Renal biopsy registries allow histopathological data to be collected to improve knowledge of different pathologies and their natural history.
AimTo analyse the data of the Castilla La Mancha Glomerulonephritis Registry (GLOMANCHA) and the evolution of the different biopsy-proven pathologies between 1994 and 2008.
MethodsThe 6 most common biopsy-proven pathologies were collected during the 14 years of the study (941 biopsies) in the 5 participant centres of the autonomous community. In 2008, we assessed patient renal survival and mortality and we evaluated associated factors to each situation for each pathology.
ResultsOf the 941 biopsies, 59% belonged to men, with a mean age of 48±18 years. At the time of the biopsy, the median glomerular filtration rate was 50.3 (25.5–76.3) ml/min/1.73m2 and median proteinuria was 3.4 (1.5–6.4) grams per day. The most common pathology were lupus nephropathy, followed by focal segmental glomerulosclerosis and membranous nephropathy. Lupus nephropathy and minimal change disease achieved the best renal prognosis during follow-up (mean 7.3±4.8 years). Rapidly progressive glomerulonephritis type 3 and focal segmental glomerulosclerosis had the worst renal prognosis. In addition, rapidly progressive glomerulonephritis type 3 presented the worst vital prognosis.
ConclusionsIn GLOMANCHA, we demonstrate the poor prognosis of rapidly progressive glomerulonephritis type 3, in contrast to minimal change disease or lupus nephropathy. Renal function is an independent predictor of renal survival and mortality in this study.
Los registros de biopsias renales permiten la recogida de datos histopatológicos que, puestos en su conjunto, ayudan a comprender enfermedades y su historia natural.
ObjetivosAnalizar los datos del Registro de Glomerulonefritis de Castilla-La Mancha (GLOMANCHA) y la evolución de las diferentes enfermedades biopsiadas (1994-2008).
MétodosSe recogieron las 6 enfermedades biopsiadas más prevalentes durante los 14 años (941 biopsias) en 5 hospitales de la comunidad autónoma. En 2008 se evaluó la situación vital y renal de los pacientes y se analizaron los factores asociados a dicha situación en cada enfermedad.
ResultadosDe las 941 biopsias, el 59% fueron de varones con una edad media de 48±18 años. En el momento de la biopsia la mediana de filtrado glomerular (FG) era de 50,3 (25,5-76,3) ml/min/1,73 m2 y la de proteinuria de 3,4 (1,5-6,4) g al día. La enfermedad más frecuente fue la nefropatía lúpica, seguida de la glomeruloesclerosis focal y segmentaria, y de la membranosa. El mejor pronóstico renal durante el seguimiento (media 7,3±4,8 años) fue el de la nefropatía lúpica y la nefropatía por cambios mínimos; la glomeruloesclerosis focal y segmentaria y las glomerulonefritis rápidamente progresivas de tipo 3 tuvieron el peor pronóstico renal. Esta última, además, tuvo el peor pronóstico vital.
ConclusionesGLOMANCHA demuestra el mal pronóstico de las glomerulonefritis rápidamente progresivas de tipo 3, a diferencia de la nefropatía por cambios mínimos y lúpica. La función renal es un predictor independiente de supervivencia renal y de mortalidad en nuestra población.
Percutaneous biopsy is the test of choice for the diagnosis of kidney diseases. Nephrologists make decisions regarding treatment and prognosis by studying the biopsy's histological data.1
This technique is safe and relatively simple to perform. Renal biopsy complications are very limited and when they do occur they are mainly minor.2 Some centres even advocate biopsies in an outpatient setting, without the need for hospital admission.3 Furthermore, practically half of biopsied patients have a potentially treatable disease that tends to include the use of immunosuppressant drugs, thus requiring a certain degree of specificity in the diagnosis due to the adverse effect profile of these treatments.4 Choosing suitable candidates for biopsy is, therefore, a challenge for nephrologists because the benefits of early and treatable diagnosis outweigh the risks if carried out correctly.
