Diabetic kidney disease, a common complication in patients with type 2 diabetes mellitus, is associated with a markedly increased morbidity and mortality, especially of cardiovascular origin, and faster progression to end-stage renal disease. To date, reducing cardiovascular and renal risk in this population was based on strict control of cardiovascular risk factors and the renin-angiotensin system blockade. More recently, sodium-glucose cotransporter type 2 inhibitors have demonstrated to offer cardiovascular and renal protection, but the residual risk remains high and their antihyperglycemic efficacy is limited in moderate-severe CKD.
Therefore, drugs with a potent antihyperglycemic effect, independent of the glomerular filtration rate, with a low risk of hypoglycemia, that reduce weight in overweight/obese patients and that provide cardiovascular and renal protection, such as GLP-1 receptor agonists, are needed. However, these drugs require subcutaneous administration, which may limit their early use. The recent availability of oral semaglutide may facilitate the early introduction of this family with proven cardiovascular and renal benefits and excellent safety profile. In this review the family is analyzed as well as their cardiovascular and renal effects
La enfermedad renal diabética, frecuente en pacientes con diabetes mellitus tipo 2, se asocia con un marcado incremento de la morbimortalidad, especialmente cardiovascular, y de progresión a enfermedad renal crónica terminal. Hasta la fecha, la reducción del riesgo cardiovascular y renal en esta población se ha basado en el control estricto de los factores de riesgo cardiovascular y el uso de inhibidores del sistema renina-angiotensina. Mas recientemente, los inhibidores del cotransportador sodio-glucosa tipo 2 han demostrado ofrecer protección cardiovascular y renal, pero el riesgo residual sigue siendo alto y su eficacia antihiperglucemiante es limitada en ERC moderada-severa.
Por ello se precisan fármacos con un potente efecto antihiperglucemiante, independiente del filtrado glomerular, con bajo riesgo de hipoglicemia, que reduzcan el peso en pacientes con sobrepeso/obesidad y que proporcionen una protección cardiovascular y renal, como los agonistas del receptor de GLP-1. Sin embargo, estos fármacos precisan de su administración subcutánea, lo que puede limitar su uso temprano. La reciente disponibilidad de semaglutida oral puede ayudar a la introducción precoz de esta familia con beneficios cardiovasculares y renales probados y excelente perfil de seguridad. En esta revisión se revisa la familia y sus efectos cardiovasculares y renales.
Type 2 diabetes mellitus (DM2) affects a 13.8% of the Spanish population1 and its prevalence is expected to increase in the future.2 The presence of DM2 increases significantly the risk of cardiovascular events, chronic kidney disease (CKD), and premature death3,4 and as its prevalence increases it is expected to rise the incidence of microvascular and macrovascular complications.5 Until the last decade, the control of blood glucose, blood pressure, dyslipidemia, and the use of inhibitors of the renin-angiotensin system (iSRA) have been the only strategies available to reduce cardiovascular and renal risk in this population. Although strict glycemic control has beneficial effects on microvascular complications (retinopathy, nephropathy or neuropathy), it does not seem to reduce the risk of cardiovascular disease, and it is associated with an increased risk of hypoglycemia in patients with DM2.3,6 Until recently, the antihyperglycemic drugs available have improved glycemic control, but without much benefits on cardiovascular and/or renal risk. Furthermore, some were these drugs were associated with potential undesirable effects, such as the risk of heart failure or fractures (complications that are especially prevalent in patients with CKD), this is the case of thiazolidinediones, and the use of sulfonylureas and insulin are associated with weight gain or the risk of hypoglycemia. In addition, the antihyperglycemic effect of sodium-glucose cotransporter type 2 (iSGLT2) inhibitors is reduced as glomerular filtration rate (GFR) decreases, especially if GFR < 45 ml/min/1.73 m2.7–9
CKD affects 20–40% of the patients with diabetes10; in Spain, its current prevalence ranges between 28–33%11,12 and it is a recognized as a cardiovascular risk factor and increases the mortality in this population.13–15 Furthermore, in patients with DM2 and advanced CKD there are limitations in the prescription of certain antihyperglycemic drugs (e.g., metformin or sulfonylureas) and there is an increased risk of hypoglycemia with antihyperglycemic treatment,16 a complication that is associated with increased cardiovascular risk17 and hinders the achievement of glycemic control targets.
The intensive treatment of the multiple risk factors reduces the development/progression of renal disease18 which is why guidelines insist on comprehensive management of the patient with diabetes (glycemic control and control of cardiovascular risk factors, use of renin-angiotensin system inhibitors, as well as lifestyle changes, including smoking cessation or weight loss in overweight/obese individuals, among others).19 However, the residual renal risk remains very high in optimally treated patients.20 More recently, iSGLT2 has been shown to reduce the risk of cardiovascular and renal events, but still, the residual risk of renal disease progression remains high.21,22 Nevertheless, although the rate of diabetes-related complications (such as myocardial infarction, stroke, lower limb amputations or mortality) have decreased significantly in recent decades, this is not the same for CKD patients requiring renal replacement therapy attributed to diabetic kidney disease (DKD).23 Therefore new treatments are needed that improve glycemic control independently of GFR, with a low risk of hypoglycemia and able to reduce cardiovascular and renal risk in this population.
Glucagon-like peptide-1 (GLP-1) receptor agonists are a family of antihyperglycemic drugs that have demonstrated potent HbA1c reduction, low risk of hypoglycemia, with associated weight reduction, and proven cardiovascular and renal benefits.24 In this review we describe this type of drugs, their cardiovascular and renal benefits, as well as their clinical potential in patients with DKD, especially with the new oral semaglutide formulation, now available on the market.
