Objetivos: El síndrome de robo es una complicación grave del acceso vascular. Nuestro objetivo es presentar los resultados iniciales de un tratamiento simple y eficaz: la interposición de un segmento protésico en la vena yuxtaanastomótica. Métodos: Entre 2009 y 2012, 14 pacientes (57 % varones, edad media 71 años) con síndrome de robo severo por un acceso vascular nativo (grados II-IV), y tras un estudio clínico y ecográfico sistemático, y angiográfico selectivo, fueron tratados mediante la interposición de un segmento de prótesis tubular, politetrafluoroetileno (PTFE) de 6 mm de diámetro en la vena yuxtaanastomótica, más reparación selectiva de estenosis arterial mediante parche (2 casos) o ligadura de colaterales venosas no útiles (8 casos). Se utilizó anestesia local o regional y un régimen ambulatorio en todos los casos. Resultados: El éxito técnico fue del 100 %. El estudio ecográfico pre y posoperatorio mostró una reducción del flujo posoperatorio del acceso del 39 % y un aumento del flujo arterial radial del 477 %. Los síntomas isquémicos se resolvieron en 12 pacientes (86 %); los otros dos requirieron procedimientos adicionales por síntomas isquémicos persistentes. Ocurrió una ruptura venosa posoperatoria, que requirió una ligadura definitiva. Ningún caso sufrió amputaciones ni hubo pérdidas ni trombosis del acceso en el seguimiento. Las permeabilidades primaria y primaria asistida libres de nuevos síntomas isquémicos fueron del 78 % y del 78 % a los 12 meses, y del 62 % y del 78 % a los 24 meses. Conclusiones: La interposición de un segmento protésico yuxtaanastomótico es una técnica sencilla, rápida y eficaz en el tratamiento del síndrome de robo vascular, con resultados prometedores a dos años de seguimiento.
Objective: Steal syndrome is a severe complication of vascular access. Our aim is to present the initial results of a simple and effective treatment: the interposition of a prosthetic segment in the juxta-anastomotic vein. Method: Between 2009 and 2012, 14 patients (57 % male, average age 71) with severe steal syndrome due to vascular access (stages II-IV), following a clinical and systematic echographic study and a selective angiographic study, were treated through the interposition of a 6 mm segment of PTFE in the juxta-anastomotic vein, in addition to selective patch repair of arterial stenosis (2 cases) or ligature of useless venous collaterals (8 cases). Local or regional anaesthesia was used and outpatient care was given in all cases. Results: There was 100 % technical success. The pre- and post-operative echographical study showed a reduction of post-operative access flow by 39 % and an increase of radial artery flow by 477 %. Ischaemic symptoms were resolved in 12 patients (86 %); the other two patients required additional procedures due to persistent ischaemic symptoms. There was a post-operative venous rupture, which required a definitive ligation. No patient suffered amputations, nor were there losses or access thrombosis during the follow-up. The primary and primary-assisted patencies free of new ischemic symptoms were 78 % and 78 % at 12 months, and 62 % and 78 % at 24 months. Conclusions: The interposition of a prosthetic segment in the juxta-anastomotic vein is a simple, quick and effective technique in the treatment of vascular steal syndrome, with promising results at the 2 year follow-up.
INTRODUCTION
Arteriovenous fistula (AVF) is the method of choice for vascular access during haemodialysis sessions in patients with end-stage renal failure, because, compared with prosthetic arteriovenous or central venous catheters, it is associated with lower complication rates.1,2 However, there is a specific complication of AVF or prosthetic grafts that can be severe and even jeopardize the viability of the affected limb: steal syndrome.
Steal syndrome is caused by a decrease in distal blood perfusion due to a proximal preferred blood flow outlet through the vascular access vein, with less resistance than the distal arterial bed.3 Despite being a physiological phenomenon observed in up to 73% of AVFs and 91% of prostheses (steal syndrome grade I),4 it can cause symptoms in up to 8% of accesses5: pain during dialysis (grade II), pain at rest (grade III) or ulceration and necrosis (grade IV).6 When these symptoms occur, especially grades III and IV, endovascular or surgical procedures are required to restore distal blood flow and prevent irreversible injury or amputation, maintaining patency and the use of the vascular access, if possible.