Nevertheless, there are no established criteria for performing renal biopsy; the criteria vary according to different centres or countries.1 In general, nephrotic syndrome, nephritic syndrome and acute renal failure tend to be main indications for performing a biopsy.5–7
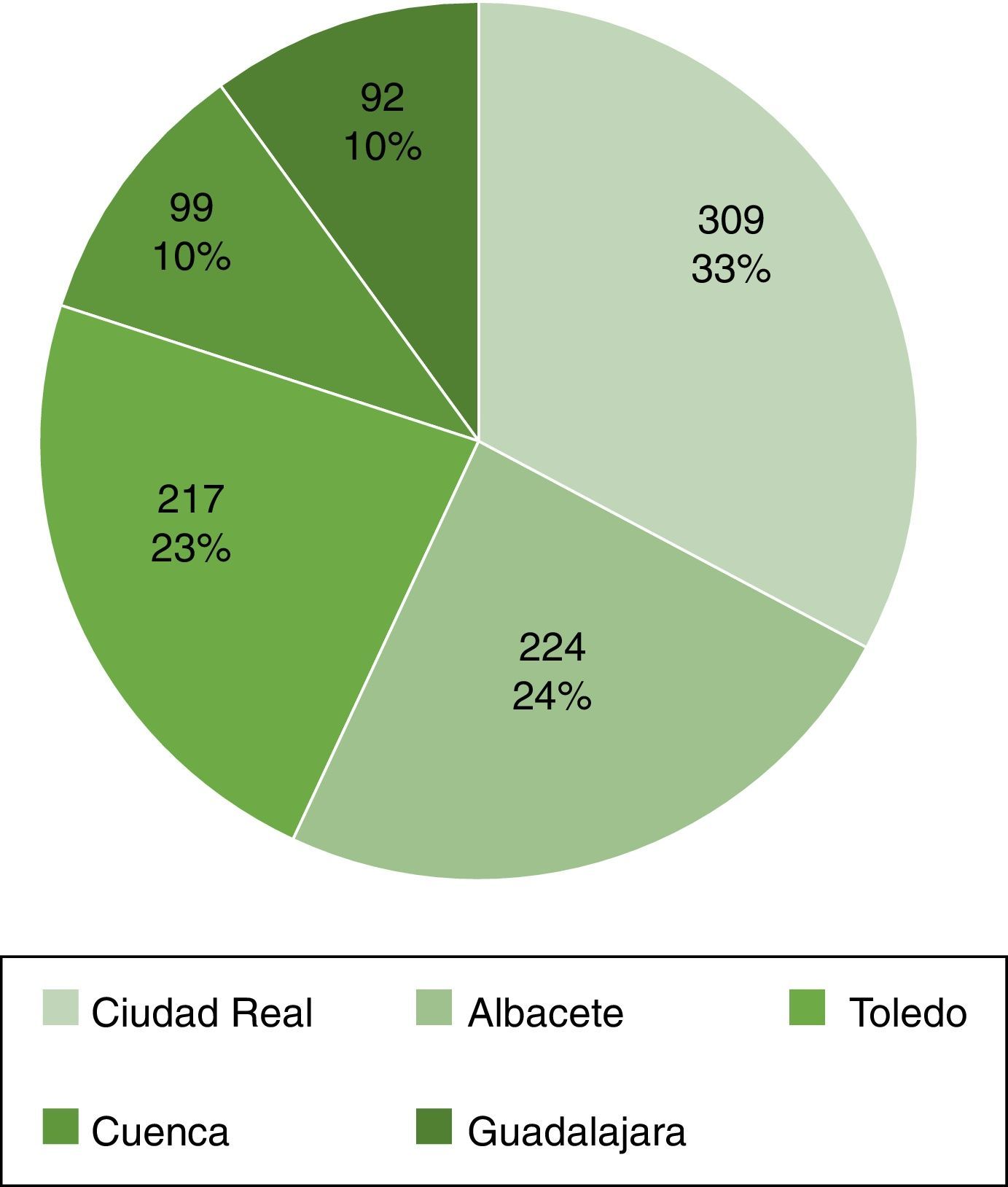
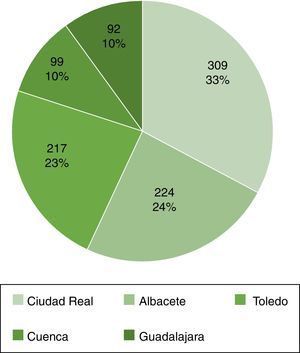
Spain's largest histopathological data collection is the Spanish Registry of Glomerulonephritis sponsored by the Spanish Society of Nephrology, which has recorded more than 22,000 biopsies to date.8,9 In the region of Castilla-La Mancha, all the biopsies performed between 1994 and 2008 at hospitals in Albacete, Ciudad Real, Cuenca, Guadalajara and Toledo have been recorded.
This study aims to explore the epidemiological and clinical characteristics of biopsied patients with a diagnosis of glomerulonephritis in Castilla-La Mancha (GLOMANCHA) and to analyse renal survival and mortality during follow-up.
