negativa que no seroconvierten incluso ni tras recibir las dosis de recuerdo o boosters. En ese contexto, el manejo probablemente pasa por el uso de anticuerpos monoclonales dirigidos frente a SARS-CoV-2 que han sido recientemente aprobados, asumiendo que pueden perder efectividad con el cambio del genoma viral. La presente revision tiene por objeto resumir y analizar la situación actual de la vacunación frente a SARS-CoV-2 en el espectro de la ERC enfatizando en los resultados del estudio SENCOVAC. Asimismo, a lo largo de la revisión se discuten los predictores de la respuesta a las diferentes dosis y tipos de vacunas y la integración de estas con los anticuerpos monoclonales y los agentes antivirales.
Chronic kidney disease (CKD) is one of the fastest growing causes of death, projected to become the fifth global cause of death by 2040 and the second cause of death in countries with long life expectancy before the end of the century.1,2 While the increased risk of cardiovascular disease has received most attention,3 persons with CKD are at increased risk of death from non-cardiovascular causes and indeed the absolute increase in the risk of death from non-cardiovascular causes, including infection, is larger than for cardiovascular causes, especially in the elderly.4
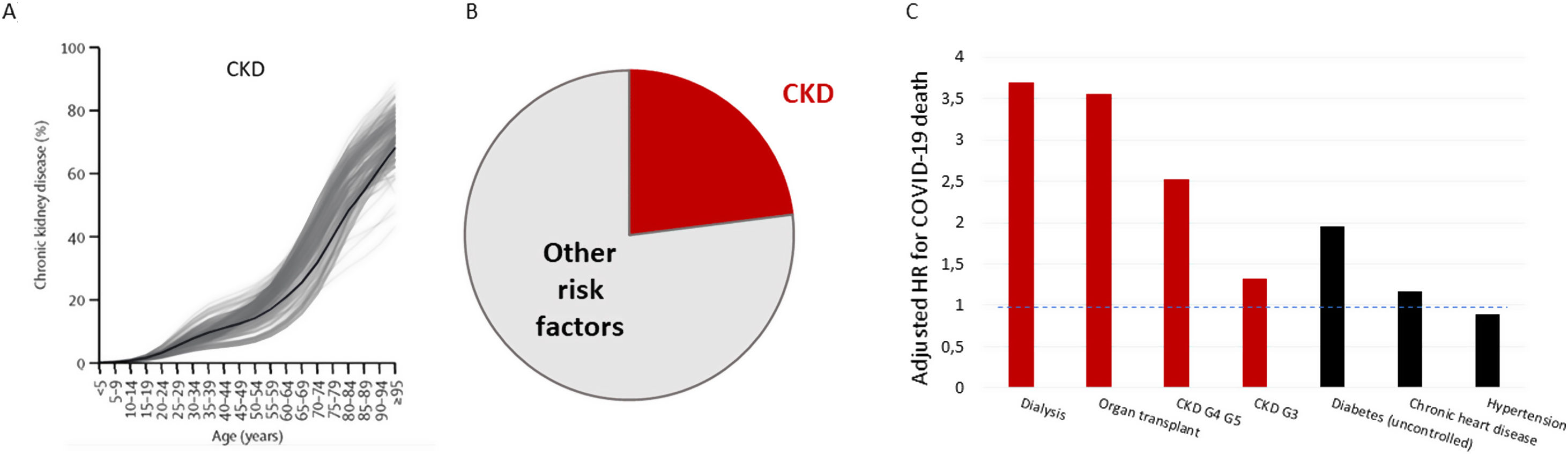
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causes Coronavirus disease 2019 (COVID-19), a condition of variable severity that may result in death. Initial reports of COVID-19 indicated that 20% of hospitalized patients had diabetes mellitus (DM), 15% hypertension and 15% had cardiovascular disease (CVD), but CKD was not mentioned5 and was not considered a risk factor for many months. It is now clearly established that CKD is the main risk factor for severe COVID-19.6 According to the Global Burden of Disease (GBD) collaboration, CKD is the most prevalent risk factor for severe COVID-19 worldwide7 (Fig. 1A). Additionally, CKD is the coexistent condition that most increased the risk of COVID-19 death.8 OpenSAFELY analyzed data from 17,278,392 adults and 10,926 COVID19-related deaths in England. In a fully adjusted model, patients cared for by nephrologists, such as dialysis patients (Adj HR 3.69), transplant recipients (Adj HR 3.55) and CKD (Adj HR 2.52 for patients with eGFR<30ml/min/1.73m2, GFR category G4-5) patients were among the top 4 risk categories (Fig. 1B).8 However, within the dialysis/transplant populations (kidney replacement therapy, KRT), the additional risk associated to classical risk factors differs from the