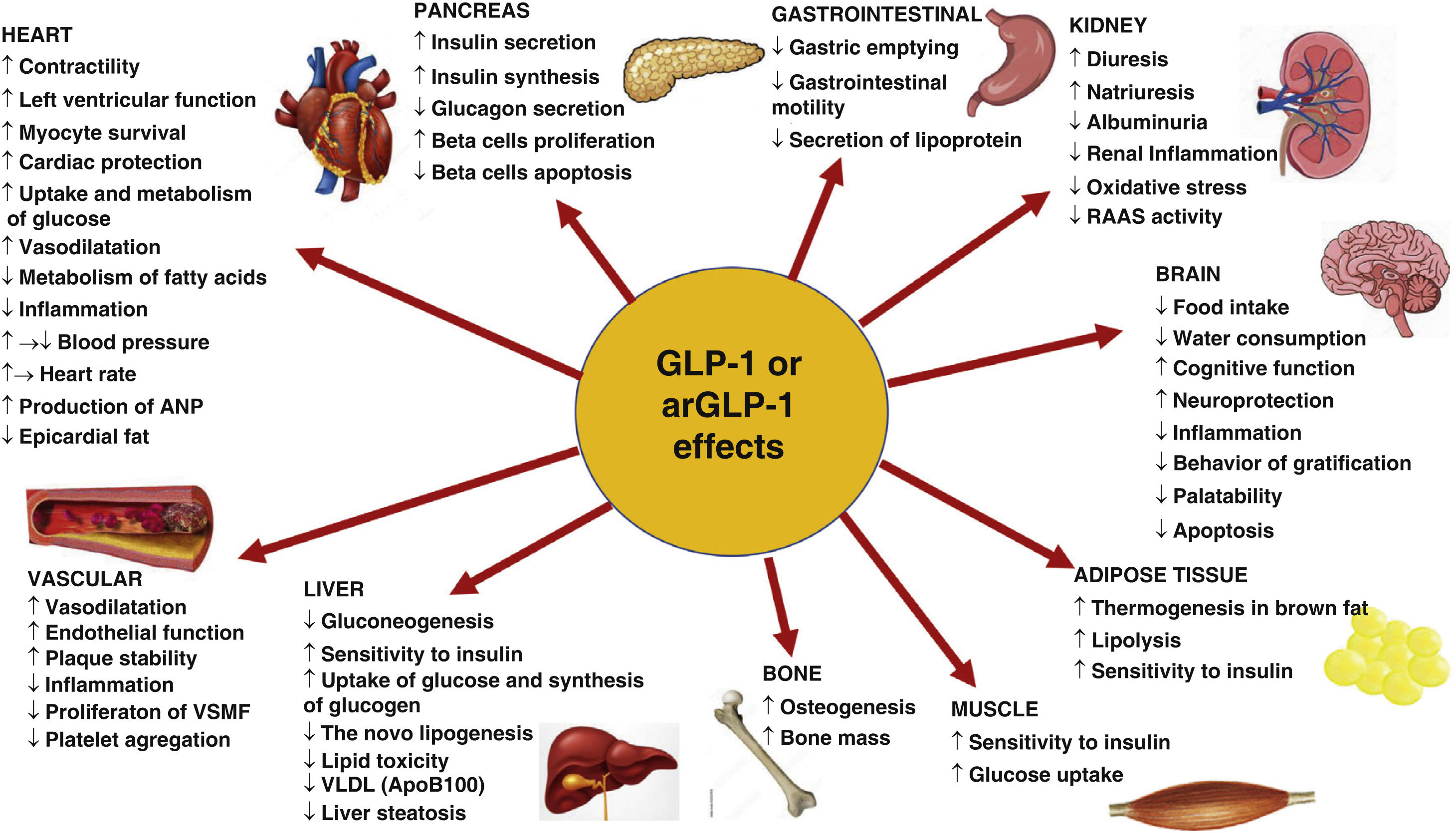
Glucagon-like peptide type 1 receptor agonistsIncretin effect and systemic effects of glucagon-like peptide type 1 and its agonistsThe incretin effect describes the phenomenon whereby oral glucose elicits higher (2–3 times) insulin secretory responses than does intravenous glucose.25 This is due to incretin hormones, such as GLP-1 or glucose-dependent insulinotropic polypeptide, which stimulate insulin secretion in response to food intake. GLP-1 is a peptide secreted by L-type enteroendocrine cells in the ileum and colon in response to different nutrients, mainly glucose and lipids, whereas glucose-dependent insulinotropic polypeptide is produced by K cells present in the duodenum and the first portion of the jejunum. GLP-1, acting on its receptor, has physiological actions on multiple target organs: it increases glucose-stimulated insulin secretion in pancreatic beta cells, reduces glucagon secretion by α cells, improves insulin sensitivity and reduces food intake.26 In cellular and murine models GLP-1 promotes β cell neogenesis and inhibits their apoptosis.27 Insulin secretion induced by GLP-1 is dependent of the glycemia and therefore does not induce hypoglycemia (Fig. 1). In patients with DM2 the incretin response is diminished, but the insulinotropic effect of GLP-1 is conserved.28 Under normal conditions GLP-1 has a short half-life (approximately 2 min), as it is rapidly degraded by dipeptidylpeptidase 4, so GLP-1 analogues resistant to this enzyme have been developed for the treatment of DM2.29
arGLP-1: glucagon-like peptide 1 receptor agonist; VSMF: vascular smooth muscle fiber; ANP: atrial natriuretic peptide; RAAS: renin-angiotensin-aldosterone system. Modified from Müller et al.29
The GLP-1 receptor (rGLP-1) belongs to the B family of G protein-coupled receptors and is expressed in the pancreas, gastrointestinal tract, central nervous system (CNS), kidneys, lungs and heart. They are also expressed in other tissues, such as blood vessels or adipose tissue29,30 although their localization in different tissues and organs yields discrepant results.31
GLP-1 or its agonists slow gastric emptying. They also act on the CNS promoting satiety, reducing food intake and favoring weight loss.27,29 Agonists of rGLP-1 (arGLP-1) have also demonstrated neuroprotective and antiapoptotic effects, a reduction of oxidative stress and inflammation, and improve cognitive and memory function (Fig. 1).29,32
With respect to the vascular system, rGLP-1 activation induces modest decreases in blood pressure,33,34 it has a vasodilator effect, improves arterial stiffness, and prevents atherosclerotic plaque formation-and has plaque stabilizing effects-through its anti-inflammatory and antiapoptotic effects as demonstrated in animal models.29,32,35–38 Despite controversies about rGLP-1 expression in vascular smooth muscle and endothelial cells, studies have shown that arGLP-1 has antiproliferative effects on these cells,39 reduces oxidative stress, improves endothelial function, and increases nitric oxide production in experimental models.35–40
In the heart they produce a discrete increase in heart rate (2–4 beats/min), although this does not seem to be associated with an increase in arrhythmias or cardiovascular events. They increase glucose uptake and utilization and improve cardiac metabolism, as well as left ventricular function36,41; increase the release of atrial natriuretic peptide; have anti-inflammatory, anti-ischemic and anti-fibrotic effects and inhibit myocardiocyte apoptosis.29,32,42 More recently, it has been reported that they also reduce epicardial fat.43 They also inhibit platelet aggregation and thrombosis.44
In the lungs, rGLP-1 activation conferred protection against lipopolysaccharide-induced acute lung injury.45
In adipose tissue they favor adipocyte differentiation, decreases lipid deposition in white fat and facilitate its conversion to brown fat, as well as thermogenesis and energy consumption29,32,46–48; the effect on the intestine results in a reduction of postprandial lipidemia and chylomicron secretion.48,49 In the muscles, they improve insulin sensitivity and increase glucose uptake.