MethodsPatientsRegistry of patients who have undergone biopsy in the Autonomous Community of Castilla-La Mancha between 1994 and 2008. Data have been provided by different hospitals from the community provinces (Albacete, Ciudad Real, Cuenca, Guadalajara and Toledo). Forms were sent that included epidemiological data (age and gender) and renal function data (glomerular filtration rate [GFR] and proteinuria) at the time of renal biopsy and at the cut-off date in 2008 (death, alive and requiring renal replacement therapy or alive with no need for renal replacement therapy). Each centre presented its own indications for renal biopsy.
Histological diagnosisThe following histological diagnoses were collected in the registry:
- -
Minimal change disease.
- -
IgA nephropathy.
- -
Rapidly progressive type III glomerulonephritis (vasculitis).
- -
Membranous nephropathy.
- -
Lupus nephritis.
- -
Focal segmental glomerulosclerosis.
Other biopsied diseases were excluded from the analysis (n=103) due to the small number of cases recorded (acute glomerulonephritis, membranoproliferative glomerulonephritis types I, II and III, extracapillary types I and II glomerulonephritis and others), as were paediatric patients who underwent biopsy.
Follow-upFollow-up was ended in 2008, when vital status (alive or dead) and renal status (on renal replacement therapy [RRT] or not) were recorded. We checked for an association between the different factors collected (age, gender, proteinuria and renal function) and survival (renal and overall) and multivariate models were carried out adjusted according to these factors. Proteinuria was analysed dichotomously according to the median for each disease.
Statistical analysisThe variables are expressed as mean (standard deviation) or median (interquartile interval) according to their distribution. The Student's t-test or Chi-squared test were used for parametric variables and the Mann–Whitney test for non-parametric variables. Overall and renal survival studies were performed using univariate and multivariate Cox regression adjusted according to the variables studied (age, gender, GFR and proteinuria). With an aim to show clinically significant predictive parameters in this study, the gender and GFR variables were treated as continuous variables and median proteinuria was taken for each of the diseases studied. Moreover, renal and overall survival curves were performed for the different diseases using the Kaplan–Meier estimator. A p-value <0.05 was considered to be significant. All the analyses were carried out using SPSS v18.0 (SPSS Inc., Chicago, IL, USA).
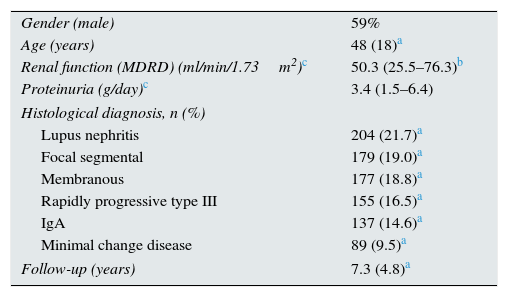
ResultsBaseline characteristicsOf the 941 biopsies recorded, 555 (59%) were men, with a mean age of 48±18 years. Median GFR estimated by MDRD at the time of biopsy was 50.3 (25.5–76.3) ml/min/1.73m2, with a median proteinuria of 3.4 (1.5–6.4) g per day. The most common diseases were lupus nephritis, focal segmental glomerulosclerosis and membranous nephropathy. The rest of the data is shown in Table 1. The distribution according to centre is shown in Fig. 1.
Baseline characteristics of the population biopsied.
| Gender (male) | 59% |
| Age (years) | 48 (18)a |
| Renal function (MDRD) (ml/min/1.73m2)c | 50.3 (25.5–76.3)b |
| Proteinuria (g/day)c | 3.4 (1.5–6.4) |
| Histological diagnosis, n (%) | |
| Lupus nephritis | 204 (21.7)a |
| Focal segmental | 179 (19.0)a |
| Membranous | 177 (18.8)a |
| Rapidly progressive type III | 155 (16.5)a |
| IgA | 137 (14.6)a |
| Minimal change disease | 89 (9.5)a |
| Follow-up (years) | 7.3 (4.8)a |
Table 2 shows the data collected from the different diseases biopsied. There was male predominance in all the entities – the most striking was IgA glomerulonephritis – except for lupus nephritis (19.4% of the total), in which more women were diagnosed. In terms of age of presentation, lupus nephritis affected patients at the youngest age (34±15 years) in contrast to rapidly progressive glomerulonephritis type III (63±13 years). Data on renal function and proteinuria vary according to the different diseases studied. Minimal change disease and membranous nephropathy present normal renal function medians (75.3 and 67.1ml/min/m2, respectively) with proteinuria greater than 7g per day. Meanwhile, at the time of biopsy, rapidly progressive glomerulonephritis type III presented a very deteriorated GFR (13.6; 7.6–21.55ml/min/1.73m2) with less proteinuria than the other diseases (1.5; 0.8–2.5g per day).
