Autosomal dominant polycystic kidney disease is a multi-organic hereditary disorder. It is responsible for 7-10% of cases of end stage renal failure. It is caused by mutations in the genes PKD1 and PKD2. The diagnosis of this disease can be performed through ultrasounds, but the molecular diagnosis offers some advantages, such as the early detection of asymptomatic individuals who carry this genetic defect, in order to perform a preventive monitoring and genetic counselling. In this work, we present the results of the clinical analysis of 48 patients diagnosed with autosomal dominant polycystic kidney disease. The objectives of this work were to analyze the main clinical aspects of the disease. The average age of appearance of the first symptoms was 41.17 ± 13.41 years in women and 49.91 ± 12.52 years in men (p <0.05). Arterial hypertension was the first sign of the disease (68.42%), with more cases in men than in women (p <0.05), followed by chronic renal failure (68.29 %). The most common renal symptom during the evolution of the disease was chronic renal failure, which was present in all the patients of the study, followed by proteinuria (92.31%), end-stage renal failure (89.58%) and arterial hypertension (87.23%). In summary, our results reveal a high prevalence of patients with polycystic kidney disease who received a late diagnosis. This could possibly explain the high morbi-mortality associated to this condition. Given the high prevalence of chronic renal failure and end-stage renal failure secondary to polycystic kidney disease in our study, the early diagnostic of the disease would carry better pronostic in relation with a more strict clinical follow-up. The arterial hypertension was the most frequent clinical manifestation of the disease in our study by what this entity should be included in all the hypertense patients of unknown etiology and on the other hand, the infectious complications should be a sign of alert in every patient with polycystic kidney disease.
La poliquistosis renal autosómica dominante es una enfermedad hereditaria multiorgánica, responsable del 7-10% de los casos de insuficiencia renal crónica terminal que precisan tratamiento renal sustitutivo, causada por mutaciones en los genes PKD1 y PKD2. El diagnóstico de esta enfermedad puede realizarse fácilmente mediante pruebas radiológicas; la ecografía constituye el método de elección, pero el diagnóstico molecular ofrece la ventaja de la detección precoz de individuos asintomáticos portadores del defecto genético. En este trabajo, presentamos los resultados del análisis clínico de 48 pacientes diagnosticados de poliquistosis renal autosómica dominante. Los objetivos de nuestro trabajo fueron analizar los principales aspectos clínicos de la enfermedad, las causas de morbimortalidad e identificar a los individuos de riesgo afectados y sus manifestaciones clínicas precoces. En nuestro estudio, la hipertensión arterial fue la manifestación inicial más frecuente (68,42%), mientras que en la evolución de la enfermedad lo fue la insuficiencia renal crónica (100%). A pesar de que la edad media del diagnóstico de la poliquistosis renal en este estudio fue menor en las mujeres, la evolución de la enfermedad fue más tórpida en los hombres, lo que determinó el inicio más precoz del tratamiento renal sustitutivo y, consecuentemente, la mayor mortalidad. En este estudio se observó una prevalencia similar de muertes de origen cardiovascular (42,1%) e infeccioso (42,1%). En resumen, nuestros resultados revelan una alta prevalencia de pacientes con poliquistosis renal diagnosticados tardíamente, lo que podría explicar la elevada morbimortalidad. Dada la alta prevalencia de insuficiencia renal crónica e insuficiencia renal crónica terminal secundaria a poliquistosis renal en nuestro estudio, el diagnóstico precoz de la poliquistosis conllevaría un mejor pronóstico en relación con un seguimiento clínico más estricto. Por tanto, al ser la hipertensión arterial la manifestación clínica más frecuente en el momento del diagnóstico, debería incluirse esta entidad nosológica en todos los casos con hipertensión arterial de etiología no filiada y, por otra parte, las complicaciones infecciosas deberían ser un signo de alerta en todo paciente con poliquistosis renal autosómica dominante.
