The clinical spectrum of COVID-19 ranges from asymptomatic cases to those who develop acute-respiratory distress syndrome and require intensive care unit (ICU) admission.1,2
Previous studies have identified chronic kidney disease (CKD) as a risk factor for severe outcomes following SARS-CoV2 infection.3 This observation derives from data from small cohorts of hospitalized patients mainly from China4–7 and a UK cross-sectional survey describing 16,749 patients hospitalized with COVID-19.8 Recently, the largest nationwide cohort study describing the impact of clinical risk factors on COVID-19 related deaths has been published and reports the negative impact of CKD on mortality among an unselected population of cases with COVID-19.9 Whether the same association is observed among non-hospitalized patients from different regions is yet to be confirmed. We aimed to determine the modifying impact of CKD among a Portuguese nationwide cohort of cases of SARS-CoV2 infection, including those with and without hospitalization.
This was a retrospective analysis from a nationwide prospective registry, including all confirmed (nasal/pharynx swab real-time polymerase chain reaction) cases of SARS-CoV2 infection notified to the Directorate-General of Health from March 02 until April 21, 2020, in Portugal. The country (population of 10.6 million inhabitants) has been using the National Epidemiological Surveillance System (SINAVE) to capture cases of SARS-CoV2 infection occurring nationally (25 administrative regions, 100 hospitals, and 357 primary care centres) since January 01, 2020. CKD was defined according to the physician who notified the case of SARS-CoV2 infection.
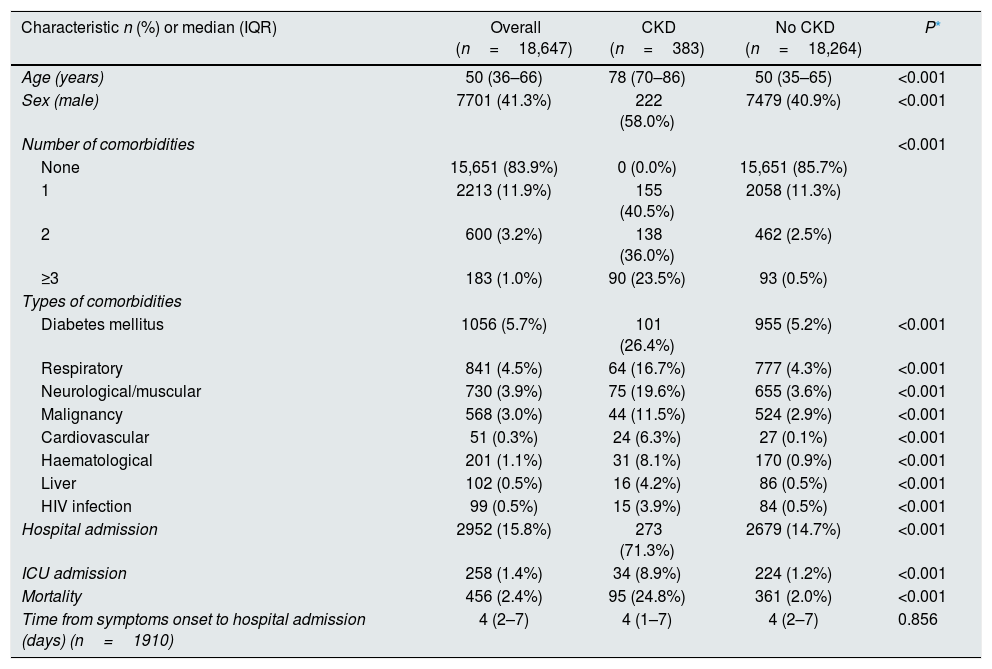
Overall, 18,647 cases were included in our analyses, following exclusion of 1623 (8.0%) cases without hospital admission status and 23 (0.1%) cases without outcome status. Among all cases, median (IQR) age was 50 (36–66) years (Table 1). Male sex accounted for 7701 (41.3%) of all cases. While 15,651 (83.9%) cases did not have any comorbidity, the remainder of cases had the following number of comorbidities: one in 2213 (11.9%) cases, 2 in 600 (3.2%) cases, and ≥3 in 183 (1.0%) cases. Median (IQR) follow-up was 27 (19–33) days. Peak ICU bed occupancy rate occurred on April 06 (58.1% of national standard official capacity). Median (IQR) time from symptoms onset to hospital admission was 4 (2–7) days. Overall, 2952 (15.8%) or 258 (1.4%) cases required hospital or ICU admission, respectively. All-cause mortality occurred in 456 (2.4%) cases. There were 687 (3.7%) cases admitted to the ICU or deceased.