Renal expression of rGLP-1 has not been fully characterized due to insufficient specificity and sensitivity of the antibodies used, although it have been non-uniformly detected in endothelial cells, macrophages, juxtaglomerular cells and proximal tubule in several animal models and human tissue. GLP-1 infusion increases natriuresis by reducing tubular sodium reabsorption and reduces hyperfiltration in obese subjects.50 Chronic treatment with arGLP-1 in patients with DM2 has shown no effect on measured GFR and effective renal plasma flow in the fasting or postprandial state, however it maintains its natriuretic effect.51 GLP-1 and its agonists induce natriuresis by inhibition of the Na+/H + exchanger type 3 in the proximal tubule, although interactions with the renin, insulin, or glucagon system cannot be excluded52–54 because they reduce circulating levels of RAS components51,53–55 (Fig. 1).
Metabolic effects of glucagon-like peptide-1 receptor agonistsThe arGLP-1 s show a potent antihyperglycemic effect with decreases between 0.5% and 1.3% of HbA1c, which is greater in those with a long half-life56 with low risk of hypoglycemia, as has been shown in cardiovascular safety studies, where there were no differences in hypoglycemia rates versus placebo, except if associated with sulfonylureas or insulin.57–64 The low risk of hypoglycemia has been observed even in patients with DKD.65–67 who are more susceptible to this complication.16
It is also important to highlight the loss of weight and waist circumference with arGLP-1, mainly as a consequence of a reduction in body fat and not in lean mass, which is maintained during long-term follow-up.68 This weight loss is attributed to the slowing of gastric emptying which increases satiety, to the increase in resting energy consumption and its direct effects on appetite and satiety control centers in the CNS.
Obesity and overweight, which are common in patients with DM2, are a factors that favor the development/progression of CKD through both direct and indirect effects.69 Although the precise mechanisms the association between obesity and CKD are not totally defined it has been proposed that hypertension, glomerular hyperfiltration, hyperglycemia, lipid toxicity, inflammation or the role of adipose tissue and its adipokines are involved.70 Weight reduction by bariatric surgery, pharmacological treatment or dietary measures are associated with a nephroprotective effect,71–73 so that weight reduction is a fundamental objective in the nephroprotection of patients with DKD.
Hepatic steatosis is closely related to insulin resistance, diabetes mellitus or metabolic syndrome, and is associated with increased risk of cardiovascular and renal disease.74 Hepatic steatosis has been associated with the prevalence and incidence of renal disease. The decreased in GFR or albuminuria has been associated with the presence of nonalcoholic steatohepatitis or the severity of fibrosis, whereas improvement in liver histology is associated with improvement in CKD.75 arGLP1 improves insulin levels, regulates autophagy, inhibits hepatic neoglycogenesis, reduces hepatic fat content, plasma liver enzyme levels and hepatic histological improvements have been described with liraglutide and semaglutide.49,76 Likewise, epicardial and perirenal fat are associated with increased cardiorenal risk,77 so that the reduction in ectopic fat43 as demonstrated with the use of arGLP-1 has could be beneficial in CKD.
Finally, arGLP-1 has been shown to improve the pre- and postprandial lipid profile, with a reduction in the levels of free fatty acids, triglycerides and LDL cholesterol, as well as the fraction of small and dense LDL and a reduction of the increase levels of triglycerides and apo-B48 postprandial.78–82 Likewise, it has been reported a reduction in markers of inflammation, plasminogen activator inhibitor type 1 and leptin whereas adiponectin levels increase.47,48
Regarding the adverse effects of this family, the most important are gastrointestinal, especially nausea and vomiting of mild-moderate intensity, which are reduced with time.
All this translates into an improvement in glycemic control and dyslipidemia, a reduction in blood pressure, weight, and ectopic visceral fat deposits, as well as inflammation and coagulation, which could translate into greater cardiovasculorenal protection with the use of these drugs.
Key differential aspects of the group of glucagon-like peptide-1 receptor agonist drugsThere are several arGLP-1 s approved or under study for the use in DM2: exenatide (exenatide and exenatide LAR or weekly), liraglutide, lixisenatide, dulaglutide, semaglutide, albiglutide (withdrawn from the market), and efpeglenatide (in clinical development). Almost all are administered subcutaneously, although oral semaglutide is currently available in Spain (see below). These drugs are divided into: incretinmimetics, derived from exendin-4 (a peptide obtained from the saliva of the Gila monster [Heloderma suspectum]); or analogs of human GLP-1, with changes in the amino acid sequence that make them resistant to dipeptidylpeptidase 4. The arGLP-1 s present other differences in clinically relevant biological properties (half-life, dosing interval and molecular weight).
Short half-life agents (exenatide twice daily or lixisenatide) produce spikes with transient increases in plasma arGLP-1 levels, resulting in intermittent activation of the receptor and have a potent effect on gastric emptying and postprandial glycemia by reducing the rate of glucose entry into the duodenum.
Those with a long half-life (weekly exenatide, efpeglenatide, liraglutide, semaglutide, albiglutide and dulaglutide) produce a sustained activation of rGLP1, resulting in a greater reduction in HbA1c, basal glycemia and body weight and would have fewer gastrointestinal side effects; they do not have a significant effect on gastric emptying due to a tachyphylaxis phenomenon, and have a modest effect on postprandial glycemia.24,78
Those of small molecular weight (exenatide, lixisenatide, liraglutide and semaglutide) can penetrate the blood-brain barrier more easily than those of high molecular weight (dulaglutide, albiglutide and epfeglenatide) and could generate differential effects on central appetite signaling pathways and weight loss.24,33,83
Exendin-4 analogues are eliminated by the the kidneys, so their use is not recommended in patients with advanced CKD (GFR < 30 ml/min/1.73 m2), while GLP-1 analogues have other elimination routes and can be prescribed in patients up to CKD stage 4 (GFR15 ml/min/1.73 m2).