INTRODUCTION
Autosomal dominant polycystic kidney disease (ADPKD) is a hereditary multiorgan disease characterised by the progressive growth and development of renal cysts that destroy functional parenchyma. It affects one in every 1,000 people and is responsible for the 7-10% of cases of chronic kidney disease which require renal replacement treatment.1 ADPKD is caused by a mutation in the PKD1 gene, responsible for 85-90% of cases, in the PKD2 gene, responsible for 10-15% of cases and, possibly in a third gene, PKD3, which has not yet been identified.2-4
ADPKD is a multisystemic disease, with renal and extrarenal manifestations derived from the formation of renal cysts and, in many cases, asymptomatic cysts in the liver and pancreas.5 The main renal symptoms are abdominal or lumbar pain, haematuria, arterial hypertension (AH) and, less frequently, urinary infection, lithiasis, renal failure and palpable renal mass.6 The main extrarenal manifestations are cerebral aneurysms, hepatic cysts, cardiac valve disease, colonic diverticula and abdominal hernias.7,8
Diagnosis of ADPKD is usually established by means of imaging techniques; renal echography is the method of choice in patients over 30 years of age. In younger patients computed tomography (CAT) or nuclear magnetic resonance (NMR) should be used.9 In patients under 30, diagnostic certainty is offered by genetic study which provides additional information in genetic counseling.10,11
In this study, our purpose was to analyse the clinical characteristics of all ADPKD patients who required medical attention in our centre during the 1994-2005 period.
MATERIAL AND METHODS
Study population
We carried out a clinical study of 48 ADPKD patients who required medical attention in the Nephrology Service of the University Hospital of Salamanca between 1994 and 2005. The patients had been diagnosed with ADPKD according to clinical and radiological criteria.
Clinical follow-up
The clinical aspects of 48 patients diagnosed with ADPKD who required medical intervention were analysed by our service through the eleven years from 1994 to 2005, and they were followed-up clinically until March 2008. The information on all the patients was taken from their clinical records. The principal parameters analysed were prevalence and age of onset of the disease, aspects related to its initial clinical manifestations, to the clinical manifestations that appeared during evolution, to extrarenal manifestations and to the survival and morbidity and mortality of patients diagnosed with ADPKD.
Statistical method
The statistical study was conducted with the SPSS® software package for Mac® OS X (version 13.0). Qualitative variables were expressed as percentages and numeric variables as mean and standard deviation (X ± SD).
The statistical method used was the Student’s t-test to compare hypothesis on means in populations with normal distribution. When the principle of normality was not met the Mann-Whitney non-parametric statistical test was used. The relation between the qualitative variables of two or more categories was determined by proportion using the Chi-squared test.
Independently of the analysis and the nature of the variable, probability values of less than 0.05 (p < 0.05) were considered to be significant.
RESULTS
48 ADPKD patients, 21 women and 27 men, were clinically studied. The average age of onset of the first symptoms was 46.07 ± 13.49 years, 41.17 ± 13.41 years in women and 49.91 ± 12.52 years in men (p < 0.05). 41.18% of the women had the first manifestations of the disease between 30 and 40 years of age while diagnosis in men was fundamentally between 50 and 60 years.
No statistically significant differences were found in the prevalence of the disease according to the sex of the patient.
Initial clinical renal manifestations
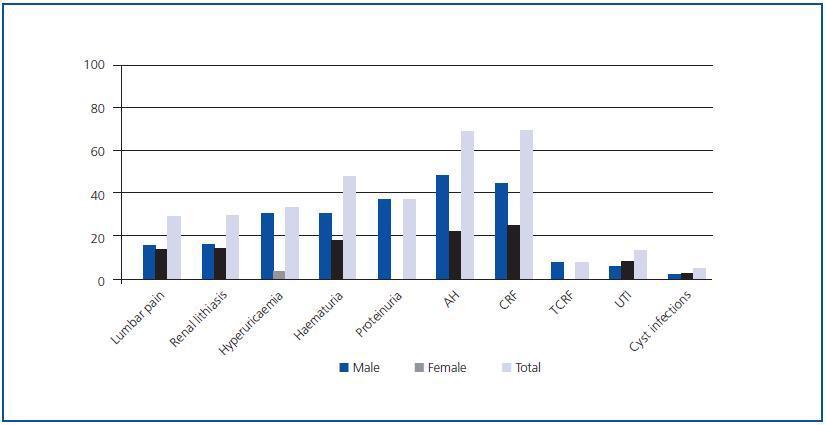
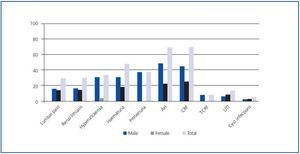
As an isolated manifestation, AH was the most frequent disorder and the initial sign of the disease in 68.42% of the patients, predominantly in men with respect to women (p < 0.05), followed by chronic renal failure (CRF), present in 68.29% of patients with ADPKD (Figure 1).