general population. Thus, male sex or coexistent diabetes, hypertension, coronary artery disease or lung disease do not further increase the risk of COVID-19 death,9 contrary to observations in persons not on KRT.8 Furthermore, the adverse impact of older age is smaller in persons on KRT: older KRT patients have a ∼4-fold increase in risk vs those in their fifties, as opposed to a ∼15-fold increase in the general population.6 In the early pandemic waves in the most affected countries or cities, such as Madrid, a 30% mortality was reported in dialysis patients.10 This soon decreased to 13–16%, as it became apparent that, as for other populations, there was also a high prevalence of asymptomatic COVID-19 among hemodialysis (HD) patients.11,12 Despite this, mortality remained above that of the general population and had a dramatic impact on the epidemiology of KRT. As an example, in Madrid in 2020, KRT incidence decreased 12% versus 2019 while KRT prevalence decreased (−1.75%) for the first time since records began. COVID-19 was the most common cause of death in 2020 (33% of all deaths) and singlehandedly increased the annual mortality of KRT patients to 10.2% (34% higher than the mean for 2008–2019), more so in kidney transplant recipients (+68%). However, the bulk of COVID-19 deaths (209/285, 73%) occurred during the first COVID-19 wave, which roughly accounted for all the excess mortality of KRT patients in 2020. This temporal pattern of COVID-19 mortality, despite further larger COVID-19 waves observed before vaccines became available, suggests that appropriate healthcare may improve COVID-19 outcomes in KRT patients.13,14
Global prevalence of CKD and impact of CKD on COVID-19 mortality. (A) Global prevalence of CKD by age according to Global Burden of Disease (GBD) data. Gray lines represent individual countries. (B) According to these data, CKD accounts for the increased risk of severe COVID-19 in 5.1% of the global population, or 23% of the global population at increased risk of severe COVID-19, as represented in panel B, being the highest contributor to global risk. Data for (A) and (B) from Ref. 7. (C) Risk of COVID-19 death for selected preexistent conditions from Ref 8. Dialysis and transplantation were the conditions that most increased the risk of death from COVID-19 in the OpenSAFELY study. Additionally, the increased risk conveyed by CKD G4/G5 was still higher than that conveyed by several of the often-cited risk factors for severe CVOD-19, including diabetes, chronic heart disease and hypertension. Even CKD G3 conveyed a higher risk of COVID-19 death than heart disease or hypertension. The discontinuous horizontal blue line indicates neutral risk (Adj HR=1.0).
