Las nuevas guías internacionales del consorcio KDIGO (Kidney Disease: Improving Global Outcomes) sobre la enfermedad renal crónica (ERC) y sobre el manejo de la presión arterial (PA) en pacientes con ERC constituyen la actualización de las correspondientes guías KDOQI (Kidney Disease Outcomes Quality Initiative) de 2002 y 2004. El objetivo de estos documentos es ofrecer una guía actualizada para el diagnóstico, la evaluación, el manejo y el tratamiento del paciente con ERC. La primera guía conserva la definición de ERC de 2002, pero ofrece una clasificación pronóstica mejorada. Además, se revalúan los conceptos sobre el pronóstico de la ERC, y se establecen recomendaciones para el manejo de los pacientes y sobre los criterios de derivación al especialista en nefrología. La segunda guía conserva el objetivo de una PA < 130/80 mmHg para pacientes con ERC que curse con una albuminuria elevada (cociente albúmina/creatinina en muestra aislada de orina entre 30 y 300 mg/g) o proteinuria (cociente albúmina/creatinina en muestra aislada de orina > 300 mg/g), pero recomienda el objetivo menos estricto de PA < 140/90 mmHg para pacientes con albuminuria normal. El desarrollo de las guías siguió un proceso predeterminado de revisión y evaluación de las evidencias disponibles. Las recomendaciones sobre el manejo y el tratamiento están basadas en la revisión sistemática de los estudios relevantes. El sistema GRADE (Grading of Recommendations Assessment, Development and Evaluation) se utilizó para evaluar la calidad de la evidencia y emitir el grado de recomendación. También se discuten las áreas de incertidumbre de los distintos aspectos tratados.
The new Kidney Disease: Improving Global Outcomes (KDIGO) international guidelines on chronic kidney disease (CKD) and the management of blood pressure (BP) in CKD patients are an update of the corresponding 2002 and 2004 KDOQI (Kidney Disease Outcomes Quality Initiative) guidelines. The documents aim to provide updated guidelines on the assessment, management and treatment of patients with CKD. The first guidelines retain the 2002 definition of CKD but present an improved prognosis classification. Furthermore, concepts about prognosis of CKD, recommendations for management of patients, and criteria for referral to the nephrologist have been updated. The second guideline retains the <130/80mmHg-goal for management of BP in patients with CKD presenting increased albuminuria or proteinuria (albumin-to-creatinine ratio 30-300 mg/g, and >300 mg/g, respectively) but recommends a less-strict goal of <140/90mmHg in patients with normoalbuminuria. The development of the guidelines followed a predetermined process in which the evidence available was reviewed and assessed. Recommendations on management and treatment are based on the systematic review of relevant studies. The GRADE system (Grading of Recommendations Assessment, Development and Evaluation) was used to assess the quality of evidence and issue the grade of recommendation. Areas of uncertainty are also discussed for the different aspects addressed.
INTRODUCTION
The epidemiological view of chronic kidney disease has experienced a significant change in the last twenty years. Initially restricted to relatively low incidence pathologies, such as glomerular diseases or hereditary nephropathies, and to a specialised care setting (Nephrology), CKD that is predominant today affects a large percentage of the population and is related to highly prevalent diseases or phenomena, such as old age, high blood pressure (HBP), diabetes or cardiovascular disease. CKD is often a comorbidity suffered by patients followed up by many medical specialties, particularly Primary Care, Internal Medicine, Cardiology, Geriatrics, Endocrinology and any other medical or surgical specialty in which patients at risk of developing CKD are treated, especially those related to old age. Advanced CKD patients included in renal replacement therapy programmes using dialysis and transplantation are considered the visible part of the iceberg that is the major public health problem of CKD in the population.
In the last ten years, scientific nephrology societies have worked hard to collect information on CKD and research this disease. In 2002, the US National Kidney Foundation published K/DOQI (Kidney Disease Outcome Quality Initiative)1,2 guidelines, which established the current definition of CKD, the classification into grades and the basic assessment methods, such as renal function estimation using equations to calculate the glomerular filtration rate (GFR) based on the determination of serum creatinine and the assessment of albuminuria using the albumin/creatinine ratio in an isolated urine sample. In 2004, the first K/DOQI guidelines on HBP management in CKD patients were published3. The K/DOQI CKD classification was included in the first Spanish Society of Nephrology (S.E.N.) guidelines that were published after this time4,5. In 2003, the Kidney Disease Improving Global Outcomes (KDIGO) organisation was founded as an international and independent expert group, with Spanish participation, in order to develop initiatives for the prevention and management of CKD (http://www.kdigo.org/). The first guidelines on the definition and classification of CKD were published in 2005 and they endorsed the approach of the 2002 K/DOQI guidelines6. In 2008, the S.E.N. and the Spanish Society of Family and Community Medicine (semFYC) developed the S.E.N.-semFYC CKD Consensus Document, which established the bases for joint management and prevention of kidney disease between Primary Care and Nephrology7. The S.E.N. developed a very extensive CKD training and research activity programme8, one of the main exponents of which is the EPIRCE (Epidemiological Study of Renal Failure in Spain) study, which revealed a CKD prevalence of 9.16% in the general population9. In parallel, the main international and national guidelines on hypertensive patient management incorporated this CKD diagnostic system and included a decrease in renal function and albuminuria amongst the main cardiovascular risk variables10,11. Lastly, in recent months, a CKD consensus document promoted by the S.E.N. was published by ten Spanish scientific societies12. In this context, new KDIGO guidelines were published in December 2012 and January 2013 on the assessment and treatment of CKD and on antihypertensive treatment in patients with this disease13,14, the full version of which is available free at http://www.kdigo.org. The objective of the present article was to create a short, practical document that includes the main points of these two latest KDIGO guidelines.