The treatments proposed for steal syndrome include a variety of techniques: ligature (usually effective, but resulting in access loss), repair of associated arterial lesions proximal or distal7, ligature of the distal radial artery (DRAL)8 or proximal vascular access (PRAL)9 in steal cases at the palmar arch in distal cephalic fistulae, proximalisation or distalisation of the anastomosis (PAVA and RUDI)10,11, flow reduction techniques (such as banding, with unpredictable results if performed without intraoperative monitoring, or variants of it, such as MILLER12, external banding with prosthetic bands13,14 or simple ligature of non-useful collateral veins15,16), or DRIL3,17 (distal revascularization by interposition of an arterial bypass and ligature of the native artery). The results offered by these techniques are variable, maintaining access and resolving ischemic symptoms in up to 77% of cases treated with DRIL. However, many of them are complex and induce morbidity.3
The aim of this study is to present the initial results of a simple and effective technique to treat steal syndrome: reduction of vascular access flow (and increased distal arterial perfusion) by interposing a prosthetic graft segment in the juxta-anastomotic vein.
MATERIAL AND METHOD
Our vascular access unit (UFAV, Hospital Clínic, Universidad de Barcelona; Barcelona, Spain) offers a multidisciplinary approach to patients requiring vascular access for haemodialysis, and is a referral centre for patients in our health district and other centres with regards to creation and repair of vascular access. All patients evaluated and treated by our unit are systematically entered prospectively into a multidisciplinary database, which collects basic data, comorbidities, previous vascular accesses, a detailed history and physical and routine ultrasound examinations. All surgical procedures are also noted, once again with a physical examination and intraoperative and immediate postoperative results as also postoperative follow-up.
For this study, we included all patients that came to our unit between 2009 and 2012 for symptomatic steal syndrome secondary to a native arteriovenous vascular access performed in our unit or in other centres. Symptomatic steal syndrome was defined as pain at dialysis, constant pain or ulceration (grades II, III and IV) in the hand or forearm of the vascular access, usually associated with other signs of hand ischemia (pallor, coldness) and after ruling out other differential diagnoses by physical examination and selective complementary examinations (carpal tunnel syndrome, joint pain, high venous pressure, etc.).
During the study period, our unit evaluated 51 patients with persistent symptomatic steal syndrome. After an individual assessment and discussion of each case, a physical examination, routine ultrasound, and selective angio-radiological study, we excluded from the series renal transplant patients with normally functioning kidneys (they underwent access ligature), subsidiary accesses to be repaired with other techniques (proximal or distal arterial stenosis treated with endovascular procedures, distal accesses treated by DRAL, simple ligature of useless venous collaterals in proximal accesses), useless accesses, presence of severe distal artery diseases, those cases associated with very extensive ulcerated lesions, poor health conditions or patient preference.
In brachio-basilic side to side fistulae with several output drainage veins (basilic, elbow perforating and/or cephalic vein), we chosed to perform a simple ligature of useless venous collaterals when the diameter of one of these was greater than 4 mm, and to add a prosthetic interposition to the useful vein (in addition to useless collaterals ligature) when said collaterals were all smaller. We dismissed making this decision based on flow, given the difficulty and unreliability of calculating this with ultrasounds, especially for the perforating elbow vein.