Baseline characteristics of the patients according to histological diagnosis.
| MCD | FSGS | MN | IgA | RPGN type III | LN | |
|---|---|---|---|---|---|---|
| Gender (male) | 56.2% | 70.9% | 59.3% | 81.9% | 55.5% | 19.4% |
| Age (years)a | 46 (20) | 48 (16) | 52 (16) | 42 (15) | 63 (13) | 34 (15) |
| Renal function (MDRD) (ml/min/1.73m2)b,c | 75.3 (50.5–96.4) | 45.1 (29.3–63.1) | 67.1 (41.5–83.6) | 48.1 (28.5–73.05) | 13.6 (7.6–21.55) | 65.9 (46.9–85.3) |
| Proteinuria (g/day)b,c | 7.5 (5.0–11.2) | 3.0 (1.7–5.5) | 7.1 (4.9–11.4) | 2.0 (1.1–3.7) | 1.5 (0.8–2.5) | 2.8 (1.2–4.9) |
MCD: minimal change disease; RPGN type III: rapidly progressive glomerulonephritis type III; FSGS: focal segmental glomerulosclerosis; IgA: IgA nephropathy; LN: lupus nephritis; MN: membranous nephritis.
During follow-up (7.3±4.8 years), 146 patients died (15.5%); 74 prior to starting RRT (7.8%) and 72 (7.6%) who were already part of the programme. A total of 53 patients were on dialysis at the end of follow-up (5.6%), 74 had received a kidney transplant (7.8%) and 609 (64.7%) were alive and had not required RRT. Fifty-nine (59) patients were lost to follow-up (6.2%).