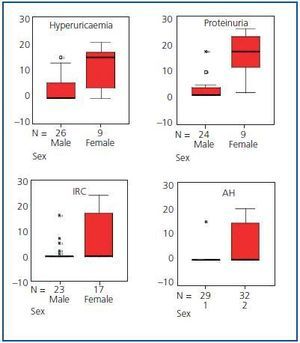
No statistically significant differences were found in the prevalence of the different initial clinical renal manifestations by gender, with the exception of AH, proteinuria, hyperuricaemia and renal lithiasis in which the prevalence was greater in men (p < 0.05) than in women. Quantification of proteinuria at the time of diagnosis was always less than 300mg/24 h.
Clinical renal manifestations during evolution
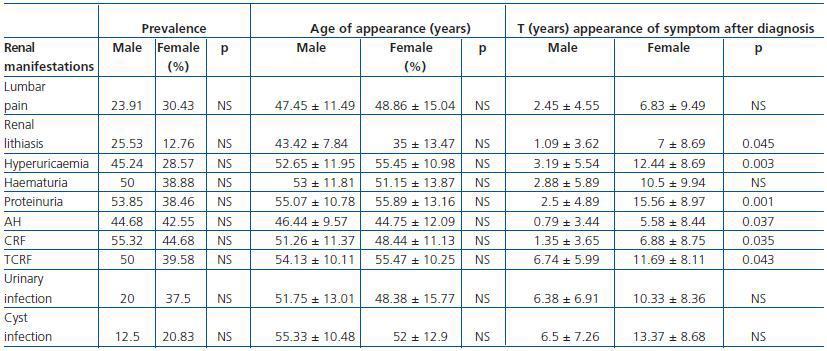
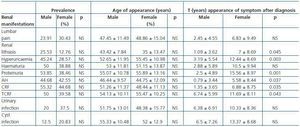
The most frequent renal manifestation during evolution was CRF, present in 100% of the studied patients, followed by proteinuria (92.31%) and terminal chronic renal failure (TCRF) (89.58 %) (Table 1). AH was present in 87.23% of the sick patients, statistically significant in the 30-60 age group (p < 0.01). The prevalence of renal manifestations during evolution showed no significant differences between men and women (Table 1).
Although the prevalence of urinary tract infections was higher in women, it had no statistical significance. Enterobacteria were responsible for 94.12% of the episodes and Escherichia coli was the most frequently isolated aetiological agent (70.59%).
The mean age at CRF onset was 50.02 ± 11.21 years, and that of TCRF 54.74 ± 10.07 years, independently of sex (p > 0.05) (Table 1). Patients diagnosed with CRF were found predominantly in the 30-60 age group (p < 0.05), as were patients with TCRF, in whom the difference was not statistically significant. When the disease was transmitted by the father, the mean age of onset of CRF and of TCRF was lower (p < 0.01).
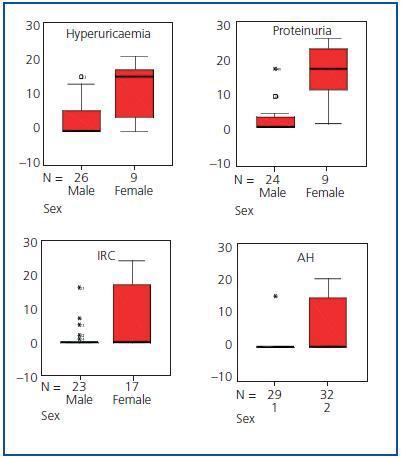
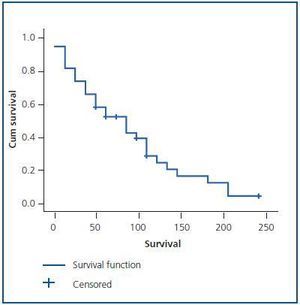
The evolution time to CRF and TCRF (Figure 2) from diagnosis of the disease was shorter in men than in women, as was the elapsed time from diagnosis of CRF to the start of renal replacement therapy (p < 0.05) (Table 1 and Figure 3). Patients with AH were more likely to exhibit CRF (p < 0.05).