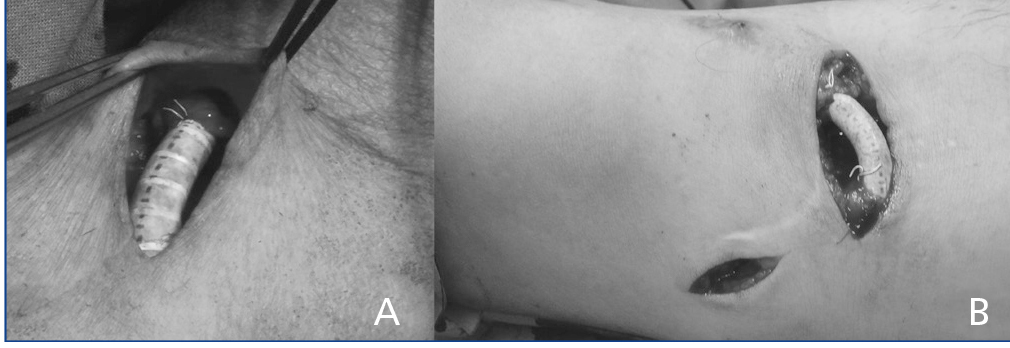
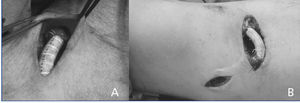
Finally, 14 cases were included, which were treated with the prosthetic interposition technique. A segment of straight tubular 6 mm prosthesis made of polytetrafluoroethylene (PTFE) graft, 2cm long, was interposed in the juxta-anastomotic venous segment by end to end anastomosis of both venous stumps (Figure 1). Useless venous collaterals (less than 4mm in diameter) were ligated in side to side brachio-basilic AVF, and when the preoperative study (ultrasound and/or angiography) showed perianastomotic artery stenosis, this was repaired by patch angioplasty, in addition to interposition of the juxta-anastomotic prosthetic segment.
In addition to patients’ basic and clinical preoperative, the following were collected in a prospective database:
Preoperative Examination: physical exam (grade of steal syndrome), ultrasound examination (prior vascular access type, location, venous drainage, arterial and venous diameters, systolic, diastolic and mean speeds in the proximal artery [2cm proximal to the anastomosis] and distal radial artery in the wrist).
Intraoperative Examination: procedure performed, immediate physical examination (pulse, palpable thrill, murmur at access, distal arterial pulse) and ultrasound (same preoperative measurements).
Follow-up: postoperative follow-up of clinical symptoms and adequacy of access for haemodialysis sessions, patency and need for secondary procedures.
All surgeries and examinations were performed by the same surgeon, using the same ultrasound device: SonoSite MicroMaxx Ultrasound System (Sonosite Inc, WA, USA) and a HFL38/13-6 MHz 38 mm linear array transducer, adjusting and steering the pulsed wave doppler angle to 60º to vessel direction and gate size to vessel diameter. Intra-operative measurements were recorded in the operating room, during surgery and after anastomosis creation; aseptic conditions were taken into account. These measurements were not considered in surgery decisions. Velocities (PSV, EDV, MV) and RI were automatically calculated, and vessel diameter (adventitia-adventitia) was manually measured. Flows (mL/min) were calculated using manufacturer proposed parameters: cross sectional area (0.785xD2, in cm2) x time averaged mean velocity (MV, in cm/s) x conversion factor (0.06). All pre- and intra-operative measurements were always taken in the same place.Vascular access flow was estimated according to the flow of the proximal artery as the most reliable method,18,19 dismissing vein flow rates (less reliable18,19 and as several draining veins exist which in many cases affect this interpretation). Total distal arterial perfusion flow was not calculated, because there are several drainage distal arteries (radial, ulnar, interosseous) that should be added and due to the technical and computer complexity that this entails, in spite of, changes in distal radial artery flow were calculated and considered an estimate of overall changes in distal perfusion.
Statistics
Frequency and descriptive statistics were obtained, and comparisons were performed using SPSS software, version 19.0, describing medians and ranges or interquartile ranges (25-75 percentiles) and percentages. Changes in flows were defined as percentages of change ([preoperative-postoperative]/preoperative), and were described as medians and interquartile ranges. Statistical differences between groups were evaluated using the Wilcoxon signed rank test for comparisons of pre-postoperative flows. Measures of time to event were analyzed using Kaplan- Meier survival analysis, with estimated percentages at 12 and 24 months. A P value <.05 was considered to be statistically significant.
RESULTS
The 14 patients included in this study (57% male, median age 72 years) had a high percentage of comorbidities (86% hypertension, 78% smoking history, 71% dyslipidaemia, 57% ischaemic heart disease, 43% diabetes mellitus, 29% severe symptomatic peripheral artery disease, 21% chronic obstructive pulmonary disease). All were on renal replacement therapy by hemodialysis for a median of 1.1 years (range 0.2 to 5.1), although the actual arteriovenous access was made 1.5 years earlier (range 0.1 to 12.7), it was a secondary access in 50% of cases. All had symptomatic steal syndrome (grade II: 2, grade III: 8, grade IV: 4).