All this means that there are differences between these compounds in terms of their potency in reducing HbA1c, baseline glycemia or weight reduction, among other factors, which should be considered when choosing drugs from this family.78,83,84
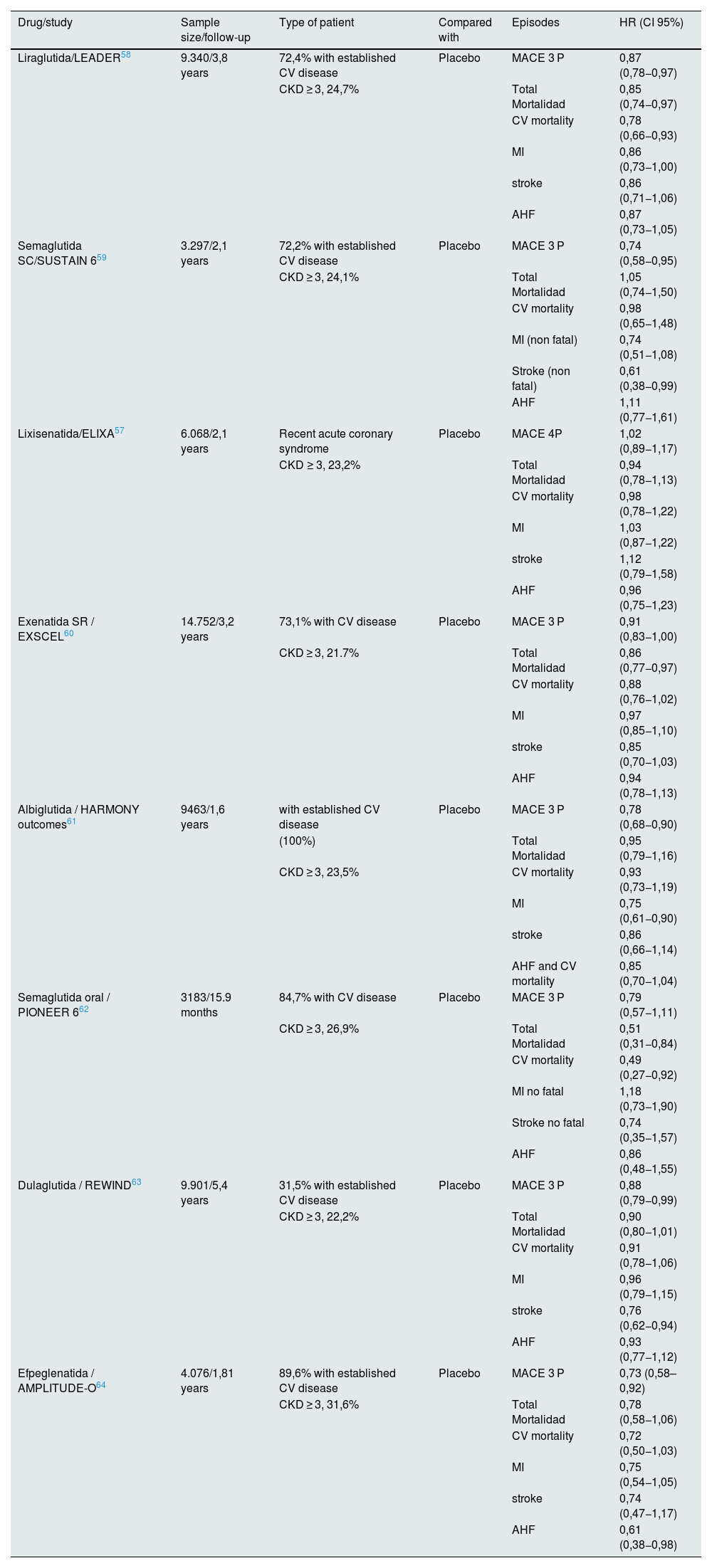
Cardiovascular protection of glucagon-like peptide type 1 receptor agonistsSeveral randomized clinical trials have analyzed the cardiovascular safety of arGLP1 versus standard therapy. All trials analyzed the composite of 3 major adverse cardiovascular events (MACE), which included cardiovascular death, non-fatal myocardial infarction, or non-fatal stroke, except the lixisenatide study that considered 4-point MACE, which also included hospitalization for heart failure (Table 1).57–64
Although all the studies with arGLP-1 have demonstrated cardiovascular safety (noninferiority vs. placebo), most of them (liraglutide, semaglutide SC, dulaglutide, albiglutide, and efpeglenatide) have also demonstrated a reduction in MACE with respect to placebo, while weekly exenatide or oral semaglutide showed a tendency to reduce MACE. In the LEADER study with liraglutide, in patients with CKD stage ≥3, this cardiovascular benefit proved to be greater.58 Although the cardiovascular safety studies were not powered to detect differences in individual components of MACE, some differences between studies were seen in these individual components; thus liraglutide and oral semaglutide significantly reduced cardiovascular death, subcutaneous semaglutide and dulaglutide significantly reduced nonfatal stroke, albiglutide reduced the risk of nonfatal myocardial infarction, and epfeglenatide reduced the incidence of heart failure, while weekly exenatide or lixisenatide showed no significant benefit on any component of MACE.
However, it is difficult to discern whether these differences between studies are due to real differences between the molecules in terms of their cardiovascular effects. Potential explanations include differences in the type of patients included, adherence to study treatment, sample size, duration of drug exposure, and GLP1 receptor activation with greater HbA1c reduction with longer-acting human GLP1-based analogs than with some short-acting exendin-4 analogs. Recent results from the AMPLITUDE-O trial with efpeglenatide64 suggest that the cardiovascular benefits of this family are not exclusive of the human GLP-1 analogs. Likewise, the cardiovascular benefit of efpeglenatide was independent of GFR or the concomitant use of iSGLT-2, suggesting its additive and independent effect from the latter,85 as was also observed in the EXSCEL study.86
A recent meta-analysis of the 8 published cardiovascular safety clinical trials, and which included a total of 60. 080 patients, concluded that, overall, arGLP-1 reduces MACE by 14% (HR 0.86, 95% CI: 0.80−0.93, p < 0.0001), with no significant differences regarding structural homology with human GLP-1, risk of cardiovascular events (based on the risk of MACE in the placebo group as low, medium or high), presence or not of cardiovascular disease, baseline HbA1c, body mass index, duration of follow-up, administration interval (daily or weekly) or renal function. Analyzing the individual components of MACE, the use of arGLP-1 reduced the risk of cardiovascular death (HR 0.87, 95% CI: 0.80−0.94; p = 0.001), fatal and non-fatal myocardial infarction (HR 0.90, 95% CI: 0.83−0.98; p = 0.020) and fatal or non-fatal stroke (HR 0.83, 95% CI: 0.76−0.92; p = 0.0002). ArGLP-1 also reduced total mortality (HR 0.88, 95% CI 0.82−0.94, p = 0.0001) and hospital admission due to heart failure (HR 0.89, 95% CI 0.82−0.98, p = 0.013), all without increasing the risk of severe hypoglycemia, pancreatic problems or retinopathy.87 A recent post hoc analysis of CVOT with semaglutide has shown a gain in time free of cardiovascular disease of 1.7 years in those patients with CKD vs. 0.2 years in diabetic patients with cardiovascular risk factors only, suggesting an additional benefit of semaglutide in this population with high cardiovascular risk.88
The mechanisms involved in the cardiovascular benefits of this family are various. A meta-regression study suggested a linear relationship between HbA1c reduction and MACE risk in patient with arGLP-1.89 Publication on mediation analyses also suggest that cardiovascular benefit could be mediated in part by effects on HbA1c, on blood pressure or reduction in urine albumin-creatinine ratio90,91 (see below), as well as by their effect on lipid profile.81 However, in subgroup analyses of cardiovascular safety studies, baseline HbA1c, weight, previous cardiovascular disease, or renal function did not predict the beneficial effects of these drugs on MACE,87 so other mechanisms, such as the previously mentioned anti-inflammatory, antifibrotic, antiatherogenic, vasodilator, or endothelial function-enhancing effects, have been suggested to influence the cardiovascular benefit of these drugs29,78 (Fig. 1).