Several factors may account for the high impact of COVID-19 on KRT patients, especially during the first wave. Some of them may have been transient, thus contributing to the severe impact of the first COVID-19 wave while others may be permanent, accounting for a persistent increased risk of severe COVID-19. Thus, HD patients were unable to shield, as they had to travel to dialysis facilities thrice weekly. In Spain this entailed shared vehicles with other patients at a time when health authorities were actively suppressing masking.15,16 In this regard, shared transport emerged as a key risk factor for COVID-19.17 Moreover, at a time of severe constraints in access to ICU care, KRT patients may have been considered to have too low chances of survival, in a self-fulfilling prophecy. Finally, early therapy for COVID-19 was not evidence-based, and some of the useless medication combinations used may have increased the risk of fatal arrhythmia associated with long QT in dialysis patients.18 Among more permanent factors, we find the spontaneous immune suppression of uremia, the immunosuppressants prescribed to kidney transplant (KT) recipients and the high prevalence of frailty among KRT patients.
SENCOVAC study: rationale and designThe development of SARS-CoV-2 vaccines opened a new era in the COVID-19 pandemic. Phase III pivotal studies for assessing safety and efficacy of the different vaccines demonstrated protection against severe COVID-19.19–22 Indeed, vaccination was associated to lower rates of admissions and complications derived from SARS-CoV-2 infections, including mortality, for at least 5 months after the initial schedule.23,24 However, and beyond the pivotal clinical trials, a very low number of patients included in prospective studies presented CKD at baseline.25
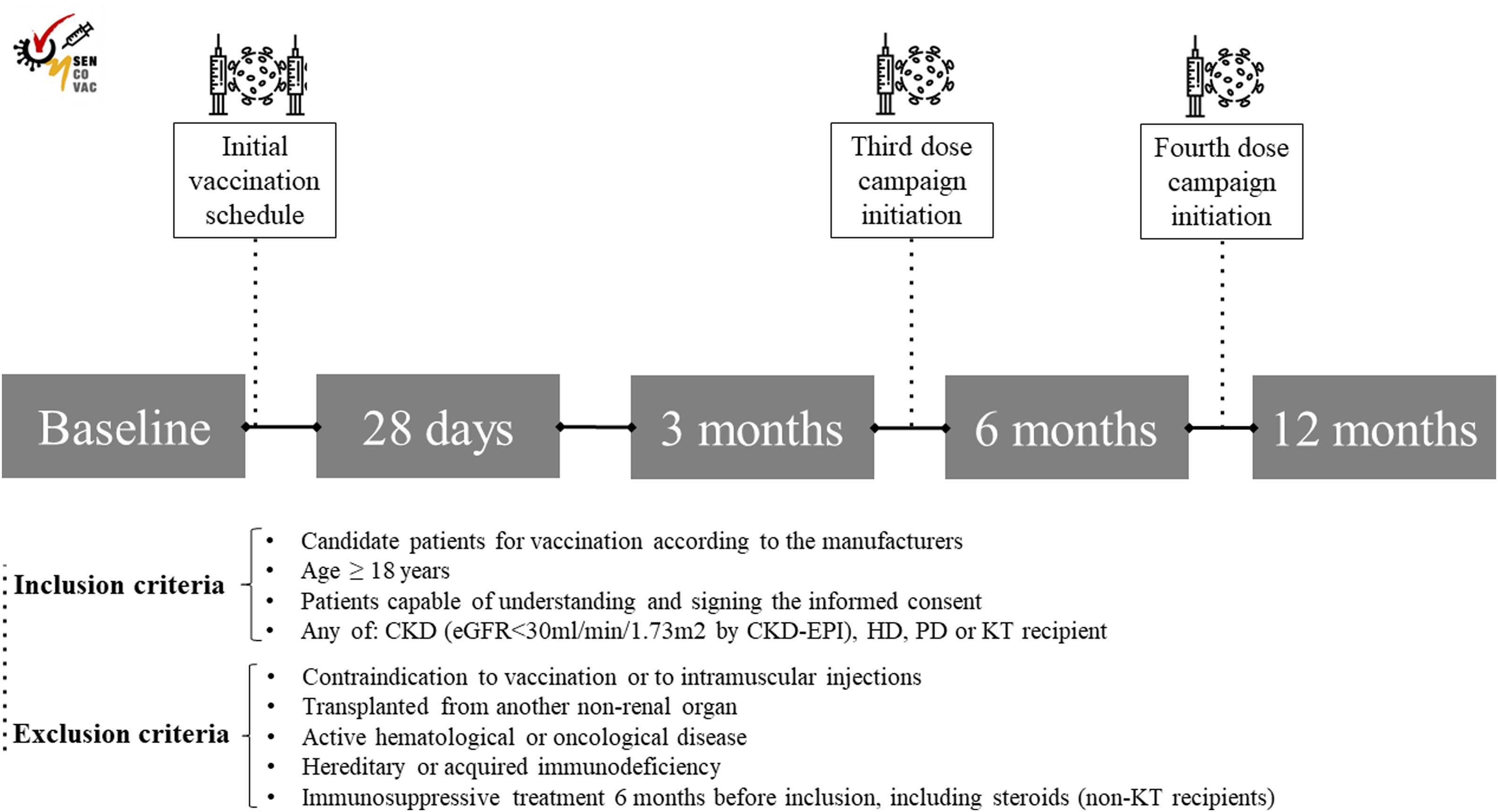
With this background, we designed the SENCOVAC study with the aim of evaluating humoral response to SARS-CoV-2 vaccines in the CKD spectrum (i.e. patients with advanced CKD [eGFR<30ml/min/1.73m2, peritoneal dialysis [PD], HD and KT recipients).26 This real-world study had a prospective and observational design and included more than fifty centers in Spain. Inclusion and exclusion criteria are summarized in Fig. 2. At baseline, investigators collected epidemiological data, comorbidities, long-term treatments, and data specific for each cohort (Kt/Vurea, dialysis vintage, technique and vascular access for HD and PD patients; immunosuppressive therapy for KT; eGFR for ND-CKD and KT recipients).