Baseline characteristics stratified by the presence of chronic kidney disease.
| Characteristic n (%) or median (IQR) | Overall (n=18,647) | CKD (n=383) | No CKD (n=18,264) | P* |
|---|---|---|---|---|
| Age (years) | 50 (36–66) | 78 (70–86) | 50 (35–65) | <0.001 |
| Sex (male) | 7701 (41.3%) | 222 (58.0%) | 7479 (40.9%) | <0.001 |
| Number of comorbidities | <0.001 | |||
| None | 15,651 (83.9%) | 0 (0.0%) | 15,651 (85.7%) | |
| 1 | 2213 (11.9%) | 155 (40.5%) | 2058 (11.3%) | |
| 2 | 600 (3.2%) | 138 (36.0%) | 462 (2.5%) | |
| ≥3 | 183 (1.0%) | 90 (23.5%) | 93 (0.5%) | |
| Types of comorbidities | ||||
| Diabetes mellitus | 1056 (5.7%) | 101 (26.4%) | 955 (5.2%) | <0.001 |
| Respiratory | 841 (4.5%) | 64 (16.7%) | 777 (4.3%) | <0.001 |
| Neurological/muscular | 730 (3.9%) | 75 (19.6%) | 655 (3.6%) | <0.001 |
| Malignancy | 568 (3.0%) | 44 (11.5%) | 524 (2.9%) | <0.001 |
| Cardiovascular | 51 (0.3%) | 24 (6.3%) | 27 (0.1%) | <0.001 |
| Haematological | 201 (1.1%) | 31 (8.1%) | 170 (0.9%) | <0.001 |
| Liver | 102 (0.5%) | 16 (4.2%) | 86 (0.5%) | <0.001 |
| HIV infection | 99 (0.5%) | 15 (3.9%) | 84 (0.5%) | <0.001 |
| Hospital admission | 2952 (15.8%) | 273 (71.3%) | 2679 (14.7%) | <0.001 |
| ICU admission | 258 (1.4%) | 34 (8.9%) | 224 (1.2%) | <0.001 |
| Mortality | 456 (2.4%) | 95 (24.8%) | 361 (2.0%) | <0.001 |
| Time from symptoms onset to hospital admission (days) (n=1910) | 4 (2–7) | 4 (1–7) | 4 (2–7) | 0.856 |
IQR: interquartile range. HIV: human immunodeficiency virus. ICU: intensive care unit.
Cases with CKD (n=383; 2.1%) had higher median (IQR) age [78 (70–86) vs. 50 (35–65) years; P<0.001), were more frequently men (58.0% vs. 40.9%; P<0.001) and had higher burden of disease (any comorbidity 100% vs. 14.3%; P<0.001) compared to those without CKD.
All types of comorbidities were more frequently reported in cases with CKD compared to those without CKD (Table 1).
Cases with CKD had higher odds of hospital admission [OR 14.4 95%CI (11.5–18.1)], ICU admission [OR 7.9 95%CI (5.4–11.4)], mortality [OR 16.4 95%CI (12.7–21.1)] and composite endpoint of mortality or ICU admission [OR 14.0 95%CI (11.1–17.7)] compared to cases without CKD. In multivariable analysis with logistic regression, only higher age [aOR (95%CI) 1.02 (1.01–1.04), P=0.01] was associated with higher risk of death or ICU admission among the population with CKD, irrespective of gender [male gender aOR 1.07 (0.69–1.68), P=0.76] or number of comorbidities [≥2 vs. 1 (ref) aOR 1.21 (0.76–1.92), P=0.42]. Interestingly, median (IQR) age (years) of cases with CKD [78 (70–86)] was similar to cases that were admitted to the ICU or died [80 (69–87)] among the entire population.10
The interpretation of our findings should consider the following limitations. Firstly, the absence of a prespecified definition of CKD or serum creatinine measurements might have excluded from our analysis patients with earlier stages of the disease and interfered with the measurement of the strength of the association between CKD and clinical outcomes. However, the standardized national electronic reporting system through SINAVE has likely minimized such limitation. Secondly, we reported no laboratory data to characterize SARS-CoV2 infection among CKD cases because the majority of cases were not admitted to the hospital. While we recognize their importance to predict worse outcomes among hospitalized cases, the performance of the model previously published by our group10 strongly suggests that demographics and chronic conditions greatly impact clinical outcomes, irrespective of acute disease severity and its specific management strategies.
Among cases with SARS-CoV2 infection at an early phase of the epidemic in Portugal CKD was associated with worse outcomes. Age seems to be the only demographic risk factor associated with worse outcomes. These findings may inform health policies designed to protect this specific subgroup of the population, especially while measures of containment are being eased in many countries.