METHODOLOGY
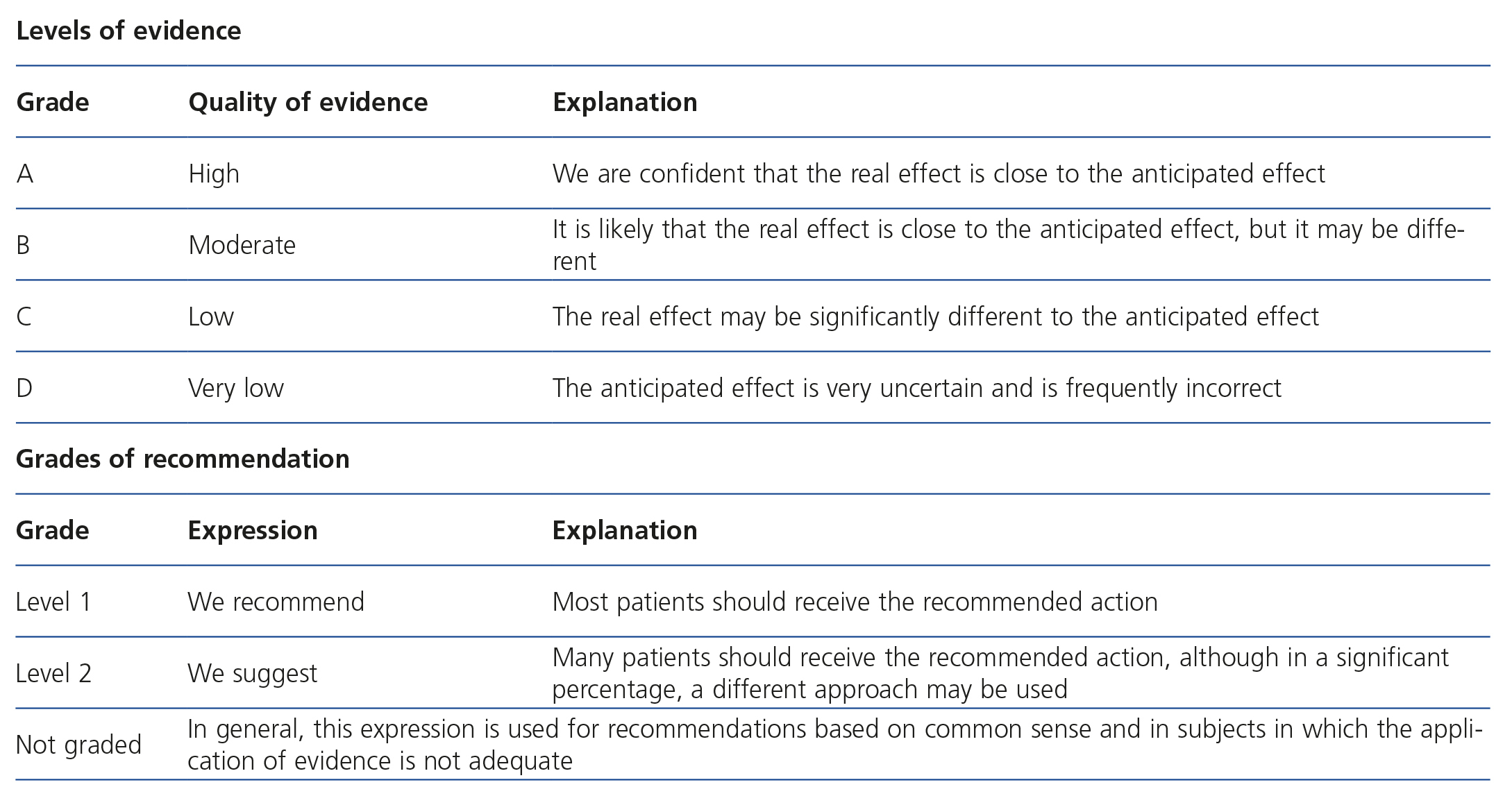
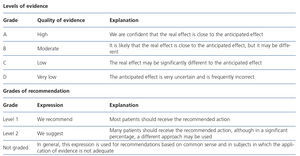
The recommendations on the management and treatment of CKD expressed in the abovementioned KDIGO guidelines were based on the systematic review of relevant studies by working groups of international nephrology experts and a team of experts in evidence-based medicine. The resulting manuscripts were subject to a public review. Those who carried out this review appear in the corresponding appendices of the guidelines. The GRADE (Grading of Recommendations Assessment, Development and Evaluation) system was used to assess the quality or level of evidence and issue the grade of recommendation (GR). Appendix 1 displays the GRADE system definitions used by the KDIGO group.
The present document, which includes the aforementioned KDIGO guidelines, was prepared using the following methodology. A drafting committee prepared summaries of the guideline chapters by subject area. The summaries were compiled by special editors and the resulting manuscript was revised by an expert committee appointed by the S.E.N. After collecting expert contributions, a definitive document was prepared, which was submitted for the approval of all the authors. Appendix 2 displays the distribution of authors in the different working groups.
This document summarises the aspects of the aforementioned KDIGO guidelines that refer to adult CKD patients in stages before renal replacement therapy, with the exception of HBP management in kidney transplant patients. Furthermore, the document aims to provide a holistic perspective on these and other daily clinical practice guidelines. The level of evidence and the GR are expressed with “we recommend” for a level 1 recommendation (most patients should receive the recommended action) and “we suggest” for the level 2 recommendation (many patients should receive the recommended action, although a significant percentage of cases may be subject to another approach). The authors of this document highlight the current small amount of grade A evidence and level 1 recommendations in the KDIGO guidelines and consider that many CKD aspects are the subject of debate. The GR and the corresponding evidence level of the main recommendations are displayed in brackets.
DEFINITION OF CHRONIC KIDNEY DISEASE
CKD is defined as abnormalities of kidney structure or function, present for at least three months with implications for health (GR, not graded). This definition is the same as the previous definition1,2,6,7, except for the added phrase “with implications for health”, which reflects the concept that there may be certain abnormalities of kidney structure or function that do not have prognostic consequences (for example, a simple renal cyst). CKD diagnosis criteria are kidney damage markers or a GFR of less than 60ml/min/1.73m2 (Table 1). Duration of more than three months of any of these abnormalities may be confirmed prospectively or be deduced from previous registries.