Renal protection with glucagon-like peptide type 1 receptor agonistsIn CVOTs of arGLP1 renal outcomes were prespecified as key secondary outcomes in 4 studies done with liraglutide, subcutaneous semaglutide, dulaglutide, and efpeglenatide,58,59,63,64 or were collected as safety parameters in other studies.60 Overall, the prevalence of CKD stage ≥3 in these studies was low (20–30%) (Table 1).
Cardiovascular safety clinical trials with arGLP1 in patients with type 2 diabetes mellitus.
| Drug/study | Sample size/follow-up | Type of patient | Compared with | Episodes | HR (CI 95%) |
|---|---|---|---|---|---|
| Liraglutida/LEADER58 | 9.340/3,8 years | 72,4% with established CV disease | Placebo | MACE 3 P | 0,87 (0,78−0,97) |
| CKD ≥ 3, 24,7% | Total Mortalidad | 0,85 (0,74−0,97) | |||
| CV mortality | 0,78 (0,66−0,93) | ||||
| MI | 0,86 (0,73−1,00) | ||||
| stroke | 0,86 (0,71−1,06) | ||||
| AHF | 0,87 (0,73−1,05) | ||||
| Semaglutida SC/SUSTAIN 659 | 3.297/2,1 years | 72,2% with established CV disease | Placebo | MACE 3 P | 0,74 (0,58−0,95) |
| CKD ≥ 3, 24,1% | Total Mortalidad | 1,05 (0,74−1,50) | |||
| CV mortality | 0,98 (0,65−1,48) | ||||
| MI (non fatal) | 0,74 (0,51−1,08) | ||||
| Stroke (non fatal) | 0,61 (0,38−0,99) | ||||
| AHF | 1,11 (0,77−1,61) | ||||
| Lixisenatida/ELIXA57 | 6.068/2,1 years | Recent acute coronary syndrome | Placebo | MACE 4P | 1,02 (0,89−1,17) |
| CKD ≥ 3, 23,2% | Total Mortalidad | 0,94 (0,78−1,13) | |||
| CV mortality | 0,98 (0,78−1,22) | ||||
| MI | 1,03 (0,87−1,22) | ||||
| stroke | 1,12 (0,79−1,58) | ||||
| AHF | 0,96 (0,75−1,23) | ||||
| Exenatida SR / EXSCEL60 | 14.752/3,2 years | 73,1% with CV disease | Placebo | MACE 3 P | 0,91 (0,83−1,00) |
| CKD ≥ 3, 21.7% | Total Mortalidad | 0,86 (0,77−0,97) | |||
| CV mortality | 0,88 (0,76−1,02) | ||||
| MI | 0,97 (0,85−1,10) | ||||
| stroke | 0,85 (0,70−1,03) | ||||
| AHF | 0,94 (0,78−1,13) | ||||
| Albiglutida / HARMONY outcomes61 | 9463/1,6 years | with established CV disease | Placebo | MACE 3 P | 0,78 (0,68−0,90) |
| (100%) | Total Mortalidad | 0,95 (0,79−1,16) | |||
| CKD ≥ 3, 23,5% | CV mortality | 0,93 (0,73−1,19) | |||
| MI | 0,75 (0,61−0,90) | ||||
| stroke | 0,86 (0,66−1,14) | ||||
| AHF and CV mortality | 0,85 (0,70−1,04) | ||||
| Semaglutida oral / PIONEER 662 | 3183/15.9 months | 84,7% with CV disease | Placebo | MACE 3 P | 0,79 (0,57−1,11) |
| CKD ≥ 3, 26,9% | Total Mortalidad | 0,51 (0,31−0,84) | |||
| CV mortality | 0,49 (0,27−0,92) | ||||
| MI no fatal | 1,18 (0,73−1,90) | ||||
| Stroke no fatal | 0,74 (0,35−1,57) | ||||
| AHF | 0,86 (0,48−1,55) | ||||
| Dulaglutida / REWIND63 | 9.901/5,4 years | 31,5% with established CV disease | Placebo | MACE 3 P | 0,88 (0,79−0,99) |
| CKD ≥ 3, 22,2% | Total Mortalidad | 0,90 (0,80−1,01) | |||
| CV mortality | 0,91 (0,78−1,06) | ||||
| MI | 0,96 (0,79−1,15) | ||||
| stroke | 0,76 (0,62−0,94) | ||||
| AHF | 0,93 (0,77−1,12) | ||||
| Efpeglenatida / AMPLITUDE-O64 | 4.076/1,81 years | 89,6% with established CV disease | Placebo | MACE 3 P | 0,73 (0,58–0,92) |
| CKD ≥ 3, 31,6% | Total Mortalidad | 0,78 (0,58−1,06) | |||
| CV mortality | 0,72 (0,50−1,03) | ||||
| MI | 0,75 (0,54−1,05) | ||||
| stroke | 0,74 (0,47−1,17) | ||||
| AHF | 0,61 (0,38−0,98) |
CV, cardiovascular; CKD, Chronic kidney disease stage ≥3 (FGe<60 ml/min/1,73 m2); AHF, Hospital admission due to heart failure; MI, myocardial infarction; MACE 3P, 3-point cardiovascular composite episode; MACE 4P, 4-point cardiovascular composite episode, including hospitalization due to non-stable angina.