Of all the patients who started renal replacement therapy, 83.72% were hypertensive and 13.95% normotensive. Patients with AH, proteinuria, macroscopic haematuria, lithiasis, abdominal pain, urinary tract infections, hepatic cysts and cyst infections were more likely to exhibit TCRF (p < 0.01). However, the probability of developing TCRF did not increase in patients with diverticula, macroscopic haematuria before 30 years of age or women with more than 3 children (p > 0.05).
No significant differences were found by sex in the age of onset of any of the renal manifestations (Table 1).
In addition to CRF and TCRF, the time of evolution from disease diagnosis to the appearance of hyperuricaemia, proteinuria, renal lithiasis and AH was also less in men than in women (p < 0.05) (Table 1 and Figure 3). No significant differences were found in the evolution period from diagnosis of the disease to the appearance of the other renal manifestations.
The recurrence of renal lithiasis was statistically significant in men.
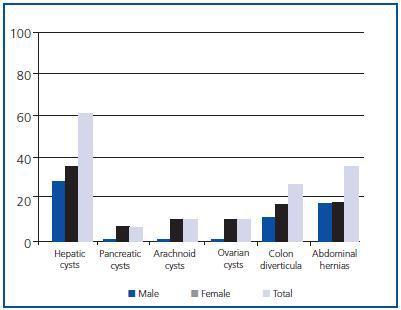
Extrarenal manifestations
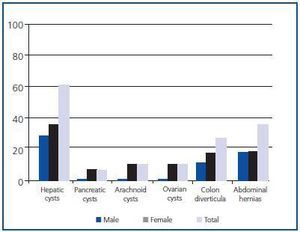
Hepatic cysts were the most frequent extrarenal manifestation, present at the time of diagnosis in 59.09% of patients, with greater prevalence in women (p < 0.05) (Figure 4). No statistically significant differences by sex were found in the prevalence of the other extrarenal manifestations diagnosed.
Survival and morbidity and mortality
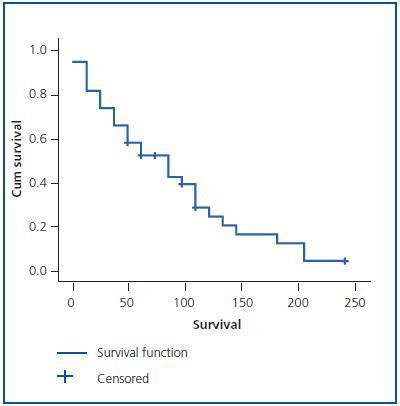
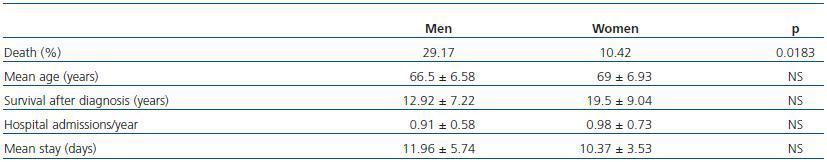
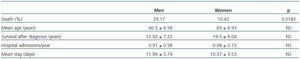
Patients with ADPKD had a total of 6.17 ± 5.81 hospital admissions in our centre during the length of the study, 0.94 ± 0.63 admissions/year, with an average stay of 11.27 ± 4.91 days. No significant differences by sex were found in survival, or in the number of hospital admissions per year, nor in the average stay (Table 2).