Chronic kidney disease categories or grades
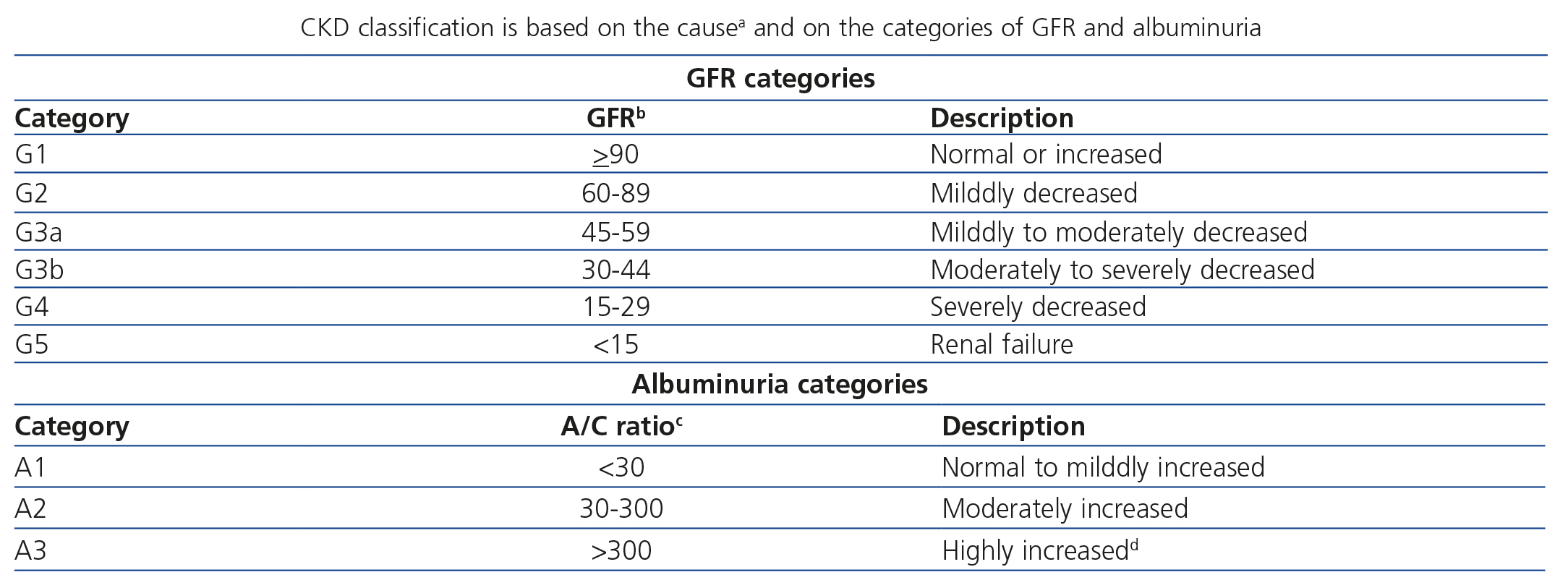
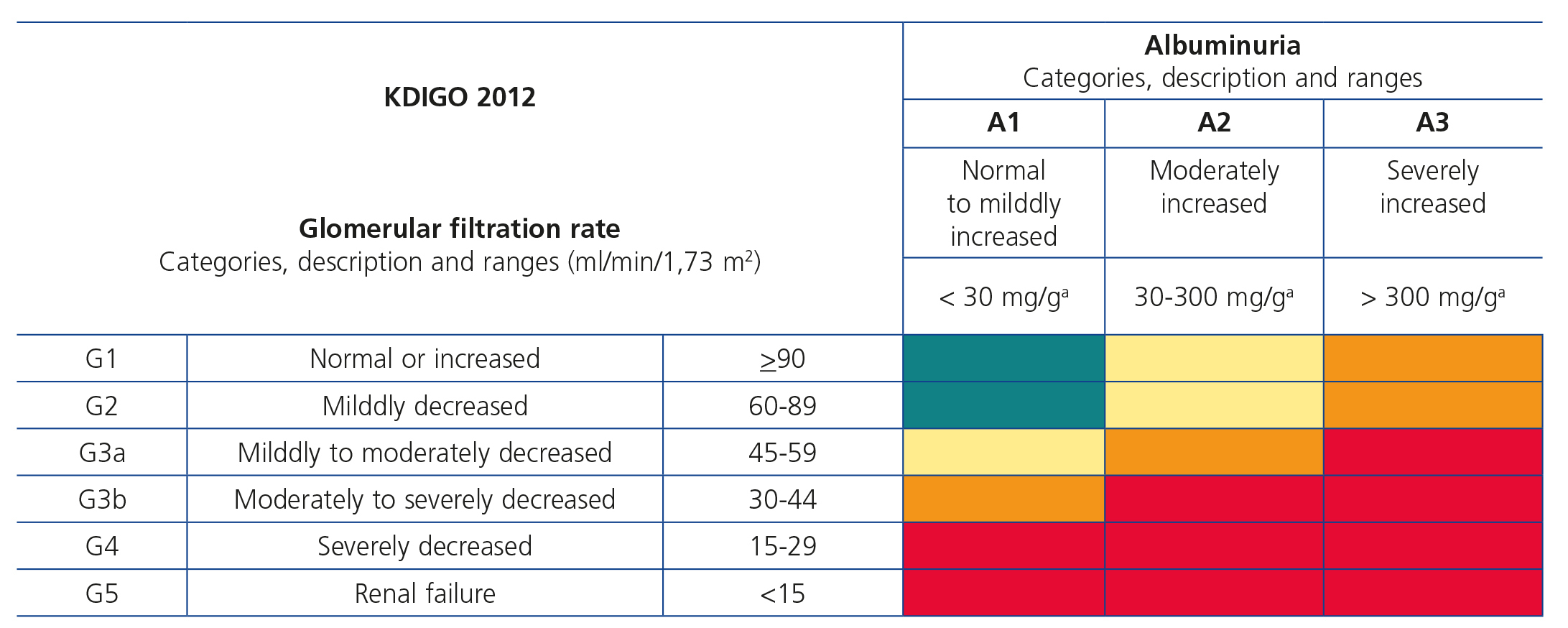
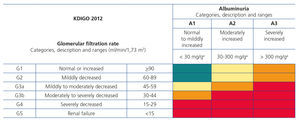
After the diagnosis is confirmed, CKD is classified according to the GFR and albuminuria categories and the aetiology (GR 1B). The cause of CKD is established according to the presence or absence of a systemic disease with potential kidney involvement or through observed or suspected pathological abnormalities (GR, not graded). The GFR (G1 to G5) and albuminuria (A1 to A3) grades are displayed in Table 2 (GR, not graded).