Original AVF accesses are described in Table 1. All of them were proximal fistulae with a cephalic or basilic drainage vein, or both. As has already been described in side to side brachio-basilic fistulae the elbow perforating vein or other useless veins (proximal basilic or cephalic) were occluded or were under 4mm in diameter. 71% of accesses were in the left arm.
All cases were treated using the juxta-anastomotic prosthetic interposition technique (described above). Arterial anastomotic stenoses were diagnosed in 2 cases and repaired by an additional arterial patch angioplasty. In the 8 cases of side to side, brachio-basilic arteriovenous fistulae, we also carried out a ligature of small useless collateral veins (perforating veins or proximal basilic of less than 4mm in diameter), and in 5 we performed a superficial transposition of the proximal basilic vein. All cases were performed under local or regional anaesthesia and as hospital outpatients without hospital admittance.
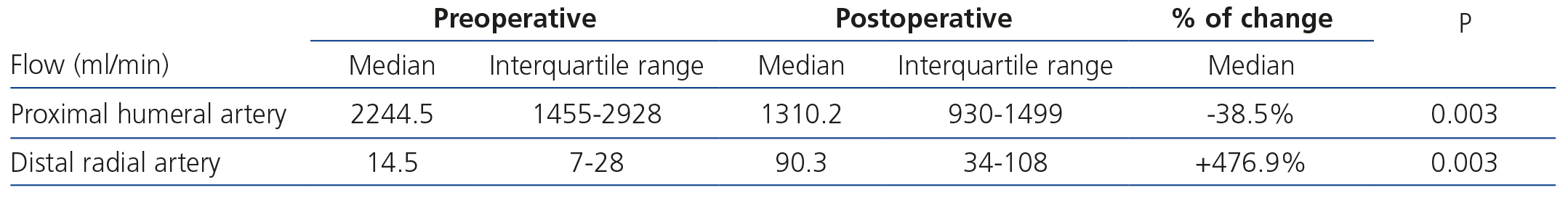
The technical success of the procedure was 100%. Pre- and postoperative ultrasonography showed immediate access flow reduction of 39% (proximal arterial flow) and an increase in the flow of the distal radial artery of 477% (Table 2).
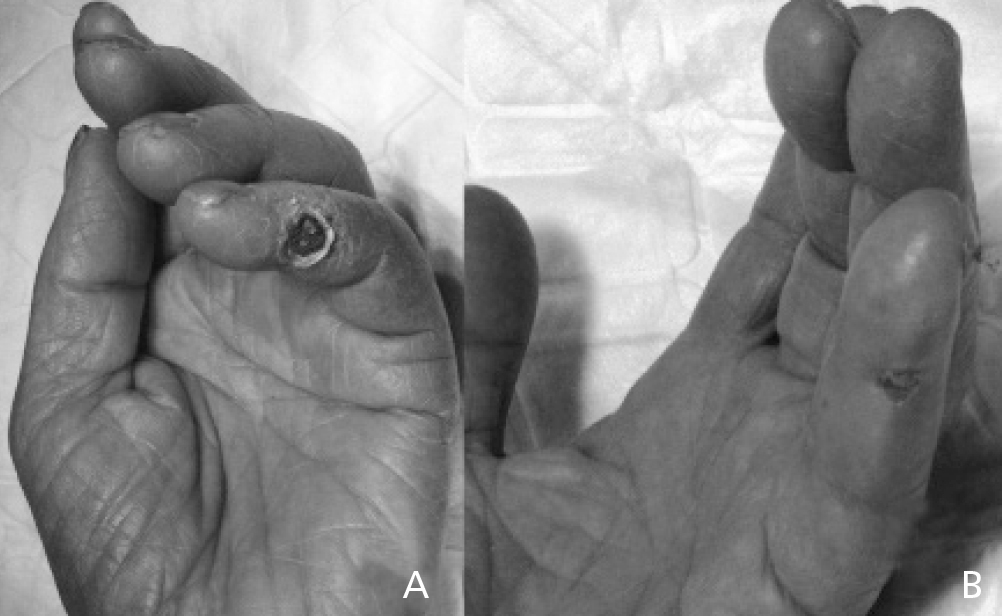
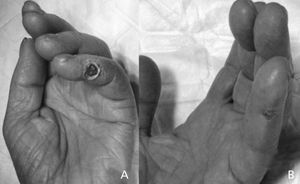
Ischaemic symptoms completely disappeared and ischaemic lesions healed following the prosthetic interposition technique in 12 cases (86%, Figure 2). However, the other two cases showed partial improvement with persistent ischemic pain, eventually requiring access ligature within three months of follow up. Technical failure was attributed to a proximal arterial stenosis not initially diagnosed and to an artery of small calibre and low flow. Both cases, after initial repair, had the lowest increase in distal blood flow and access flow decrease in all the series. The postoperative complication occurred due to rupture of an excessively dilated basilic vein that had undergone superficial transposition, three days after the initial procedure, which required a permanent access ligature. There were no minor or major amputations in any case after surgery or during follow-up.