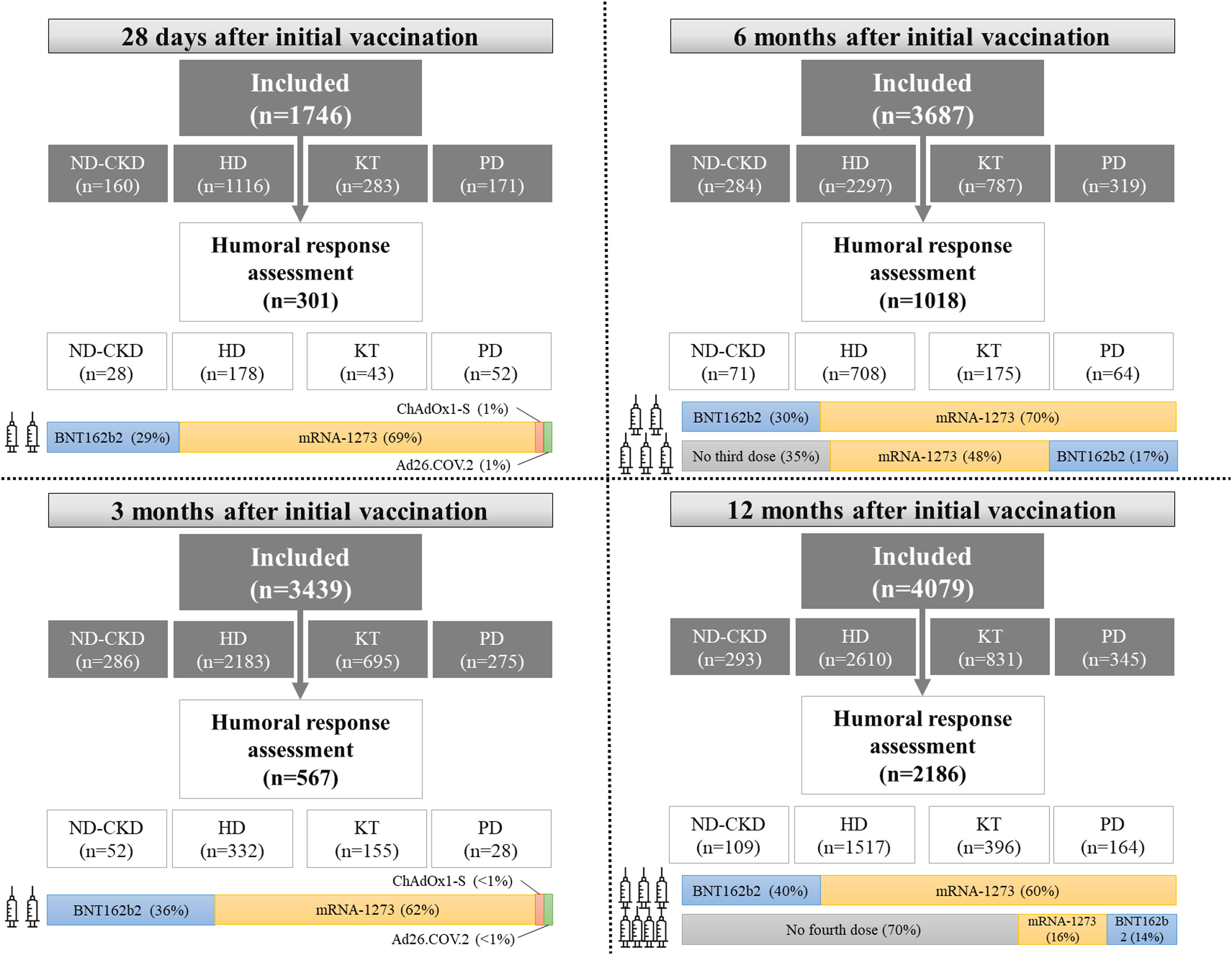
Following the study protocol, at baseline, 28 days, 3 months, 6 months, and 12 months after completing the initial vaccine schedule, serum samples were obtained and sent to a central laboratory to determine anti-Spike antibodies (Fig. 3). All samples were tested by a CE-marked commercial method, a quantitative chemiluminescence immunoassay (CLIA, Covid-19 Spike Quantitative Virclia® IgG Monotest, Vircell SL, Spain), with a sensitivity and specificity of 96% and 100% respectively that detects IgG antibodies against the SARS-CoV-2 Spike protein. This assay was calibrated against the First WHO International Standard for anti-SARS-CoV-2 human immunoglobulin (NIBSC code: 20/136) and results were expressed as IU/ml. According to the performance studies of the manufacturer, based on the analysis of prepandemic serum samples, values ≤32IU/ml were considered as negative, between 32 and 36IU/ml as equivocal and values >36IU/ml as positive, reflecting the presence of anti-Spike IgG antibodies as a consequence of either previous infection or vaccination. Investigators included an addendum to perform an interim analysis at 9 months only in patients on HD.27
During the study, some patients received a third and a fourth dose of vaccine. Although initial vaccination was based on BNT162b2 (Pfizer-BioNTech®), mRNA-1273 (Moderna®), ChAdOx1-S (AstraZeneca®) or Ad26.COV.2 (Janssen®) vaccines, subsequent boosters consisted of mRNA-based vaccines (BNT162b2 and mRNA-1273) (Fig. 3). Vaccination schedules were determined by the local public health authorities and investigators did not intervene in vaccine prescription.
The primary objective of the study was to determine the humoral response after SARS-CoV-2 vaccination (initial and boosters) in patients with advance CKD (GFR<30ml/min/1.73m2, HD, DP and KT recipients). Secondary objectives included safety, quantification of anti-Spike antibodies after vaccination, predictors of humoral responses and establishment of cut-off anti-Spike antibody titers for protection against severe breakthrough infections.
Safety and humoral response after SARS-CoV-2 vaccinationThe safety and humoral response after SARS-CoV-2 vaccination were evaluated after the initial vaccination schedule, after the third dose and after the fourth dose. Since the timing of sampling was pre-specified, at each time point patients that had received boosters could be compared with those who had not yet received the booster.