With respect to the previous CKD classification1,2,6,7, 60ml/min/1.73 m2 is retained as the threshold for defining the GFR and grade 3 is subdivided into G3a and G3b, according to whether the GFR is between 59 and 45 or between 44 and 30ml/min/1.73m2, respectively. Furthermore, albuminuria should be classified in any GFR grade. It is also recommended to replace the term “microalbuminuria” for “moderately increased albuminuria”. Albuminuria is classified as A1, A2 or A3 according to the albumin/creatinine ratio in an isolated urine sample for values <30, 30-300 or >300mg/g, respectively (or according to the corresponding urine albumin excretion values displayed in Table 2).
The reaffirmation of considering a GFR <60ml/min/1.73m2 as the value for defining CKD has been the subject of debate, particularly in older patients, given the reduction in GFR associated with age. This limit is based on results of the CKD Prognosis Consortium meta-analysis15-17. This study demonstrated the association between a GFR <60ml/min/1.73m2 and the risk of overall mortality, cardiovascular mortality, CKD progression, progression to stage 5 CKD and acute renal failure, both in the general population and in high cardiovascular risk groups. Furthermore, the risk of nephrotoxicity due to drugs and metabolic and endocrinological complications exponentially increases with a GFR <60ml/min/1.73m2.
Risk stratification
The variables that determine risk of CKD complications are the causes of the latter, e.g., GFR grade, albuminuria grade and other risk factors or comorbidities. Upon diagnosing CKD in a specific patient, the aetiology should be explained along with the GFR and albuminuria grades, for example: G3a A3 CKD probably secondary to diabetic nephropathy for a diabetic patient with a GFR between 45 and 59ml/min/1.73m2 and albuminuria >300mg/g. This system allows the prognostic classification of CKD patients in situations of moderate, high or very high risk with respect to the baseline or reference risk of patients without CKD laboratory criteria (GFR >60ml/min/1.73m2 and albuminuria <30mg/g). Figure 1 displays the CKD risk stratification table according to GFR and albuminuria categories (GR, not graded).
ASSESSMENT OF CHRONIC KIDNEY DISEASE
The basic objective of the approach in CKD patients is to assess chronicity, the cause, the GFR and albuminuria. Chronicity is verified retrospectively, reviewing the previous history, or prospectively, whenever there are no previous laboratory tests. The cause is determined by the presence or absence of a systemic disease with potential renal involvement or observed or suspected pathological abnormalities. Furthermore, a family history of the disease, sustained intake of nephrotoxic drugs and environmental factors, such as contact with metals like lead or mercury are assessed and imaging tests are performed.
GFR is assessed using a serum creatinine test and a formula for estimating the GFR (GR 1A). Serum creatinine is determined using a specific test with adequate traceability for international reference standards and with minimal deviation with respect to the isotope dilution mass spectrometry reference method. This recommendation does not vary with respect to that of previous documents1,7,18. The new guidelines recommend changing the equation for estimating GFR to the 2009 CKD-EPI (CKD Epidemiology Collaboration) formula19 (GR 1B). GFR estimation using formulas based on serum creatinine may be less accurate in certain circumstances, such as in individuals who follow special diets (strict vegetarian or hyperprotein), those with major abnormalities in muscle mass (amputations, diseases with loss of muscle mass), those with extreme body mass indexes (<19kg/m2 or >35kg/m2) or those who are pregnant. In these circumstances and in certain situations in which it is necessary to optimise GFR assessment (for example, assessment of potential kidney donors, patients with an estimated GFR of between 45 and 59ml/min/1.73m2 without other markers of kidney damage or patients who require treatments with high renal toxicity), it is suggested to test cystatin C and the estimated GFR using a cystatin C-based equation (preferably CKD-EPI cystatin) or assess creatinine clearance after urine collection over a given period of time. Furthermore, in situations of severe sodium and water retention (cirrhosis with fluid retention, congestive heart failure, advanced hypothyroidism), as well as in any severe situation with haemodynamic instability, it is not appropriate to estimate the GFR using a serum creatinine-based equation.
Albuminuria is initially assessed through an isolated urine sample (first urine of the morning), using the albumin/creatinine ratio (GR 2B). In the case of advanced albuminuria grades, the protein/creatinine ratio offers a more accurate view of proteinuria, although this test is not routinely carried out in our setting. Classic test strips are also considered as a screening method. An albumin/creatinine ratio ≥30mg/g is confirmed with a second sample. The quantification of urinary albumin or protein excretion over a certain period of time, for example, the classic 24-hour urine test, is reserved for special cases in which a more precise estimation is considered to be necessary. The classic term microalbuminuria should not be used (GR, not graded), with albuminuria being expressed in the aforementioned grades A1, A2 or A3. In any case, albuminuria should be considered as such in the absence of incidental factors that may increase it, such as urine infections, physical exercise, fever or heart failure.
PROGRESSION OF CHRONIC KIDNEY DISEASE
The progression and evolution of CKD varies a lot between patients. Since we do not have sufficient evidence to define and identify those who have rapid progression, the recommendation is to simultaneously and systematically assess the estimated GFR and albuminuria. Both the reduction of the GFR and the grade of albuminuria influence prognosis and have a synergistic effect (Figure 1)15-17.
CKD progression is defined as a sustained decrease in the GFR of >5ml/min/1.73m2 per year or a change of category (from G1 to G2, from G2 to G3a, from G3a to G3b, from G3b to G4 or from G4 to G5) whenever the latter is accompanied by a GFR loss of ≥5ml/min/1.73m2 (GR, not graded). Small fluctuations in the GFR do not necessarily indicate progression. Whenever the aforementioned progression criteria are detected, it is necessary to rule out potentially reversible exacerbation factors (progression versus exacerbation), such as obstructive uropathy, volume depletion, situations of haemodynamic instability or use of non-steroidal anti-inflammatory drugs, cyclooxygenase 2 inhibitors, nephrotoxic antibiotics, radiocontrast agents or renin-angiotensin system (RAS) blockers in certain haemodynamic conditions. In the case of progression, the aim is to identify progression factors, such as CKD aetiology, age, sex, race, smoking habits, obesity, HBP, hyperglycaemia, dyslipidaemia, previous cardiovascular disease and exposure to nephrotoxic agents and those that can be altered will be treated20,21. Patients with progressive CKD also have a higher cardiovascular risk and as such, they are suitable for the appropriate prevention measures.