Of the total number of patients with ADPKD, 39.58% died during the study, 29.17% men and 10.42% women (p < 0.05; RR = 2.3). Patient survival from the time of diagnosis was 13.4 ± 7.79 years, but it was not shown that AH, macroscopic haematuria, abdominal pain, cyst infections, renal lithiasis, urinary infections, the presence of hepatic cysts or a large number of childbirths were risk factors for mortality (p > 0.05).
The principal causes of death were of cardiovascular (42.1%) and infectious (42.1%) origin.
DISCUSSION
CLINICAL DIAGNOSIS
ADPKD is diagnosed in the majority of patients by characteristic clinical manifestations which usually appear in the third or fourth decade of life. In our study, the mean age of onset of the first clinical manifestations of ADPKD was earlier in women than in men. Studies published to date have not analysed the existence of differences by sex in age at disease diagnosis or in the prevalence of the majority of renal and extrarenal manifestations.1,4,7,9,12
In this series, the most frequent initial manifestation at diagnosis was AH, present in 68.42% of patients, with predominance in males and with a mean age of onset (45.77 ± 10.48 years) later than that described in the literature, with a mean age of 30 years;13,14 nevertheless, in our study it preceded measurable changes in renal function by several years.1,13,15 AH is, according to different publications, one of the guiding signs for ADPKD diagnosis, which is confirmed by this study. The fact that AH diagnosis in our population was later than that published1,14,16 could be explained by the current saturation of general medical consultations and by the lack of protocolised studies which clarify AH aetiology when it is light or easy to control; cases of ADPKD would thus be disguised among those in which early and vigorous treatment of complications would delay the onset of CRF and the progression of TCRF. The high prevalence in our study of CRF and TCRF secondary to ADPKD is in accordance with the ways the disease presents. During the evolution of ADPKD, AH was present in 87.23% of our patients, with no differences observed between the sexes. Evolution time from disease diagnosis to the appearance of AH was less in men. Its role in the progression of renal failure,13,17 already described, was demonstrated; we were unable to confirm this in patients under 35 given the low prevalence of young patients in our study. It has been proved, both in the general population and in sufferers of ADPKD that the prevalence of AH is significantly greater in men up to 45 years of age, at which point, although still prevalent in men, the differences are no longer significant. The shorter evolution time from disease diagnosis to AH onset in men may be due to the existence of cardiovascular risk factors (smoking habit, dyslipidaemia, central obesity) that favour the development of AH and that predominate in males between the ages of 20 and 55. From there on, hormonal influences, dyslipidaemia or central obesity cause these factors to increase significantly in women and, as a result, so does AH.14
In this series, proteinuria was one of the most frequent renal manifestations of ADPKD at the time of diagnosis, following AH, renal failure and haematuria. It was the presenting sign of the disease in 36% of the men, statistically significant with respect to the women, and the elapsed time from disease diagnosis to onset of proteinuria was shorter in men. AH and proteinuria present at the time of diagnosis has been related to worse renal prognosis and increased cardiovascular risk in ADPKD.17,18 Given that AH is correlated to the onset of proteinuria in various publications,19 the greater prevalence of AH in men at the time of diagnosis in this study would also explain the greater prevalence of proteinuria. Furthermore, AH, dyslipidaemia, central obesity and tobacco habit increase the risk of proteinuria and predominate in middle-aged men. During disease evolution, proteinuria becomes the second most frequent renal manifestation (92.31%) with no gender difference, which would explain the high percentage of patients with established kidney damage. The quantification of proteinuria was always lower than 300mg/dL, which agrees with that already described19 and there were no cases of nephrotic-range proteinuria. In the ADPKD patients with proteinuria studied, it was shown that they were more likely to present TCRF, in relation to secondary glomerular hyperfiltration.
Haematuria was a symptom presenting the disease in 47.06% of the cases, and was present in the evolution of the disease in 88.89% of our ADPKD patients. This prevalence was superior to that reported in literature, where the figure is 35-50%.1 Haematuria could be due to renal calculus, cyst infection or malignant tumours. The elevated prevalence of renal lithiasis and cyst infections in our series explains the higher prevalence of haematuria than that described. The underlying association of macroscopic haematuria with the progression of renal failure17,20 was evident, but that role was not demonstrated in patients under 30 probably due to the low prevalence of young patients with ADPKD. This deleterious effect of haematuria on kidney function can be explained both by the obstructive episodes secondary to clot formation and the hypovolaemic situations that it provokes.