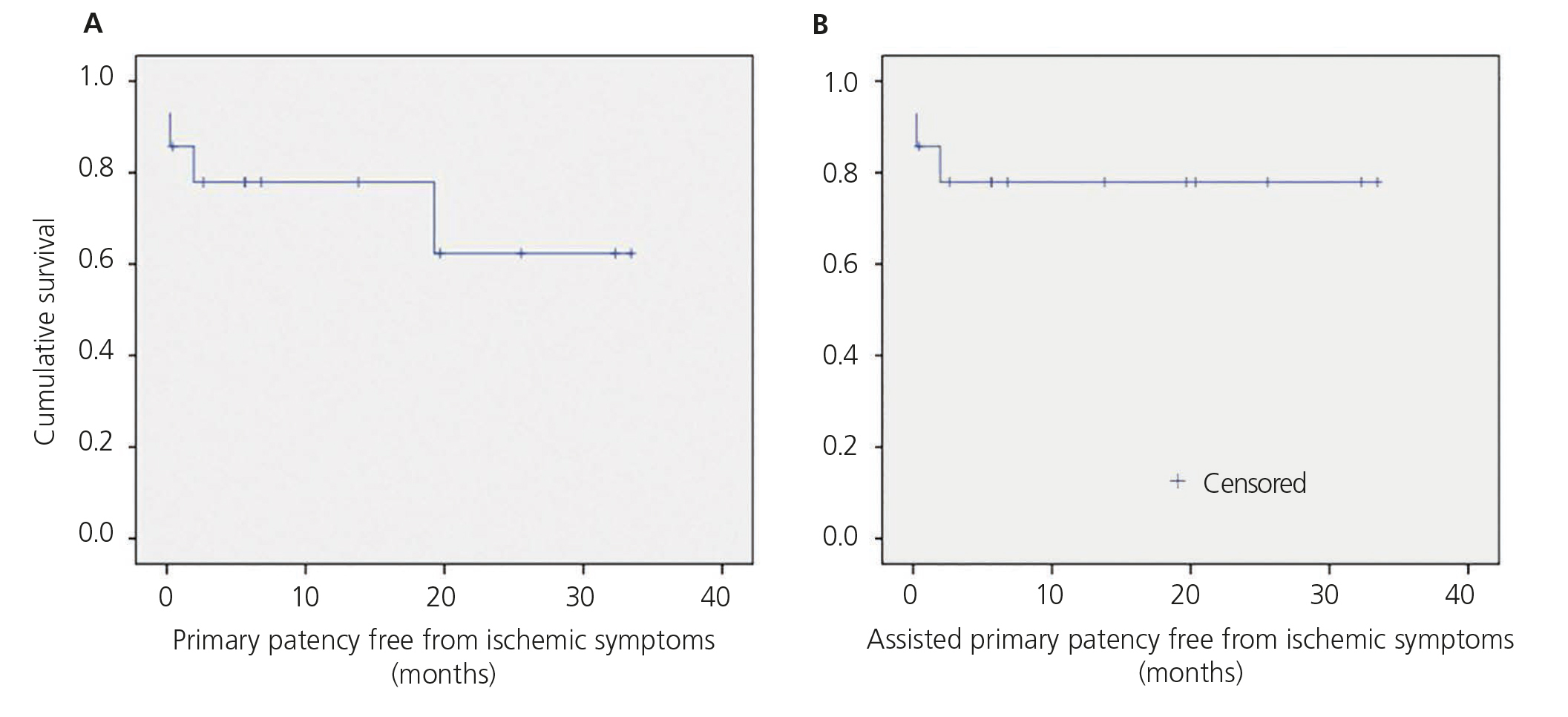
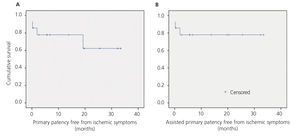
Mean follow-up was 12.04 months (range 0.2 to 33.5). There were no losses to follow-up, there were no new ischaemic symptoms, or access thrombosis and only one reintervention (balloon angioplasty) performed because of stenosis of the interposed prosthesis during follow up. Therefore, primary, assisted primary and secondary patency free from ischaemic symptoms were 78%, 78% and 78% at 12 months and 62%, 78% and 78% at 24 months, respectively (Figure 3).