The frequency of CKD patient monitoring is also subject to recommendation. As such, the frequency of check-ups is also based on the risk stratification table (Figure 1). In general terms, low-risk patients will be reviewed annually, moderate-risk patients will be reviewed every six months and high and very high-risk patients should be reviewed three, four or more times a year. This regimen is valid for stable patients. Regular repetition of the kidney function parameters is also useful for optimising the assessment of disease progression.
PREVENTING PROGRESSION AND MANAGING COMPLICATIONS OF CHRONIC KIDNEY DISEASE
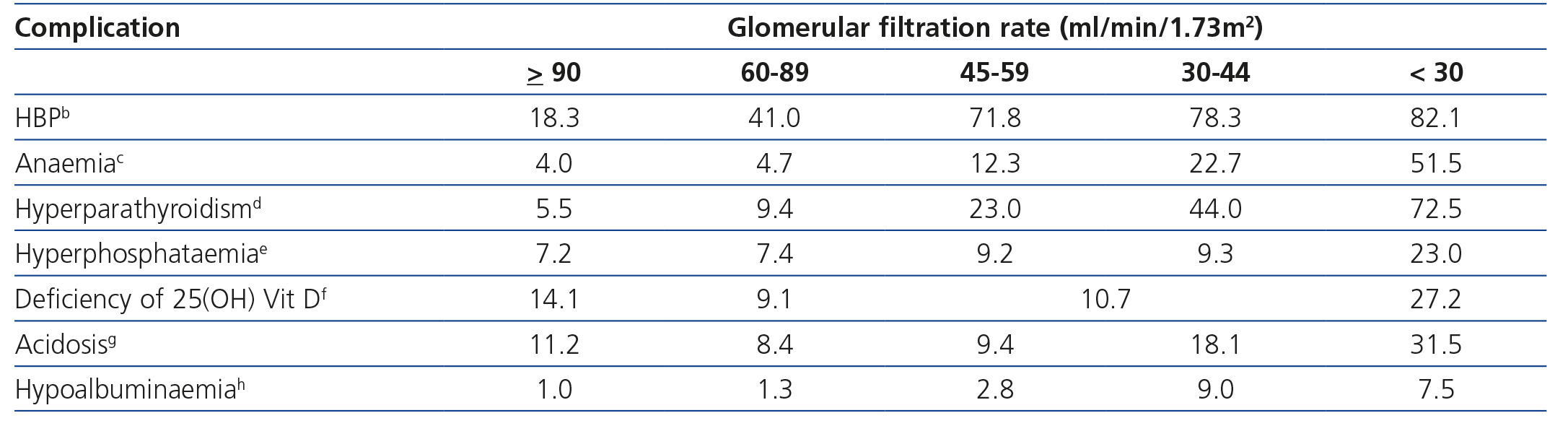
The most common complications of CKD and their prevalence according to GFR grades are displayed in Table 322,23. The comprehensive management of cardiorenal risk patients forms the basis of CKD progression prevention. Although different nuances may be established between CKD progression prevention measures and cardiovascular prevention measures, the bases for overall prevention are dietary and lifestyle changes, HBP control, RAS blockade and metabolic (mainly glycaemic and lipid) control.
General recommendations for managing high blood pressure
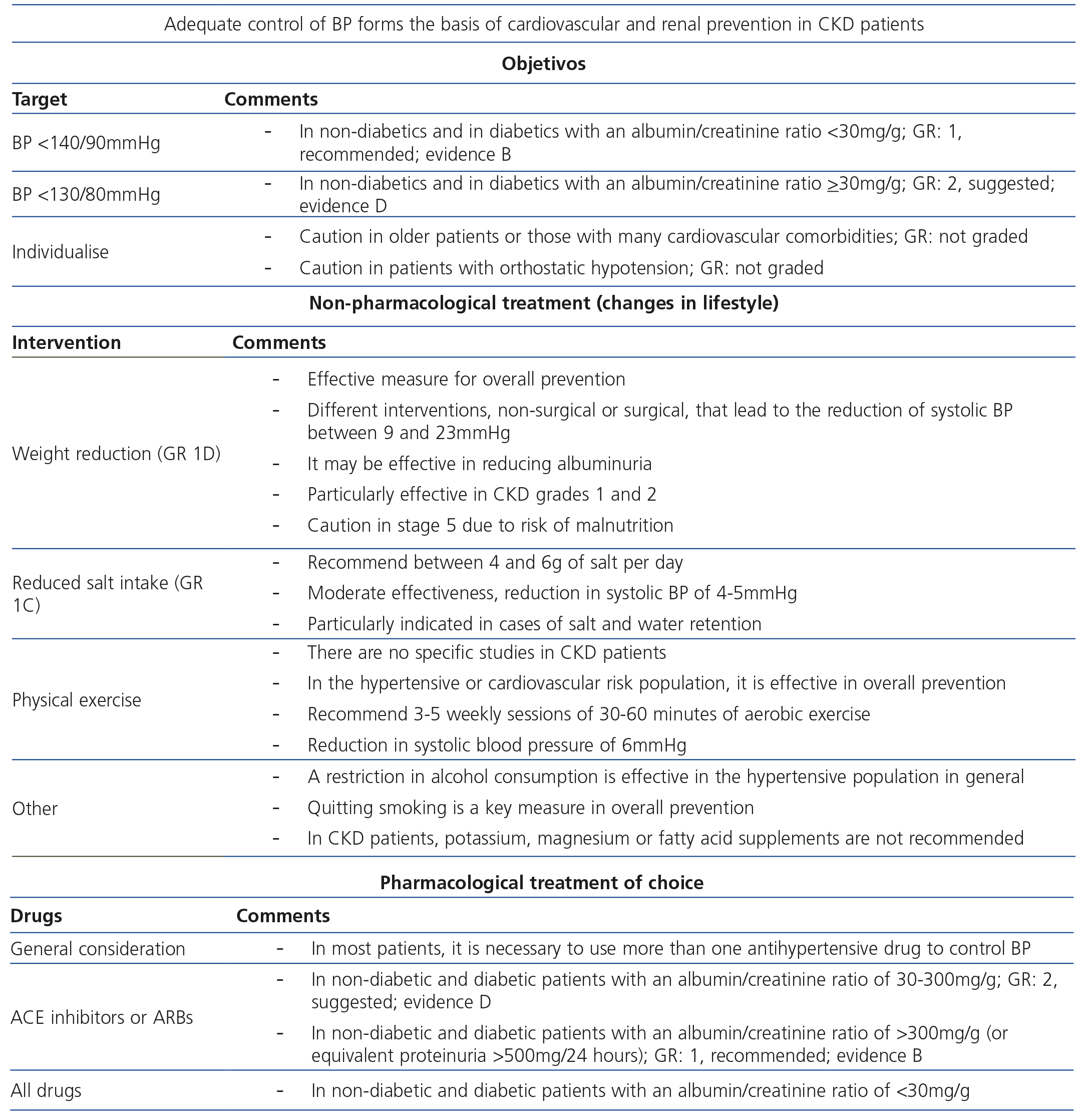
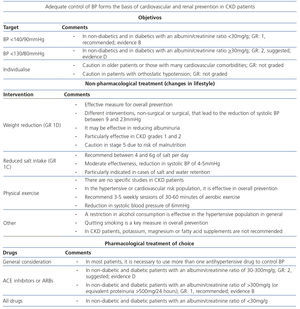
Adequate blood pressure (BP) control is the basis for cardiovascular, renal and overall prevention in CKD patients. The BP control target is <140/90mmHg in patients with an albumin/creatinine ratio <30mg/g, whether or not they are diabetics (GR 1B), and <130/80mmHg in patients with an albumin/creatinine ratio ≥30mg/g, both in non-diabetics and diabetics alike (GR 2D). The previous BP target <130/80 mmHg for all CKD patients, independently of the level of albuminuria or proteinuria, was a recommendation based particularly on observational data. However, recent data have questioned whether this target is beneficial for patients with CKD and albuminuria <30mg/g24,25, and as such, it has been proposed that the targets recommended for hypertensive patients in general be applied to CKD patients with normal albuminuria. With regard to patients with increased albuminuria or proteinuria, the suggested BP target <130/80mmHg is recognised as an expert recommendation. The BP control target in CKD patients continues to be the subject of debate26. In fact, three recent guidelines recommend BP control <140/90mmHg for hypertensive patients in general, including CKD patients27-29.