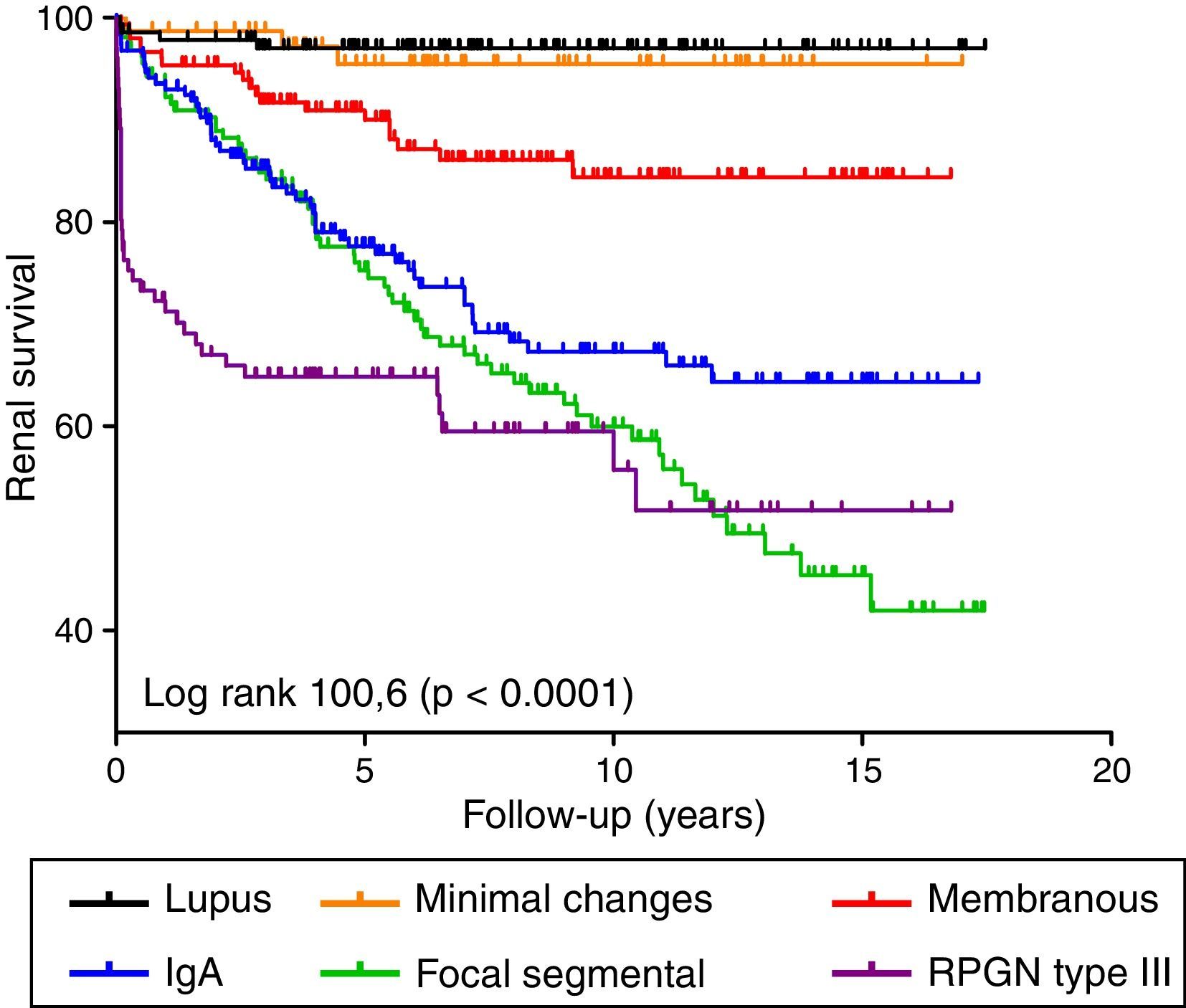
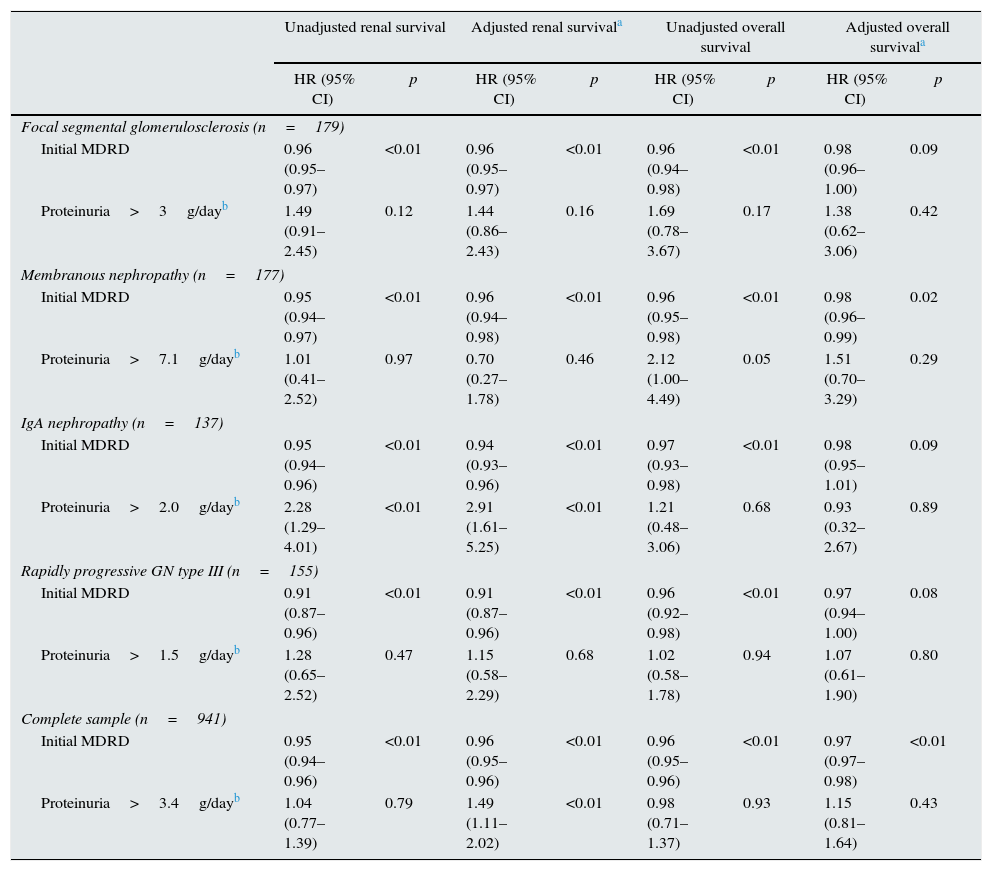
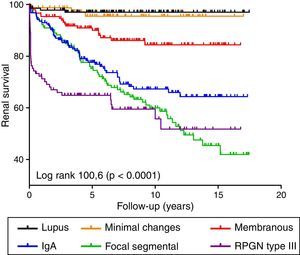
Factors associated with renal survivalA total of 199 patients (10.5%) required RRT. Fig. 2 shows the renal survival curves for the different diseases. Of these, IgA glomerulonephritis, focal segmental glomerulosclerosis and rapidly progressive glomerulonephritis type III were associated with the worst prognosis. Overall, proteinuria and a decrease in GFR were independent predictive factors for worse renal survival in a model adjusted for age and gender (Table 3). In terms of the diseases individually, deterioration in GFR was associated with worse renal prognosis in all the diseases. However, proteinuria was only an independent predictive factor in IgA glomerulonephritis (Table 3). Taking proteinuria continuously, the results overlapped (data not shown).