DISCUSSION
Steal syndrome is caused by a decrease in distal blood perfusion due to a proximal preferred blood flow outlet through the vascular access vein, with less resistance than the distal arterial bed.3 Some cases of steal syndrome can be repaired with specific treatments (proximal or distal arterial stenosis can usually be treated with endovascular techniques, steal syndromes in radio-cephalic fistulae due to palmar arch steal can be repaired by DRAL8 or other syndromes of excess venous output due to large collaterals can be treated with simple non-useful vein ligatures15,16).
However, in many cases of proximal accesses without arterial lesions or large useless collateral veins, steal syndrome is directly due to excess flow from the artery to the venous outflow to the detriment of the distal arterial system. In these cases, the treatment proposed by our group (the juxta-anastomotic vein interposition of a prosthetic 6mm graft segment, with selective repair of arterial stenoses with patch angioplasty and ligature of useless collateral veins) partially reverses this condition reducing the overall fistula flow and increasing distal blood perfusion. These flow changes have been seen by echographic measurement. Overall reduction of access flow by 39% and increase in distal radial artery flow in 477% (it therefore follows that there is a reduction in access flow at the expense of a reduction in venous outflow, thereby increasing distal arterial blood perfusion). Furthermore, results at two years follow-up are promising (assisted primary and secondary patency 78%).
Prosthetic interposition technique is a flow reduction technique (a variant of classical banding, but controlling diameter and length), thus increasing distal arterial perfusion pressure. Other flow reduction techniques have been effective, but have unpredictable results. The most widely used world-wide is banding, which is a partial ligature of the juxta-anastomotic vein. Although effective in some cases, it has been related to non-consistent results and high failure rates due to excessively tight or loose ligatures. To ensure ligature diameter, techniques such as MILLER banding have been designed (ensuring ligature diameter with an endoluminal ball),12 banding assisted by a dilator20 or monitored intraoperatively with flow and digital pressure measurements,21 thus obtaining better results. The use of prosthetic segments as external banding, to reduce vein diameter13,14, has also shown better results than usual banding, however, depending on vein diameter, there are concerns with regards to the risk of stenosis and thrombosis due to excess intra-banding fold tissue.
Other techniques such as PAVA or RUDI10,11 or interposition of long loop prosthetic segments22 are more complex procedures, which use longer prosthetic segments and have an increased risk of thrombosis, but have the same objective as the procedure presented: to reduce access flow by reducing vessel diameter to that of the interposed prosthetic segment, and therefore the same result is obtained.
The most commonly used technique to treat steal syndrome is probably DRIL3,17: using a humeral-humeral arterial bypass proximally to distally of the arteriovenous anastomosis (usually with a segment of saphenous vein) plus ligature of the native artery (between the original fistula and distal bypass anastomosis). Thus, native artery from the origin of the bypass to the arteriovenous anastomosis (of smaller diameter than the bypass, and especially and definitely smaller diameter than the outflow vein) acts like a stenosis with regards to venous outflow. From this perspective, and in spite of other less understandable haemodynamic explanations, DRIL achieves the same objective as prosthetic interposition: it causes a decrease in the size of the vessel connecting the arterial system to the venous outflow (in DRIL: the native artery, and in the interposition technique: a prosthetic segment), to produce a decreased flow from the proximal artery to the vein and increase distal arterial blood flow. However, DRIL uses a more complex technique with greater morbidity than the interposition technique for the same purpose, with the risk that bypass complications may cause arm ischaemia.3 In the interposition technique, procedure occlusion only affects arteriovenous access and not arm viability. Furthermore, DRIL has achieved a percentage of early successes and patency close to 77%, similar to the prosthetic interposition technique.