The prevalence of urinary tract infections in our study was similar to that described by others. As in the general population, because the female urethra is shorter, prevalence was higher in women, but was not statistically significant.12,16E. coli was the most frequently isolated aetiological agent. The most frequent infections were cyst infections and acute pyelonephritis, with a determinant role in the progression of renal failure, probably in relation to the deterioration of kidney function secondary to episodes of haemodynamic instability derived from severe infection.
CRF was the second most frequent clinical manifestation at diagnosis and that of greatest prevalence in the evolution of the disease in our study, while in other series it is estimated that only around half of the patients with mutation will develop the disease before the sixth decade of life.15 Also, the need for renal replacement treatment in our work was higher than that described.18 The mean age of onset of CRF (50.02 ± 11.21 years) and TCRF (54.74 ± 10.07 years) was similar to that described in the literature for patients with PKD1 gene mutation and requiring renal replacement treatment (54.3 years),3,4 which leads us to assume a greater prevalence of patients in our population with PKD1 gene mutation if we limit ourselves to a clinical analysis. The age of onset of TCRF was earlier when transmitted by the father than by the mother and evolution time to CRF and TCRF from disease diagnosis was shorter in men than in women, a fact already reflected in different studies.3,21 In this study, men had more probability of rapid progression to renal failure, which appears to be related to a direct effect of the sexual hormones.21,22 Another sign of poor prognosis in the early evolution to both CRF and TCRF was the disease being inherited from the father.
As far as extrarenal manifestations,8 these predominated in women, although only hepatic cysts were statistically significant: the greater prevalence of these in women has been associated with the number of pregnancies, the taking of contraceptive pills and replacement hormonal therapy. Although there are contradictory results, we confirm that people with hepatic cysts have worse renal function than those who do not,3,16 probably due to the prejudicial effect that repeated infection in hepatic cysts would have on renal function.
In this study, a similar prevalence was observed in deaths of cardiovascular (42.1%) and infectious (42.1%) origin. Before 1975, the most frequent cause of death in ADPKD patients was infection (30%), uraemia (28%) and cardiovascular disease (21%). With the development of dialysis, the main cause of mortality has become cardiovascular (34%) followed by infections (20.4%).23 The principal cause of death of patients in renal replacement treatment and those subjected to renal transplant is cardiovascular; nevertheless, in our study there was a markedly high prevalence of deaths of infectious origin, which could be attributed to the immunodeficiency situation derived from a population with very high prevalence of TCRF secondary to ADPKD. The death rate was higher in men, but no significant differences were found in survival.
In spite of the mean age of ADPKD diagnosis in this study being lower in women, evolution up to the appearance of renal lithiasis, hyperuricaemia, proteinuria, AH and TCRF was shorter in men. This, together with the greater prevalence in men, at diagnosis, of risk factors involved in the progression towards TCRF, determined a more torpid evolution of the disease in men, an earlier start of renal replacement treatment and, consequently, greater mortality. In women, however, the evolution time of the disease was longer, which translated to a higher number of hospital admissions. We may think that in women morbidity predominates and in men, mortality, and it is probably not a reflection of the disease but an observation common to the entire population.
In short, in our study, male carriers of ADPKD were diagnosed later than women, which could explain their worse clinical evolution. In this population, AH was the most frequent clinical manifestation at diagnosis, and this classification should, therefore, be included in all cases with unknown AH. Also, infectious and cardiovascular complications were the principal cause of mortality, implying that infectious complications are a sign of alert in all ADPKD patients, including those not subjected to transplant.
Figure 1.
Table 1. Prevalence, age of appearance and elapsed time from disease diagnosis to the appearance of the different renal clinical manifestations by sex
Figure 2.
Figure 3.
Table 2. Prevalence and statistical significance by sex of deceased patients, mean age at time of death, survival since diagnosis, hospital admissions and mean stay
Figure 4.