Univariate and multivariate Cox regression for renal and overall survival of the categories with a worse prognosis.
| Unadjusted renal survival | Adjusted renal survivala | Unadjusted overall survival | Adjusted overall survivala | |||||
|---|---|---|---|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | HR (95% CI) | p | HR (95% CI) | p | |
| Focal segmental glomerulosclerosis (n=179) | ||||||||
| Initial MDRD | 0.96 (0.95–0.97) | <0.01 | 0.96 (0.95–0.97) | <0.01 | 0.96 (0.94–0.98) | <0.01 | 0.98 (0.96–1.00) | 0.09 |
| Proteinuria>3g/dayb | 1.49 (0.91–2.45) | 0.12 | 1.44 (0.86–2.43) | 0.16 | 1.69 (0.78–3.67) | 0.17 | 1.38 (0.62–3.06) | 0.42 |
| Membranous nephropathy (n=177) | ||||||||
| Initial MDRD | 0.95 (0.94–0.97) | <0.01 | 0.96 (0.94–0.98) | <0.01 | 0.96 (0.95–0.98) | <0.01 | 0.98 (0.96–0.99) | 0.02 |
| Proteinuria>7.1g/dayb | 1.01 (0.41–2.52) | 0.97 | 0.70 (0.27–1.78) | 0.46 | 2.12 (1.00–4.49) | 0.05 | 1.51 (0.70–3.29) | 0.29 |
| IgA nephropathy (n=137) | ||||||||
| Initial MDRD | 0.95 (0.94–0.96) | <0.01 | 0.94 (0.93–0.96) | <0.01 | 0.97 (0.93–0.98) | <0.01 | 0.98 (0.95–1.01) | 0.09 |
| Proteinuria>2.0g/dayb | 2.28 (1.29–4.01) | <0.01 | 2.91 (1.61–5.25) | <0.01 | 1.21 (0.48–3.06) | 0.68 | 0.93 (0.32–2.67) | 0.89 |
| Rapidly progressive GN type III (n=155) | ||||||||
| Initial MDRD | 0.91 (0.87–0.96) | <0.01 | 0.91 (0.87–0.96) | <0.01 | 0.96 (0.92–0.98) | <0.01 | 0.97 (0.94–1.00) | 0.08 |
| Proteinuria>1.5g/dayb | 1.28 (0.65–2.52) | 0.47 | 1.15 (0.58–2.29) | 0.68 | 1.02 (0.58–1.78) | 0.94 | 1.07 (0.61–1.90) | 0.80 |
| Complete sample (n=941) | ||||||||
| Initial MDRD | 0.95 (0.94–0.96) | <0.01 | 0.96 (0.95–0.96) | <0.01 | 0.96 (0.95–0.96) | <0.01 | 0.97 (0.97–0.98) | <0.01 |
| Proteinuria>3.4g/dayb | 1.04 (0.77–1.39) | 0.79 | 1.49 (1.11–2.02) | <0.01 | 0.98 (0.71–1.37) | 0.93 | 1.15 (0.81–1.64) | 0.43 |
GN: glomerulonephritis; HR (95% CI): hazard ratio (95% confidence interval).