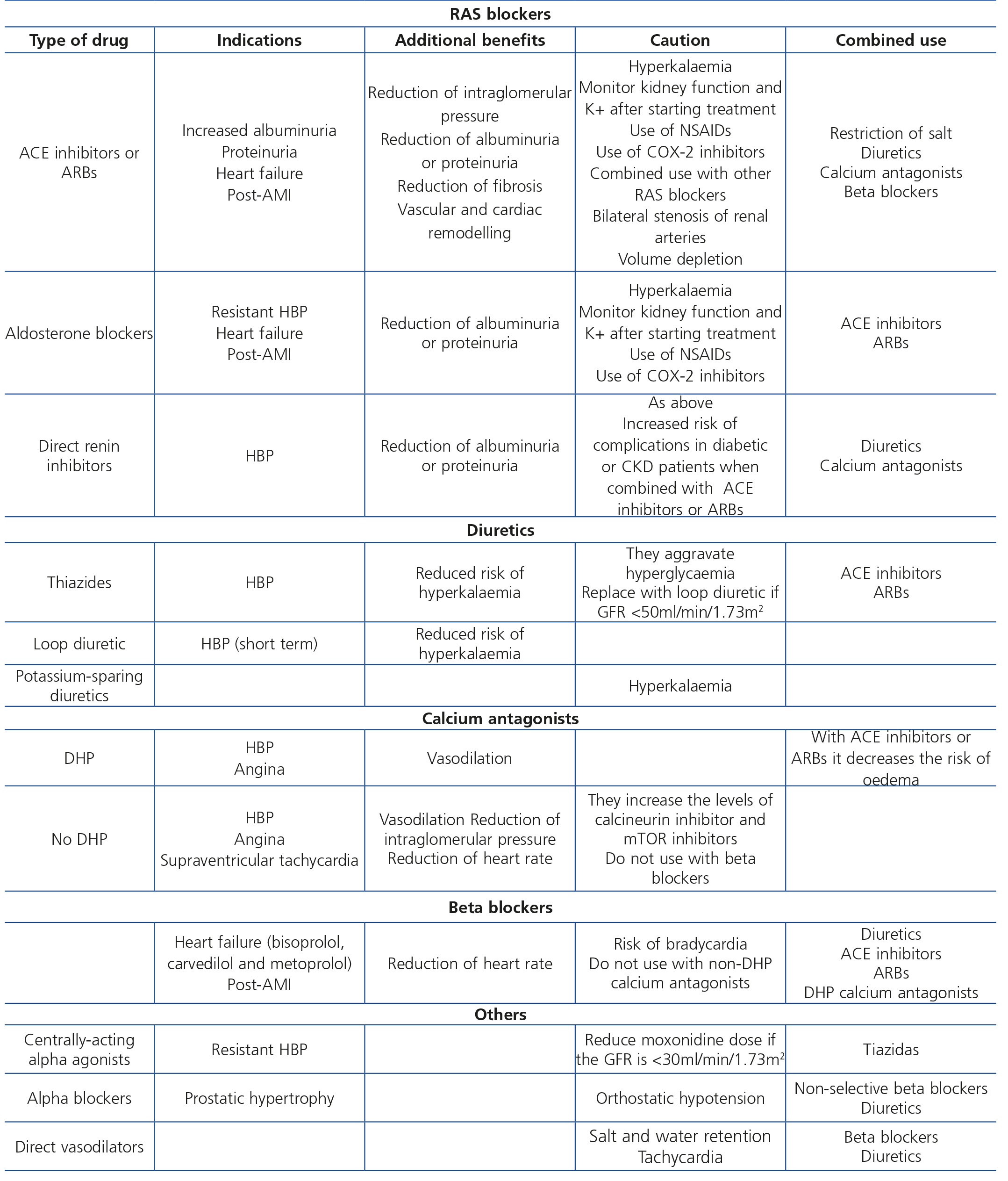
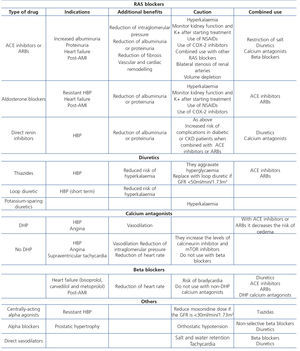
Achieving these targets is based on an individualised approach that includes non-pharmacological measures (changes in lifestyle) and pharmacological treatment. The introduction of lifestyle changes may reduce BP figures simply, cheaply and effectively and is usually accompanied by other beneficial effects30,31. With regard to pharmacological treatment, the choice of drugs that must be used should be individualised according to age, tolerance and patient comorbidities. RAS blockers, angiotensin-converting enzyme inhibitors (ACE inhibitors) and angiotensin receptor blockers (ARBs) form the basis of pharmacological antihypertensive treatment in both non-diabetic and diabetic patients with an albumin/creatinine ratio ≥30mg/g. This type of drug is suggested as the first choice in patients with an albumin/creatinine ratio between 30 and 300mg/g (GR 2D), while in patients with an albumin/creatinine ratio >300mg/g, or equivalent proteinuria (>500mg/24 hours), it is recommended (GR 1B). Independently of the type of drug chosen as the first line of treatment, most patients will require more than one antihypertensive drug for adequate control of HBP. With regard to dual blockade using ACE inhibitors and ARBs, there is not enough evidence to recommend this combination in the prevention of CKD progression, but there is with regard to potential adverse effects, such as acute deterioration of kidney function or hyperkalaemia. Table 4 displays the key aspects of HBP management in CKD patients. A comprehensive review of the preferential indications, dose, adverse effects and contraindications of the different types of antihypertensive drugs are not included in the objectives of these guidelines. However, Table 5 summarises some of the basic aspects of each antihypertensive drug group.