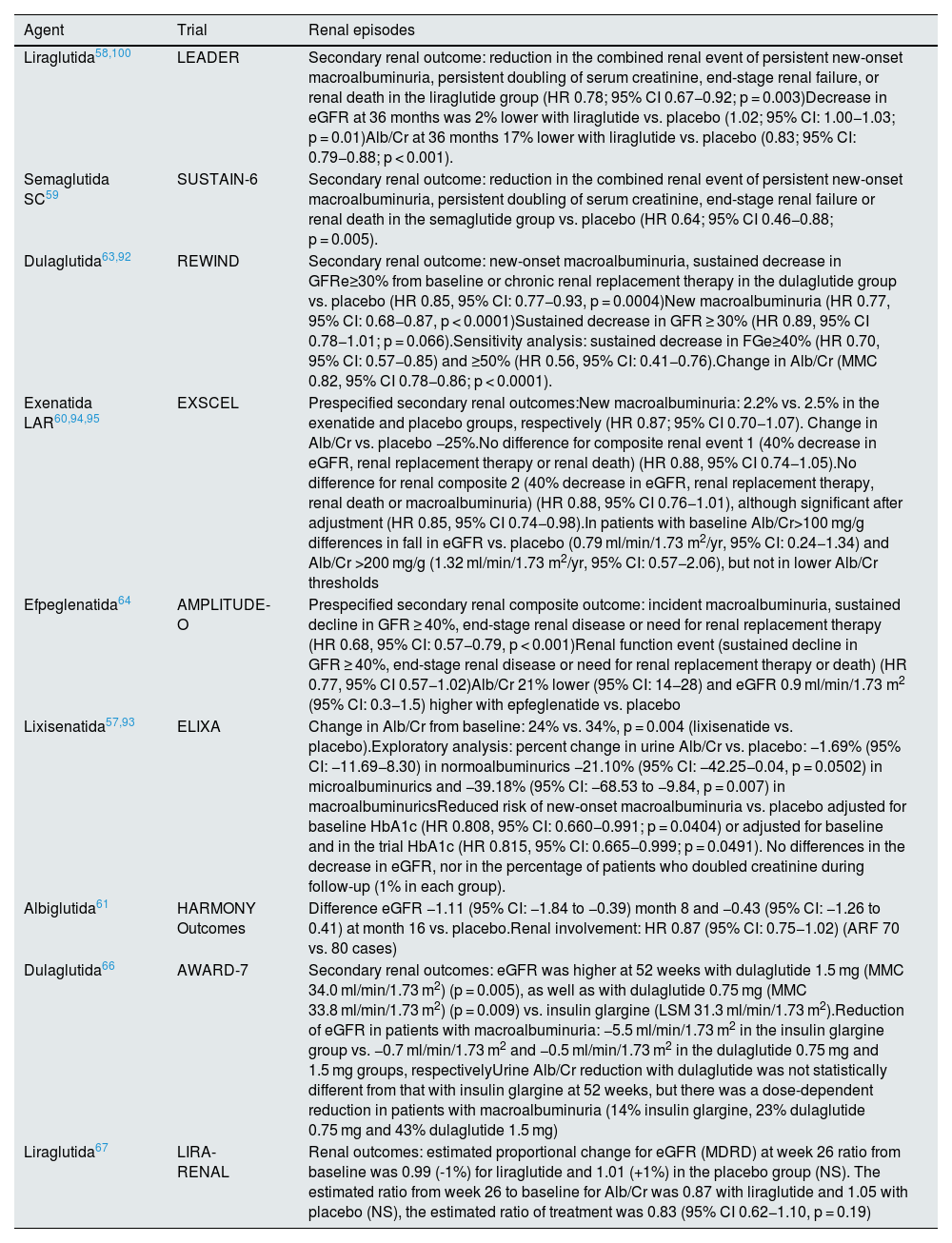
In those studies that prospectively evaluated renal targets as key secondary endpoints, included a composite renal episode that consist of the development of macroalbuminuria, the development of end-stage CKD or the need for renal replacement therapy or a significant reduction in renal function from baseline, which included a GFR<45 ml/min/1.73 m2 and a doubling of serum creatinine58,59 or a decrease in GFR ≥ 30%63 or ≥40%.64 The combined renal target was reduced by approximately 15%–35% with liraglutide, semaglutide SC, dulaglutide, or efpeglenatide compared with placebo, basically at the expense of reducing the risk of developing macroalbuminuria, without reaching statistical significance for the other components (Table 2). However, in a sensitivity analysis of the REWIND study with dulaglutide, the relative risk reduction for a 40% or 50% decrease in eGFR was 30% and 44%, both significant, in the dulaglutide group compared to placebo, respectively.92 This lower risk of developing macroalbuminuria was also confirmed in an exploratory analysis of the ELIXA (lixisenatide) study.93 In the EXSCEL study (exenatide LAR), renal episodes were analyzed as additional microvascular episodes, and no differences were observed in the risk of episodes related to decreased eGFR, but differences were observed in the composite episode, which included decreased eGFR and macroalbuminuria, although only after adjustment94 (Table 2).
Renal outcomes with arGLP1 in clinical trials.
| Agent | Trial | Renal episodes |
|---|---|---|
| Liraglutida58,100 | LEADER | Secondary renal outcome: reduction in the combined renal event of persistent new-onset macroalbuminuria, persistent doubling of serum creatinine, end-stage renal failure, or renal death in the liraglutide group (HR 0.78; 95% CI 0.67−0.92; p = 0.003)Decrease in eGFR at 36 months was 2% lower with liraglutide vs. placebo (1.02; 95% CI: 1.00−1.03; p = 0.01)Alb/Cr at 36 months 17% lower with liraglutide vs. placebo (0.83; 95% CI: 0.79−0.88; p < 0.001). |
| Semaglutida SC59 | SUSTAIN-6 | Secondary renal outcome: reduction in the combined renal event of persistent new-onset macroalbuminuria, persistent doubling of serum creatinine, end-stage renal failure or renal death in the semaglutide group vs. placebo (HR 0.64; 95% CI 0.46−0.88; p = 0.005). |
| Dulaglutida63,92 | REWIND | Secondary renal outcome: new-onset macroalbuminuria, sustained decrease in GFRe≥30% from baseline or chronic renal replacement therapy in the dulaglutide group vs. placebo (HR 0.85, 95% CI: 0.77−0.93, p = 0.0004)New macroalbuminuria (HR 0.77, 95% CI: 0.68−0.87, p < 0.0001)Sustained decrease in GFR ≥ 30% (HR 0.89, 95% CI 0.78−1.01; p = 0.066).Sensitivity analysis: sustained decrease in FGe≥40% (HR 0.70, 95% CI: 0.57−0.85) and ≥50% (HR 0.56, 95% CI: 0.41−0.76).Change in Alb/Cr (MMC 0.82, 95% CI 0.78−0.86; p < 0.0001). |
| Exenatida LAR60,94,95 | EXSCEL | Prespecified secondary renal outcomes:New macroalbuminuria: 2.2% vs. 2.5% in the exenatide and placebo groups, respectively (HR 0.87; 95% CI 0.70−1.07). Change in Alb/Cr vs. placebo −25%.No difference for composite renal event 1 (40% decrease in eGFR, renal replacement therapy or renal death) (HR 0.88, 95% CI 0.74−1.05).No difference for renal composite 2 (40% decrease in eGFR, renal replacement therapy, renal death or macroalbuminuria) (HR 0.88, 95% CI 0.76−1.01), although significant after adjustment (HR 0.85, 95% CI 0.74−0.98).In patients with baseline Alb/Cr>100 mg/g differences in fall in eGFR vs. placebo (0.79 ml/min/1.73 m2/yr, 95% CI: 0.24−1.34) and Alb/Cr >200 mg/g (1.32 ml/min/1.73 m2/yr, 95% CI: 0.57−2.06), but not in lower Alb/Cr thresholds |
| Efpeglenatida64 | AMPLITUDE-O | Prespecified secondary renal composite outcome: incident macroalbuminuria, sustained decline in GFR ≥ 40%, end-stage renal disease or need for renal replacement therapy (HR 0.68, 95% CI: 0.57−0.79, p < 0.001)Renal function event (sustained decline in GFR ≥ 40%, end-stage renal disease or need for renal replacement therapy or death) (HR 0.77, 95% CI 0.57−1.02)Alb/Cr 21% lower (95% CI: 14−28) and eGFR 0.9 ml/min/1.73 m2 (95% CI: 0.3−1.5) higher with epfeglenatide vs. placebo |