The three initial complications that occurred in our series could probably have been avoided. To superficially transpose an excessively dilated vein increases the risk of bleeding and should not have been performed, but instead replaced with a prosthetic access. A diagnostic error was responsible for not diagnosing proximal arterial stenosis, which might have been previously treated and so made it possible to avoid prosthetic interposition. And in arteries of small calibre repairs should be performed with smaller diameter prostheses (4mm).
Fistula ultrasound and compression studies showed two cases of arterial stenosis in the distal segment of the original anastomosis. These cases required, in addition to prosthetic interposition, repair by means of an arterial patch angioplasty. Possibly these would have been good candidates for repair using DRIL, saving the arterial stenosis with a bypass, but with this technique both defects were treated: the arterial stenosis and excess vein output flow. Other procedures, such as ligature of useless collateral veins or superficial transposition of a proximal basilic vein are important to complete flow reduction and increase the usefulness of the final access.
As previously described, in side to side brachio-basilic fistulae with useless collateral vein drainage (proximal basilic or elbow perforating veins), a simple ligature of these may be sufficient to resolve steal syndrome.15 But given our previous bad experience with this technique and the difficulty and unreliability when calculating collateral flow with echo-Doppler, especially for perforating elbow vein (hard to measure or selectively compress), we chose to ligate said collateral when they were patent and greater than 4mm diameter. Only when these were smaller, did we add the prosthetic interposition technique to ligature, since simple ligature could be responsible for procedure failure due to insufficient reduction of access flow. Maybe in the future, following the results presented, ultrasound monitoring of intraoperative pre and post ligature flows of simple collaterals (rather than their diameter or systematic simple ligature and clinical follow-up) could help define what side to side accesses require performance of the prosthetic interposition technique.
For this study a portable conventional duplex device for morphologic and flow diameters was used. Despite being very useful, easy to use even under intraoperative conditions and available in most services, estimated flows may be higher and there is a low correlation with other more reliable techniques (ultrasound dilution rate or quantitative speed colour indices); the reason is probably the high susceptibility of conventional Doppler to turbulent flows.23 Precisely for this reason, many groups recommend measuring flow in the proximal artery as an estimate of the extent of access flow, avoiding vein measurements: this bears a better relationship to access flow, has a more laminar flow, with a regular wall, circular area and is more difficult to compress with the probe.18,19 This was the reason for estimating arteriovenous access flow by proximal arterial flow.
There are several limitations to the validity of our study: it is a short series, without a control group, in which comparisons are difficult, and ultrasound measurements may have system errors. Moreover, as already discussed, in some cases in the series simple ligature of collateral veins could have been an alternative treatment to interposition plus collateral ligature.
In conclusion, the interposition of a prosthetic graft segment in the juxta-anastomotic vein, with selective repair of arterial stenosis with patch angioplasty and ligature of useless venous collaterals, can effectively treat symptomatic steal syndrome caused by an arteriovenous vascular access, increasing distal arterial perfusion and reducing the access flow. Despite some avoidable mistakes in our series, this technique is promising at two years follow-up. Its possible indications are symptomatic steal syndromes (grade II to IV) in proximal arteriovenous accesses without associated proximal arterial lesions or large patent useless venous collaterals, and demonstrated hyperflow.
Conflicts of interest
The authors declare that they have no conflicts of interest related to the contents of this article.
Table 1. Description of the types of arteriovenous accesses addressed in the series.
Table 2. Comparison of pre and post operative flows in the proximal humeral artery and distal radial artery (medians and interquartile ranges, and% of change)
Figure 1. Intraoperative images of the interposition of a prosthetic segment , one (B) with additional ligature of non-useful collateral veins (proximal basilic vein).
Figure 2. Ischaemic lesion in 5th finger of the hand, secondary to a steal syndrome due to a brachio-cephalic arteriovenous fistula (A); improvement of lesion 2 weeks after repair using the interposition graft technique (B).
Figure 3. Kaplan-Meier survival function.