Initial vaccination schedule responseSARS-CoV-2 vaccination in patients of the CKD spectrum was safe, with a high incidence of adverse events (>50%) but all of them not serious (i.e., local pain, general discomfort, asthenia). The second dose (in patients with a two-dose scheme, i.e., BNT162b2, mRNA-1273, ChAdOx1-S) generated more adverse events than the first one. Factors associated to local and systemic reactions included previous COVID-19 (before vaccination), being younger or a KT recipient.26
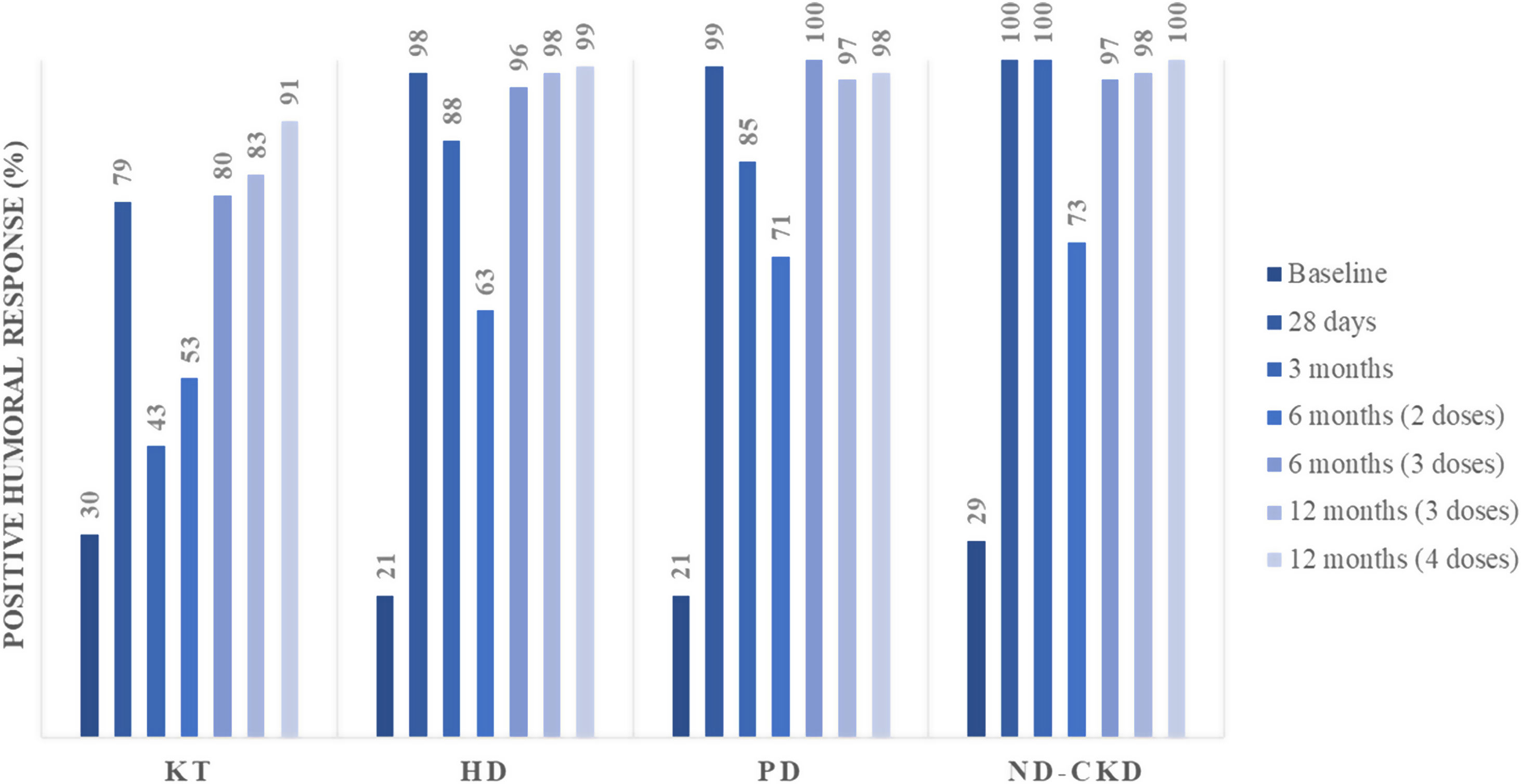
After completing the initial vaccination schedule, patients with CKD presented disparities in their humoral response. The first interim analysis of SENCOVAC included 1746 patients and analyzed humoral response 28 days after vaccination in 301 participants (28 non-dialysis CKD patients, 43 KT recipients, 52 PD and 178 HD patients).26 Anti-Spike antibodies were positive in 95% of the patients (Fig. 4). However, a negative humoral response was detected in 21% of the KT recipients, showing that this population developed suboptimal responses and should be considered at very high-risk of SARS-CoV-2 infections and of complications due to SARS-CoV-2.26 Beyond the risk of KT recipients for a suboptimal humoral response, the type of vaccine was also an independent predictor for humoral response. Specifically, the BNT162b2 (30μcg) vaccine was associated to 6-fold risk for negative humoral response than mRNA-1273 (100μcg), probably due to the different amount of vaccine in each dose.
Positive humoral response in each assessment of the SENCOVAC study across all the categories of the CKD spectrum from ref 26,27,38,42,55, 59. Positive immune response was defined as anti-Spike antibody titers above 36IU/ml as positive, while ≤32IU/ml were considered as negative, and between 32 and 36IU/ml as equivocal, based on the manufacturer evaluation of serum from exposed and non-exposed individuals. Abbreviations: ND-CKD: non-dialysis chronic kidney disease, HD: hemodialysis, PD: peritoneal dialysis, KT: kidney transplant.