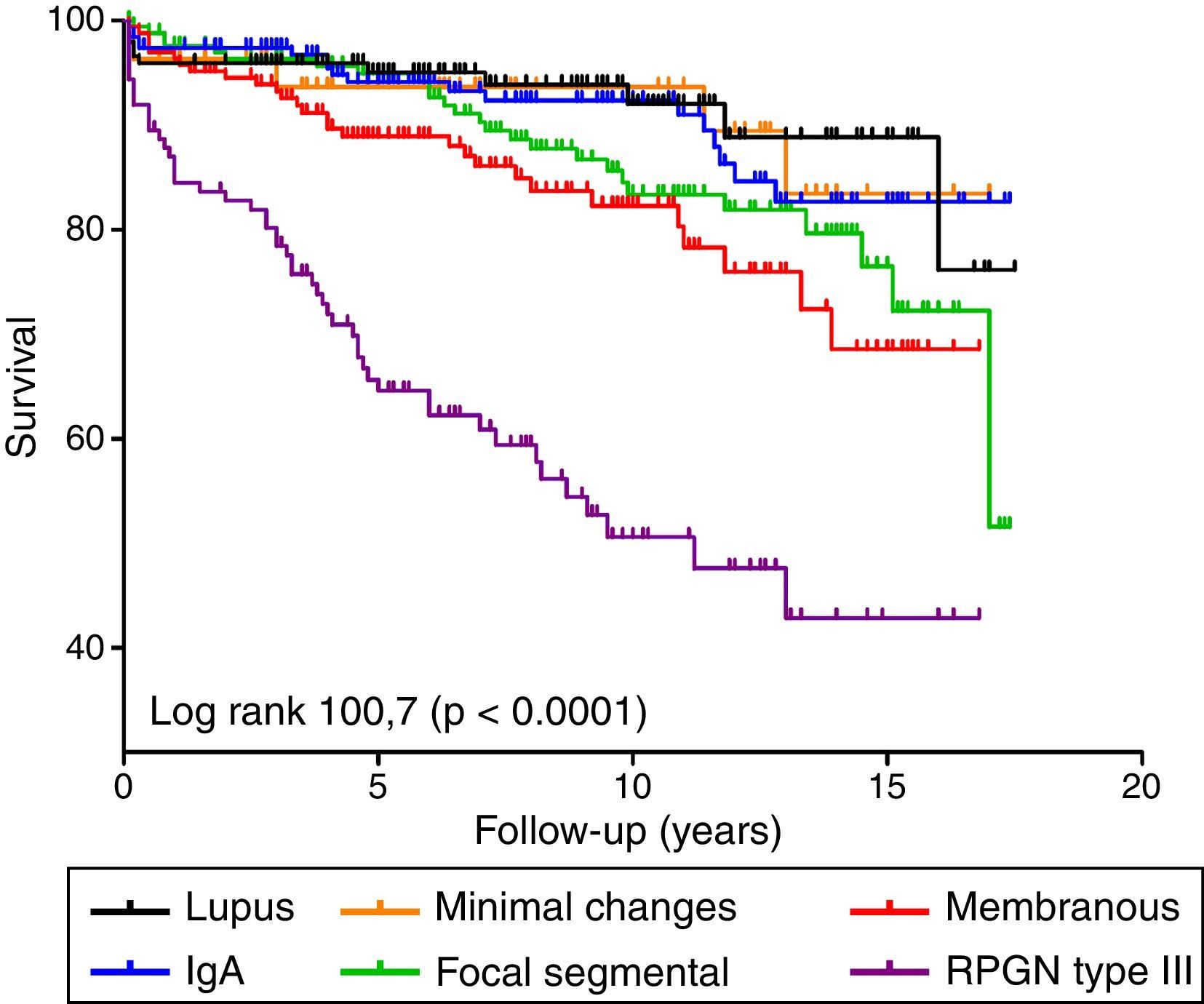
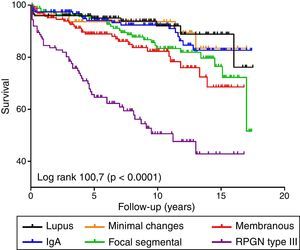
A total of 146 patients (15.5%) died during follow-up. Fig. 3 shows the survival curves for the different diseases. Rapidly progressive glomerulonephritis type III presented the worst vital prognosis. Overall, a worse GFR and age (data not shown in the table, HR 1.07; 95% CI: 1.05–1.08; p<0.001) were independently associated with mortality in a model adjusted for proteinuria and gender (Table 3). By analysing each disease separately, we found that age was the most important independent predictive factor for mortality in all diseases (data not shown) and only deterioration in GFR predicted mortality in membranous nephropathy, maintaining a trend in the other diseases (Table 3).