Management of high blood pressure in elderly patients
Despite the high prevalence of HBP and CKD in individuals ≥65 years of age and especially in very elderly individuals (≥80 years of age), there is not enough evidence to develop recommendations on their management32. Antihypertensive treatment in this type of patient involves particular step therapy and monitoring of potential adverse effects, such as electrolyte imbalances, renal failure aggravation and orthostatic hypertension (GF, not graded). A strict control of BP and the use of RAS blockers, which are key in managing adult CKD patients, may not have the same benefits for very elderly patients, and may even have harmful effects33. In the latter, there is no evidence to establish recommendations, and as such, their management should be particularly individualised34.
Management of high blood pressure in kidney transplant patients
HBP in transplant patients is a risk factor for cardiovascular graft function deterioration35. It is suggested that kidney transplant patients with BP >130/80mmHg be treated with the aim of maintaining BP <130/80mmHg independently of the level of urine albumin excretion (GR 2D). The choice of antihypertensive drug must take into account the time since transplantation, the presence or absence of increased albuminuria, use of calcineurin inhibitors and the presence of other comorbidities. When less than two years have elapsed since transplantation, calcium antagonists may have a beneficial effect added to the placebo effect or that of RAS blockers36, with dihydropyridine calcium antagonists being preferable. During the first months after transplantation, RAS blockers may have a harmful effect on kidney function recovery. However, this may subsequently be the drug type of choice, particularly in patients with increased urine albumin or protein excretion.
Cardiovascular risk in chronic kidney disease
CKD patients, particularly those with grades 3a to 5, have an increased cardiovascular risk corresponding to the accumulation of classic factors such as HBP or diabetes, and as such, they must be considered as high cardiovascular risk patients (GR 1A). Elevated albuminuria increases the risk independently of the GFR37. The risk of having a major cardiovascular complication increases from 43% in CKD grade 3a to >300% in grades 4-5 with respect to individuals without CKD38. In fact CKD patients have a greater risk of cardiovascular mortality than requiring renal replacement therapy via dialysis or renal transplantation39. For these reasons, it is recommended to consider any individual with an estimated GFR <60ml/min/1.73m2 as a high cardiovascular risk patient. There must be a comprehensive and structured plan for CKD patients for reducing cardiovascular risk, which must include abstaining from smoking, exercise, weight control, lipid profile control, optimal control of diabetes and BP, anaemia correction, phosphorus-calcium metabolism control and platelet anti-aggregation in secondary prevention. It is also advised for patients with CKD and an acute coronary event to receive the same level of diagnostic and therapeutic intervention as those without CKD (GR 1A). With regard to treatment of patients with CKD and heart failure, a similar level of intervention is suggested to that of patients without CKD (GR 2A), although any increase in treatment or any clinical deterioration must be accompanied by a stricter control of kidney function and serum potassium. Some diagnostic tests, such as those of troponins or BNP/NT-proBNP (B-type natriuretic peptide/N-terminal-proBNP), should be interpreted with caution in CKD patients, particularly those with an estimated GFR <60ml/min/1.73m2, since the standard reference values may not have the same significance as in patients without CKD.
Nutritional and metabolic control
Control of obesity is a main objective in treating CKD patients, both as a measure of cardiovascular and overall prevention and to slow down renal failure progression (GR 1D).
A reduction in salt intake to between 4 and 6g per day is recommended, unless contraindicated (GR 1C).
High dietary protein intake in CKD patients results in an accumulation of uraemic toxins, but insufficient intake may lead to malnutrition. It is suggested to reduce protein intake to 0.8g/kg/day in adults with an estimated GFR <30ml/min/1.73m2 (CKD grades 4-5) without evidence or risk of malnutrition (GR 2C).
In diabetic patients, a glycated haemoglobin (HbA1C) target <7% (GR 1A) is recommended, except in frail patients at risk of hypoglycaemia or with major comorbidities that may reduce life expectancy, for whom the HbA1C target is between 7.5% and 8% (GR 2C). In very elderly and frail patients, a more relaxed HbA1C target of <8.5% may be considered40.
With regard to hyperuricaemia treatment, it is considered that there is not enough evidence that supports or rejects the use of hypouricaemic drugs for slowing down CKD progression. However, it is indicated that in patients with CKD and symptomatic hyperuricaemia (uric acid lithiasis or gout), uric acid levels of less than 7mg/dl should be achieved. To achieve this target, patients are treated with xanthine oxidase inhibitors with doses adjusted according to kidney function. The reduction of uric acid below 7mg/dl could decrease cardiovascular risk and slow down CKD progression in patients with asymptomatic hyperuricaemia. Despite there being data in the literature that support this, there are no interventional studies with a sufficient sample for recommending the use of hyperuricaemic drugs with this objective41.