These results agree with other studies recently summarized by Ma BM et al. in a meta-analysis.28 Beyond the lower rates of positive humoral response after initial vaccination of KT recipients compared to other patients with CKD, all the CKD spectrum developed suboptimal humoral responses when compared to healthy controls.28,29 The inherent immunosuppressive state of patients with CKD and specific features of dialysis and KT (immunosuppression, comorbidities) could explain why these cohorts are not fully protected after vaccination.30
The majority of published studies assessing the humoral response to SARS-CoV-2 vaccines in CKD have been based on mRNA vaccines.31 Small reports on vaccines based on other technologies (such as inactivated virus or viral vectors) demonstrated a minimal disadvantage of these vaccines compared to mRNA-based vaccines in the immediate seroconversion in CKD (especially in KT) patients.32–34 On the other hand, and although heterologous regimens (i.e., the use of different types of vaccines) as initial regimens had been poorly explored, at least one study showed their efficacy in enhancing humoral responses over homologous regimens in HD.35
Third dose responseThe suboptimal immunization after the initial vaccination schedule was of special importance in vulnerable populations, including patients with CKD. Loss of acquired humoral response was a key point during the pandemic as anti-Spike antibody titers correlated to efficacy.36 In the CKD spectrum, 25% of patients had fully lost their immunity at 3 months with the majority of them presenting significant declines in anti-Spike antibody titers.37 Within the CKD spectrum, KT recipients achieved the higher rates of lost humoral responses, with more than 50% of the fully vaccinated patients having undetectable antibodies (Fig. 4).38 This waning dynamic was more pronounced at 6 months and affected all the CKD spectrum. This situation opened a new scenario: when to administer a new dose of SARS-CoV-2 vaccine.39,40 However, not all patients shared the same temporal pattern of the humoral immune response. Some factors predicted the loss of anti-Spike antibodies such as being a KT recipient, previous lower anti-Spike antibody titers (due to suboptimal response to vaccination or lack of prior SARS-CoV-2 infection) or the type of vaccine.38,39,41 With this background, Spanish health authorities (and other governments worldwide) decided to administrate an early booster dose of mRNA-vaccines, prioritizing vulnerable cohorts (such as CKD patients) and irrespective of the humoral situation.
The 6-month assessment of SENCOVAC included patients who had received 2 or 3 SARS-CoV-2 vaccine doses and allowed to evaluate the response to the third dose in CKD patients42 (Fig. 3). The administration of mRNA-based third doses seroconverted 69% of the previous negative patients. By groups, receiving the third dose was associated to higher proportion of positive humoral response (in comparison to 2 doses) in KT recipients (80% vs. 53%), patients on PD (100% vs. 71%), patients on HD (96% vs. 64%) and patients with ND-CKD (97% vs. 73%) (Fig. 4).42 According to these results and in agreement to other published studies, KT recipients remained as the low-protected cohort even after three vaccine doses.43 In addition to the impaired immunity of KT recipients, the emergence of new SARS-CoV-2 variants may have influenced the capacity of vaccines to elicit anti-SARS-CoV-2 neutralizing antibodies. Al Jurdi et al. conducted a small but elegant study in KT recipients that compared the neutralizing effect across different SARS-CoV-2 variants. They concluded that the third dose of SARS-CoV-2 mRNA vaccines presented lower effectivity against new SARS-CoV-2 variants (i.e., Omicron) than against the original virus.44
In contrast to the results in KT recipients, a robust humoral response was developed by the vast majority (more than 95%) of non-KT recipients that had received the booster.42,45 Although patients with CKD usually received mRNA-based vaccines, some studies explored heterologous regimens with controversial results. Despite the use of mixed vaccine types, altogether studies revealed a humoral benefit with heterologous regimens46–48 constituting an alternative for patients at high risk for low humoral response. A recently published clinical trial assessed the efficacy of a heterologous regimen (ChAdOx1 nCoV-19) vs an mRNA booster (BNT162b2 or mRNA-1273) in immunosuppressed patients who failed to respond to the initial schedule. This study demonstrated the beneficial effect of a homologous mRNA-based regimen.49 By contrast, a randomized clinical trial involving KT recipients with negative humoral response after initial vaccination, demonstrated non-inferiority of a mRNA-based third dose vs a vector-based heterologous vaccination strategy.50
Fourth dose responseAlthough anti-Spike antibody titers decreased over time, there is scarce information of the fourth dose effect in patients with CKD.51 Published studies have focused their attention to KT recipients, where a fourth dose improves humoral responses, and should be primarily offered to prior non-responders.52–54 In this regard, SENCOVAC showed that in KT recipients the fourth vaccine dose did not significantly increase anti-Spike antibodies titers (Fig. 4). More worrisome was the low rate of seroconversion achieved in previously negative KT recipients after this new booster, raising important concerns over the optimal strategy for persistently negative patients.55 In other CKD cohorts, a humoral response was observed and anti-Spike antibody titers increased after the fourth dose.55,56 In fact, among non-KT recipients, after the third dose, the prevalence of a negative humoral response was very low (<5%).55 Beyond booster administration and prior response to vaccination, steroids and mycophenolate mofetil prescription was associated to low response to a fourth vaccine dose in KT.55,56 Similarly, suboptimal responses to SARS-CoV-2 vaccines were observed in other immunosuppressed populations.57,58 However, whether anti-Spike antibody titers represent the best readout to evaluate an optimal humoral response is unclear and to our knowledge an optimal antibody titer threshold that avoids severe COVID-19 or complications has not yet been identified. Until now, all patients with CKD have been eligible for a full immunization against SARS-CoV-2 with four doses, but the optimal moment for further re-boostering needs to be established.59
A recent report from the general population in Israel disclosed that a fourth dose results in a transient increase in anti-Spike antibody titers and in increased protection from breakthrough COVID-19 for less than three months.60 This suggest that every effort should be made to time booster doses just before COVID-19 waves.