| Lixisenatida57,93 | ELIXA | Change in Alb/Cr from baseline: 24% vs. 34%, p = 0.004 (lixisenatide vs. placebo).Exploratory analysis: percent change in urine Alb/Cr vs. placebo: −1.69% (95% CI: −11.69−8.30) in normoalbuminurics −21.10% (95% CI: −42.25−0.04, p = 0.0502) in microalbuminurics and −39.18% (95% CI: −68.53 to −9.84, p = 0.007) in macroalbuminuricsReduced risk of new-onset macroalbuminuria vs. placebo adjusted for baseline HbA1c (HR 0.808, 95% CI: 0.660−0.991; p = 0.0404) or adjusted for baseline and in the trial HbA1c (HR 0.815, 95% CI: 0.665−0.999; p = 0.0491). No differences in the decrease in eGFR, nor in the percentage of patients who doubled creatinine during follow-up (1% in each group). |
| Albiglutida61 | HARMONY Outcomes | Difference eGFR −1.11 (95% CI: −1.84 to −0.39) month 8 and −0.43 (95% CI: −1.26 to 0.41) at month 16 vs. placebo.Renal involvement: HR 0.87 (95% CI: 0.75−1.02) (ARF 70 vs. 80 cases) |
| Dulaglutida66 | AWARD-7 | Secondary renal outcomes: eGFR was higher at 52 weeks with dulaglutide 1.5 mg (MMC 34.0 ml/min/1.73 m2) (p = 0.005), as well as with dulaglutide 0.75 mg (MMC 33.8 ml/min/1.73 m2) (p = 0.009) vs. insulin glargine (LSM 31.3 ml/min/1.73 m2).Reduction of eGFR in patients with macroalbuminuria: −5.5 ml/min/1.73 m2 in the insulin glargine group vs. −0.7 ml/min/1.73 m2 and −0.5 ml/min/1.73 m2 in the dulaglutide 0.75 mg and 1.5 mg groups, respectivelyUrine Alb/Cr reduction with dulaglutide was not statistically different from that with insulin glargine at 52 weeks, but there was a dose-dependent reduction in patients with macroalbuminuria (14% insulin glargine, 23% dulaglutide 0.75 mg and 43% dulaglutide 1.5 mg) |
| Liraglutida67 | LIRA-RENAL | Renal outcomes: estimated proportional change for eGFR (MDRD) at week 26 ratio from baseline was 0.99 (-1%) for liraglutide and 1.01 (+1%) in the placebo group (NS). The estimated ratio from week 26 to baseline for Alb/Cr was 0.87 with liraglutide and 1.05 with placebo (NS), the estimated ratio of treatment was 0.83 (95% CI 0.62−1.10, p = 0.19) |
Alb/Cr, ratio albúmin/ creatinine in urine; eGFR, estimated glomerular filtration rate, ARF, acute renal failure; LSM, least squares mean.
The effects on the evolution of eGFR during follow-up are variable among the different trials, with minimal differences between the active group and placebo,57,63,64 although an exploratory analysis of the EXSCEL study demonstrated a smaller fall in eGFR in patients with albuminuria greater than 100 mg/g and 200 mg/g of urine albumin/creatinine.95 Likewise, in a post-hoc analysis of the LEADER and SUSTAIN studies, a smaller fall in eGFR was observed in those with eGFR<60 ml/min/1.73 m2.96 Reduced albuminuria (or less increase) is the most consistent finding in these studies,58,59,64,93 as well as in a post hoc analysis of the SUSTAIN 1–7 trials with semaglutide SC97 and those with dulaglutide.98
In the aforementioned Sattar meta-analysis the composite renal endpoint of renal function deterioration, need for renal replacement therapy, renal death and development of macroalbuminuria was reduced by 21% (HR 0.79, 95% CI: 0.73−0.87, p < 0.0001), but was not significant for episodes of renal function deterioration (HR 0.86, 95% CI: 0.72–1.02, p = 0.089), although they were also reduced significantly after excluding the ELIXA87 study.
The renal effects of arGLP1 in patients with CKD have been evaluated in other studies. The study "Liraglutide versus placebo as an adjunct to glucose-lowering therapy in patients with type 2 diabetes and moderate renal failure" (LIRA-RENAL) did not demonstrate a benefit of liraglutide in lowering eGFR, although the duration of the study (26 weeks) may have been insufficient to show an effect. This study also showed a non-significant 17% reduction in albuminuria.67 The AWARD-7 study analyzed the efficacy of dulaglutide in patients with DM2 and moderate-severe CKD (mean eGFR 38 ml/min/1.73 m2). The decrease in mean GFR at 52 weeks was smaller in the high (1.5 mg) and low dose dulaglutide (0.75 mg) treatment groups (both −0.7 ml/min/1.73 m2) vs. the insulin glargine group (−3.3 ml/min/1.73 m2). This benefit was greater in patients with macroalbuminuria66 (Table 2).