DiscussionThe Castilla-La Mancha biopsy registry presents extended follow-up (more than 7 years) and shows that patients undergoing this procedure are predominately men, with a mean age of 48 years, mildly decreased renal function and proteinuria greater than 3g per day. The most common histological diagnoses were lupus nephritis followed by focal segmental glomerulosclerosis and membranous nephropathy. Although these data may initially draw attention, the results must be put into context, as each centre has their own indications for renal biopsy; they do not reflect the total incidence of these diseases, but rather only those which are diagnosed histopathologically. Renal survival is excellent in lupus nephropathy and in minimal change disease, unlike focal segmental glomerulosclerosis and rapidly progressive glomerulonephritis type III. The latter, moreover, has the worst vital prognosis. All these data are consistent with the registries published previously.10,11
In terms of incidence, IgA nephropathy has always remained at the head of the Spanish Registry of Glomerulonephritis, followed by lupus nephritis. According to 2010–2013 data, focal segmental glomerulosclerosis and membranous nephropathy were in third and fourth place respectively. This difference with our series is probably due to the lack of biopsies in the IgA group, if they do not present poor prognosis data and if clinical manifestations are suggestive of this disease.
Regarding the diseases with poor renal prognosis, it was interesting that in our sample GFR at the time of biopsy was the predictor for renal survival, and not proteinuria. Only in IgA nephropathy, proteinuria greater than 2g multiplied the risk for starting RRT by 2.9. In a study that included 141 patients, the Spanish Society of Nephrology Glomerular Disease Study Group (GLOSEN) confirmed the excellent prognosis for patients with IgA nephropathy and minimum or null proteinuria in the long term.12
Rapidly progressive glomerulonephritis type III, which presents the worst prognosis of the diseases studied, has a severely decreased GFR (MDRD 13.6ml/min/1.73m2), with a mean age greater than the other diseases (63 years) at the time of biopsy. These 2 situations are consistent with data recently published by the SEN (Spanish Society of Nephrology) registry and both (GFR and age) are responsible for its poor prognosis.8
Membranous nephropathy is, together with minimal change disease, the disease with the most proteinuria at the time of biopsy (more than 7g per day). Furthermore, both present a GFR greater than 60ml/min/1.73m2. Overall and renal survival are good at 15 years follow-up (greater than 80%). Nevertheless, membranous nephropathy is probably under-diagnosed. More and more authors are advocating for performing biopsy in diabetic patients with proteinuria, as this is often erroneously attributed to diabetic nephropathy.13 In a recent study published by Soleymanian et al., renal biopsy was performed in 46 diabetic patients with proteinuria, haematuria or impaired kidney function and only 34.8% had pure diabetic nephropathy.14
At the time of renal biopsy, the patients with focal segmental glomerulosclerosis presented deterioration of renal function with GFR estimated at 45.1ml/min/1.73m2 and a median proteinuria of 3g per day. These data are consistent with other registries published in the work of Kwon et al., which included 111 Korean adults.15 There was a male predominance (70%) in the sample. Renal function marked the patients’ prognosis, whose renal survival was poor. As in our study, the prognosis factors in the Korean study were renal function and age, but also proteinuria. This is because the final variable of their study was combined, including increased serum creatinine or start of RRT, unlike our study in which we only considered the latter. Moreover, since D’Agati proposed a classification of histological variants in 2003, several studies have aimed to assess renal prognosis in each of these.16 Although there is no clear consensus on whether histological type is a prognostic factor, the collapsing variant seems to be the most aggressive.17,18
In terms of lupus nephritis, previously published data is confirmed regarding the involvement of mainly young women and proteinuria less than 2g per day. For this disease, membranous nephritis has the best renal prognosis compared to the diffuse proliferative variant.19,20
Our study is not exempt of limitations, such as the small amount of data collected from the patients (to facilitate the delivery from the collaborating centres) or its retrospective nature. Moreover, it should be highlighted that only baseline analytical patient data were recorded, which also involves certain limitations. Furthermore, we do not have the treatment data for the patients biopsied and there may be a certain degree of heterogeneity as we looked at diseases from different centres. Nevertheless, the large sample size and follow-up period grant validity to the results presented.
In conclusion, the Castilla-La Mancha Registry of Glomerulonephritis shows the poor prognosis of rapidly progressive glomerulonephritis type III, unlike minimal change disease and lupus nephritis. Renal function is an independent predictor of renal survival and mortality in our population.
FundingThis study was funded in part by the Sociedad Castellano-Manchega de Nefrología (Castellano-Manchega Nephrology Society).
Conflicts of interestNone declared.
Please cite this article as: Conde Olasagasti JL, Acevedo Ribó M, Roca Muñoz A, Vozmediano Poyatos C, Rivera F, Illescas Fernández-Bermejo ML, et al. Estudio evolutivo de las glomerulonefritis en Castilla-La Mancha (GLOMANCHA) en el periodo 1994-2008. Nefrologia. 2016;36:237–242.