Breakthrough SARS-CoV-2 infectionsProtection against severe SARS-CoV-2 infections following vaccination was demonstrated by pivotal clinical trials. The severity of COVID-19 substantially decreased after completing the initial vaccination schedule.61–63 However, vaccination did not reduce to zero the risk of COVID-19 complications and mortality still remained high, especially in KT recipients, despite new SARS-CoV-2 variants that appear be less lethal, at least in populations with a high degree of exposure to SARS-CoV-2 infection and vaccines, the availability of booster doses and increased access to intensive care unit care.64 Data of the last SENCOVAC report shows that probably patients with CKD should benefit from establishing a higher cut-off of anti-Spike antibody titers to ensure their protection, at least against COVID-19 requiring admissions.55 Indeed, we know that the optimal vaccination strategy should consider the infection background, comorbidities, immunosuppressive drugs and the amount of received vaccine (number of doses and type). Luckily, the pandemic is evolving, and we are now facing the same pathogen but with a different costume.
Cellular response after SARS-CoV-2 vaccinationCellular responses to SARS-CoV-2 vaccination were altered in patients with kidney failure, including HD patients and, as expected, more pronouncedly in KT recipients.65,66 Sattler A et al. characterized cellular response to BNT162b2 prime-boost vaccination in KT recipients and HD patients. The prevalence of spike-specific CD4+ T cell responses was similar in all studied groups (healthy controls, KT and HD patients). However, the magnitude of the response was lower in KT recipients than in controls, as assessed by the frequency of CD154+ CD137+ Th cells.66 In addition, the prevalence of spike-specific CD8 responses was lower KT recipients: 5.13% (2/39) KT responders vs less than half of responders in control and HD patients. Furthermore, there was a significant decrease in the CD45RO+ CD26L− effector memory-like phenotype in KT recipients and HD patients.66
In the same line, following the second SARS-CoV2 vaccine dose, an anti-Spike T cell-specific response was observed in 51% of KT recipients and in 100% of HD patients. The number of anti-Spike specific T-cells was similar to that found in health care workers. Belatacept-treated patients displayed the worst response, developing only a few specific T cells.67 By contrast, patients treated with mTOR inhibitors had higher specific T cell–derived IFNγ release than those without and this was concordant with better humoral immune response to SARS-CoV-2 vaccine.68
In Spain, specifically activated T cells were observed in 46 of 69 (62%) HD patients.69 In France, the ROMANOV study tested anti-spike receptor binding domain (RBD) IgG and interferon gamma-producing CD4+ and CD8+ specific-T cells in the circulation 10–14 days after the second dose of BNT162b2 vaccine in 106 HD patients. The majority of patients (72/80, 90%) generated at least one type of immune effector response, however their response was lower and incomplete as compared to healthy controls. HD and immunosuppressive therapy were identified as risk factors for the absence of anti-spike CD8+ T cells. Interestingly, previous history of COVID-19 in HD patients correlated with the generation of anti-spike CD8+ T cells at levels similar to healthy controls. The authors concluded that HD patients might benefit from a third vaccine dose before transplantation.70 In concordance, the interferon-γ release assay was suboptimal in KT recipients (8% positive after the first dose and 30% after the second one). In addition, the kinetics of Spike-reactive CD4+ T helper cells was delayed in KT recipients. The same pattern, but with a milder defect than in KT recipients, was observed in a large PD cohort (n=1256).30 In agreement with these results, Interferon Gamma Release Assay (IGRA) demonstrated a positive cellular response in 96.9% of controls, 89.3% of PD, 77.6% of HD, 61.3% of KT patients transplanted more than 12 months ago and only 36% of those transplanted within the previous 12 months. These results suggest that recent transplant patients with the high immunosuppressive therapy are at higher risk for severe COVID-19.71 Interestingly, although the third dose of the BNT162b2 vaccine in KT recipients did not improve seroconversion, which remained under 50%, it increased the Spike-specific T cell response.72 RBD-specific memory B cells were detected at similar proportions in virus-naïve patients on dialysis and vaccinated controls after two doses. However, in SARS-CoV-2-recovered patients on dialysis, anti-RBD memory B cells were unchanged after the third dose.73
In lymph nodes, KT recipients had deeply blunted SARS-CoV-2-specific germinal center B cell responses when compared to healthy controls, coupled with severely hindered T follicular helper cells, SARS-CoV-2 receptor binding domain-specific memory B cells, and neutralizing antibody responses.74
Present and future of SARS-CoV-2 infection managementSARS-CoV-2 has not disappeared and probably will never do. Thus, the updating of vaccines following viral mutations and periodic booster doses will be necessary, at least for high risk populations, following protocols that are well established for influenza. This is currently being done with booster doses, which are bivalent mRNA vaccines (BNT162b2, mRNA-1273),75,76 because they immunize against 2 strains of the virus, the original strain and the omicron variant, thus offering greater protection against the virus that is currently in circulation.
However, while vaccines decrease the risk of severe COVID-19 in the general population, KT recipients, as SENCOVAC26,27,38,42,55,59 has shown, and other immunocompromised patients remain at higher risk for breakthrough severe infection and persistent viral replication.77 Novel SARS-CoV-2 could become a problem in well-vaccinated populations like in Spain. Some authors warn that measuring antibody levels with standard assays that currently use an ancestral spike antigen does not inform on protection.78 Other questions pending to be answered are the number and frequency of SARS-CoV-2 vaccine doses in the general population and in immunocompromised people, whether the amount on mRNA in the vaccine should be higher in immunosuppressed individuals as is for hepatitis virus B, and whether the SARS-CV-2 vaccine will be permanently included in the annual vaccination schedule like the flu.