Thus, arGLP-1 reduces albuminuria in patients with ESRD, but its effects on the progression of renal disease are not conclusive. However, albuminuria is a potent predictor of eGFR decline, so these drugs could delay progression in DKD by reducing albuminuria.99 In a subgroup analysis of the LEADER trial the effect of liraglutide vs. placebo in the fall of eGFR was better among patients with macroalbuminuria or in those with eGFR between 30−60 ml/min/1.73 m2,100 and in the AWARD-7 trial the effect of dulaglutide on renal clinical outcomes and eGFR decline was more pronounced in participants with macroalbuminuria at baseline.66 Likewise, in a post hoc analysis of the LEADER study those patients who experienced a 30% reduction in albuminuria during follow-up had fewer cardiovascular and renal events,101 which confirms that a reduction in albuminuria is associated with a reduction in cardiovascular and renal events.99,102
Finally, the renal benefits of arGLP-1 appear to be additive to those of iSGLT2, which is of particular interest in patients with DKD.85,86,103
Cardiovascular and renal benefits of these drugs have been also demonstrated in real-life studies.104–105
The causes of renal protection by some arGLP-1 s are unclear. Post hoc adjustment for HbA1C and other traditional renal risk factors did not have a major influence on renal benefit by liraglutide, semaglutide, or dulaglutide in CVOTs, nor were there differences in glycemic control in the AWARD-766 study. In fact, mediation analyses performed suggested that only approximately 25% of the benefit on renal outcomes may be mediated by improved glycemic control and 9–22% by changes on blood pressure,92,106 suggesting the role of direct mechanisms: reduction of hyperfiltration, oxidative stress and inflammation, antifibrotic effects, improvement of endothelial dysfunction and/or possible reduction of angiotensin II levels.52,54,107–109 In experimental studies of DKD, treatment with arGLP-1 has been associated with histological improvements (reduction of glomerular hypertrophy, mesangial expansion, thickening of the glomerular basement membrane and tubulointerstitial damage).110,111
In fact, the KDIGO guidelines for the management of patients with diabetes and CKD rank arGLP-1 as a second line of treatment and suggest its use in patients with DM2 and CKD who have not reached individualized glycemic targets despite the use of metformin and iSGLT2, or in those who cannot use these drugs since liraglutide, semaglutide and dulaglutide (results with epfeglenatide have not been published) have demonstrated cardiovascular benefits, especially among patients with established cardiovascular disease, associated with favorable renal benefits with a substantial reduction in albuminuria and probable preservation of eGFR,112 as well as other guidelines in patients with DM2 and high/very high cardiovascular risk, such as patients with DKD.19,113 Furthermore, arGLP-1 s are more potent antihyperglycemic agents than iSGLT2s in patients with DKD, and some such as semaglutide also confer greater potential for weight loss. Finally, some clinical case studies and short series suggest their benefits also in patients with CKD stage 5D, although it should be recalled that this indication is not included in the labeling of arGLP-1 s.114,115
The Effect of semaglutide versus placebo on the progression of renal impairment in subjects with type 2 diabetes and chronic kidney disease (FLOW), a phase 3 clinical trial involving more than 3,000 patients with type 2 DM and moderate-severe CKD (eGFR 25−75 ml/min/1.73 m2 and Urine Alb/Cr ratio of 300−5,000 mg/g) is currently underway (NCT03819153). The primary endpoint is a renal composite episode defined by a decrease in GFR ≥ 50%, end-stage renal failure, and death from a renal-cause or cardiovascular-caused death, and it is scheduled for completion in 2024. This trial is the first to include renal disease outcomes as a primary target with the use of this family of drugs. This study will define the clinical efficacy and renal safety of an arGLP-1 and thereby will inform about future evidence-based recommendations for treatment of DKD.
Oral semaglutideSemaglutide oral is the first oral arGLP-1. Semaglutide oral is co-formulated with sodium N-(8-[8-[2-hydroxybenzoyl]amino) caprylate (sodium salcaprozate or SNAC) which allows absorption of semaglutide across the gastric mucosa. Oral semaglutide has been approved and recently marketed in Spain following evidence from the Phase 3 clinical trial program Peptide InnOvatioN for Early diabEtes tReatment (PIONEER). This program evaluated the efficacy and safety of oral semaglutide in patients with DM2 from monotherapy to insulin addition, including HbA1c and body weight reduction outcomes, in addition to other relevant efficacy and safety endpoints, as compared to both placebo and other common antihyperglycemic drugs. Oral semaglutide at doses of 7 and 14 mg/day reduced HbA1c and body weight across the entire spectrum of DM2, as well as other endpoints, such as fasting blood glucose. Oral semaglutide was superior to placebo and other antihyperglycemic drugs, such as iDPP4 (sitagliptin), iSGLT2 (empagliflozin) or other arGLP-1 (liraglutide) in reducing HbA1c or weight.116 Oral semaglutide was well tolerated, with a safety profile similar to that of other arGLP-1 s. The most frequently reported side effects were gastrointestinal side effects of mild-moderate intensity and were transient over time. Its cardiovascular safety in patients with cardiovascular disease or high cardiovascular risk was demonstrated in the aforementioned PIONEER-6 study.62 In the PIONEER 5 study, in patients with CKD stage 3, it was evaluated the efficacy and safety of a 14 mg dose, resulting superior to placebo in terms of reduction of HbA1c (-0.8% vs. placebo) and body weight (−2.5 kg vs. placebo), with a higher percentage of patients achieving HbA1c<7.0% levels (57.8% vs 22.6% in placebo) and weight loss ≥5% (35.7% vs. 9.7% in placebo). The most frequent adverse effects consisted of mild-moderate nausea that decreased over time (19% vs. 8% on placebo) and with low risk of symptomatic or confirmed hypoglycemia (5.5% vs. 1.9% on placebo). There were no differences in eGFR between the two groups, but there was a reduction in albuminuria with oral semaglutide vs. an increase in the placebo group.117
Results from the PIONEER program demonstrate that oral semaglutide is effective, safe, and well tolerated for glycemic control in patients with DM2, including those with DKD, with the specific benefits of this family of drugs. The availability of oral semaglutide may help to broaden treatment options and facilitate the adoption of earlier treatment with arGLP-1 in the management of DM2. A heart disease study of semaglutide in patients with type 2 diabetes (SOUL, NCT03914326) evaluating the cardiovascular benefit of oral semaglutide is currently underway.
Conclusions- -
The prevalence of CKD is high in DM2 and DKD is associated with increased cardiovascular risk, mortality and progression to end-stage CKD, requiring early and intensive treatment to minimize these complications.
- -
The arGLP-1 s have demonstrated a CV benefit in patients with DM2 that appears to extend to patients with reduced renal function.
- -
Some arGLP-1 s reduce progression to overt albuminuria, decrease albuminuria during follow-up, and may delay progression of renal dysfunction in DKD, especially in the subgroup with established CKD.
- -
Oral semaglutide is effective, safe and well tolerated in patients with DM2, even in those with DKD, with the benefits inherent to this family of drugs, which may facilitate the earlier introduction of an arGLP-1 in the management of DM2.
Aleix Cases declares having received a grant, consulting and speaking fees from Vifor Pharma, speaking and consulting fees from Astellas, Astra Zeneca, Bayer and Novo Nordisk; consulting fees from Boehringer Ingelheim, GSK and Otsuka and speaking fees from Amgen and Sanofi (Mexico), but has not received funding in relation to the writing of this article.