Other protective measures recommended for immunocompromised persons,79 including prophylactic, early access to and use of antivirals, and nonpharmaceutical interventions, such as the use of well-fitting masks or respirators, should also be considered.
Neutralizing Human monoclonal antibodies (mAbs) against SARS-CoV-2 is a promising and attractive approach for rapid prophylactic and treatment settings. However, some of these antibodies have already been deemed obsolete and are no longer used since they identify epitopes that have evolved as a consequence of mutations, and they no longer recognize the most recent SARS-CoV-2 variants. The FDA has currently issued emergency use authorizations for eight anti-SARS-CoV-2 monoclonal antibody products. Cilgavimad and tixagevimad may be used for pre-exposure prophylaxis, treatment or in situations where vaccines are less effective, such as in immunocompromised individuals. Indeed, there is evidence on the use of tixagevimab/cilgavimab in pre-exposure prophylaxis in both immunocompromised patients79 and specifically in transplant recipients.80–82
Due to the potential of newly emerging SARS-CoV-2 variants in the future, vaccines and anti-SARS-CoV-2 mAbs will need to be constantly reassessed for their efficacy. Current data indicate that tixagevimab/cilgavimab neutralize Omicron BA.2 and BA.2.12.1, but this effect may be attenuated for BA.4 and BA.5.83–86
Antiviral agents, such as nirmatrelvir–ritonavir (Paxlovid) or remdesivir, are also in clinical use. However, despite being indicated in high-risk groups, including kidney transplantation and CKD patients with additional comorbidities, they cannot be administered to patients with eGFR<30ml/min/1.73m2.87
ConclusionsVaccination against SARS-CoV-2, availability of health care resources, advances in treatment and the evolving virus have changed the impact of COVID-19 on patients with CKD. Initial vaccination and booster doses generate suboptimal humoral and cellular responses in patients with CKD, especially in KT recipients. Future strategies should include the administration of updated vaccines (for emerging SARS-CoV-2 variants) in combination with the use of neutralizing human monoclonal antibodies for high-risk patients and effective antiviral agents that can be prescribed to patients with advanced CKD. Although the protective titer of antibodies has not been established in CKD, stratification of the risk for COVID-19 complications should be based on comorbidities, prescribed treatments and humoral response to prior vaccination.
FundingThe present project has been supported by Diaverum, Vifor Pharma, Vircell, Fundación Renal Iñigo Álvarez de Toledo and Instituto de Salud Carlos III (ISCIII) RICORS program to RICORS2040 (RD21/0005/0001) funded by European Union – NextGenerationEU, Mecanismo para la Recuperación y la Resiliencia (MRR), FEDER funds.
Conflict of interestB. Quiroga has received honoraria for conferences, consulting fees and advisory boards from Vifor-Pharma, Astellas, Amgen, Bial, Ferrer, Novartis, AstraZeneca, Sandoz, Laboratorios Bial, Esteve, Sanofi-Genzyme, Otsuka. MJ. Soler reports honorarium for conferences, consulting fees and advisory boards from Astra Zeneca, NovoNordsik, Esteve, Vifor, Bayer, Mundipharma, Ingelheim Lilly, Jansen, ICU Medical, and Boehringer. A. Ortiz has received consultancy or speaker fees or travel support from Astellas, Astrazeneca, Amicus, Amgen, Fresenius Medical Care, Bayer, Sanofi-Genzyme, Menarini, Kyowa Kirin, Alexion, Otsuka and Vifor Fresenius Medical Care Renal Pharma and is Director of the Catedra Mundipharma-UAM of diabetic kidney disease and the Catedra Astrazeneca-UAM of chronic kidney disease and electrolytes. P. de Sequera reports honorarium for conferences, consulting fees and advisory boards from Amgen, Astellas, Astra Zeneca, Baxter, Braun, Fresenius Medical Care, GlaxoSmithKline, Nipro, Otsuka, Sandoz, Nipro, Vifor-Pharma. She is the present president of the Spanish Society of Nephrology (S.E.N.).
- •
Patients of the CKD spectrum present an increased risk for COVID-19 complications and death.
- •
Patients on dialysis and, especially, kidney transplant recipient develop suboptimal responses to initial vaccination and booster doses.
- •
Impaired humoral and cellular responses differ across the CKD spectrum. Predictors of humoral response include previous response to vaccination, number of vaccine doses, background immunosuppression and, in some analyses type of vaccine.
- •
Future directions to protect patients with CKD include the administration of new boosters with efficacy against currently spreading SARS-CoV-2 variants, the use of neutralizing anti-SARS-CoV-2 monoclonal antibodies and of antiviral agents that can be prescribed to this population.
We thank all the involved centers and healthcare workers for their implication in this project. In addition, we want to thank the support of the Sociedad Española de Enfermería Nefrológica (SEDEN), Organización Nacional de Trasplantes (ONT) and Sociedad Española de Trasplante (SET).