Patients with advanced CKD grades may require specific dietary advice simultaneously (salt, carbohydrates, proteins, potassium, phosphorus), and will receive specialised and individualised care (GR 1B).
Anaemia
Anaemia contributes significantly to the symptoms and quality of life of patients and has a major impact on the prognosis of CKD. Serum haemoglobin tests should be carried out at least once a year in patients with grades 3a and 3b and at least once every six months in patients with an estimated GFR <30ml/min/1.73m2. The treatment and therapeutic targets mentioned are addressed in the corresponding guidelines42, although the key aspects include: 1) ruling out secondary causes, mainly iron deficiency, 2) giving patients iron supplements in cases of deficiency and 3) using erythropoiesis-stimulating agents with target haemoglobin not exceeding 11.5g/dl. Treatment with erythropoiesis-stimulating agents is not recommended in cases of active malignant disease. Basic studies on assessing anaemia in CKD patients include a complete blood count and testing levels of reticulocytes, serum ferritin, the transferrin saturation index, vitamin B12 and folate. The first step in treating anaemia associated with CKD is iron supplements if the transferrin saturation index is ≤30% and serum ferritin is ≤500ng/ml. Treatment with erythropoiesis-stimulating agents is assessed with haemoglobin levels <10g/dl42. The GR and levels of evidence corresponding to these considerations on anaemia in CKD are displayed in the corresponding KDIGO guidelines42.
Mineral and bone metabolism disorders
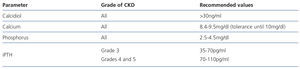
Mineral and bone metabolism disorders may begin at initial CKD grades and increase as the disease progresses (Table 3). These changes are grouped under the heading of mineral and bone metabolism disorders and include related renal osteodystrophy and extraskeletal (vascular) calcifications. Renal osteodystrophy in turn includes osteitis fibrosa cystica (hyperparathyroidism), osteomalacia and adynamic bone disease. The current KDIGO guidelines refer to the recommendations of the previous specific guidelines43, which is also expressed in the corresponding S.E.N. guidelines44. It is advised to assess serum levels of calcium, phosphorus, alkaline phosphatase, intact parathyroid hormone (iPTH) and vitamin D (25 OH D3) in patients with an estimated GFR <45ml/min/1.73m2 (grades 3b to 5). In patients with these CKD grades, it is suggested to maintain phosphorus concentration within the normal range. The absence of evidence with regard to the optimal level of iPTH in these patients is recognised, and as such, if an increased iPTH level is detected in these patients, it is suggested to assess the possibility of hyperphosphataemia, hypocalcaemia and vitamin D deficiency. Table 6 displays the values of these parameters recommended in the S.E.N. guidelines for managing mineral and bone metabolism disorders in CKD patients not on dialysis44. With regard to the indication of testing mineral and bone density metabolism and using bisphosphonates, it is suggested not to indicate bone density scans routinely in patients with an estimated GFR <45ml/min/1.73m2 and to avoid the aforementioned prescription in patients with an estimated GFR <30ml/min/1.73m2. The GR and levels of evidence corresponding to these considerations on mineral and bone density metabolism disorders in CKD are displayed in the corresponding KDIGO guidelines43.
Acidosis
As with other aforementioned complications, the prevalence and severity of acidosis increases as CKD deteriorates (Table 3). Treatment with oral bicarbonate supplements in patients with bicarbonate concentrations <22mEq/l is suggested, if it is not contraindicated.
OTHER SAFETY ASPECTS IN CHRONIC KIDNEY DISEASE PATIENTS
CKD patients are vulnerable to certain very common situations in healthcare and they must therefore receive the appropriate preventive measures.
Risk of acute deterioration of kidney function
Kidney function deterioration worsens the prognosis of any acute or chronic pathology. It is recommended to consider all CKD patients as a population at risk of acute renal failure (GR 1A). This consideration should particularly be taken into account in cases of intercurrent disease and, above all, in any situation of hospitalisation or any diagnostic or therapeutic procedure.
Use of drugs in kidney disease patients
The main recommendations with regard to the use of drugs in CKD patients are: 1) use GFR for drug dose calculation (GR 1A); 2) temporarily discontinue potentially nephrotoxic treatment or treatment that is preferentially eliminated via the kidneys in patients with an estimated GFR <60ml/min/1.73m2 in circumstances of severe intercurrent disease, given the risk of acute function deterioration; the drugs that must be taken into account in this recommendation are mainly ACE inhibitors, ARBs, aldosterone antagonists, diuretics, non-steroidal anti-inflammatory drugs, metformin, lithium and digoxin (GR 1C); 3) do not use herbal medicine; 4) do not use metformin in patients with a GFR <45ml/min/1.73m2; and 5) monitor kidney function, electrolytes and drug levels in patients who receive potentially nephrotoxic drugs, mainly aminoglycoside antibiotics, lithium, calcineurin inhibitors and digoxin.
Use of radiological contrasts
In patients with a GFR <60ml/min/1.73m2 who are going to receive an iodine contrast, it is recommended to avoid high osmolarity agents, use the minimum possible dose of the radiocontrast agent, discontinue potentially nephrotoxic drugs beforehand, particularly metformin, administer adequate hydration with saline solution before, during and after the procedure and monitor the GFR 48-96 hours after the latter (GR 1, A to C according to each specific recommendation). The use of N-acetylcysteine or ascorbic acid as prophylaxis for nephropathy due to iodine contrasts has not consistently demonstrated to be beneficial and as such, it has not been included as a recommendation in the KDIGO guidelines. However, the guidelines of the S.E.N. on acute renal failure recommend prophylaxis with N-acetylcysteine before the administration of the iodine contrast and it is assigned a B evidence level45.
It is recommended to avoid the use of gadolinium-based contrasts in patients with an estimated GFR <15ml/min/1.73m2 (GR 1B), except when there is no alternative. In patients with an estimated GFR <30ml/min/1.73m2, it is advised to avoid the use of gadodiamide and give preference to other preparations, such as gadoteridol, gadobutrol o gadoterate. Although the KDIGO guidelines do not make recommendations for dialysis patients, they recommend carrying out a dialysis session immediately after the procedure and probably also after 24 hours. The role of dialysis in patients with an estimated GFR <15ml/min/1.73m2 who are not receiving renal replacement therapy is uncertain.
Likewise, it is recommended not to use oral preparations with phosphates for intestinal preparation in patients with an estimated GFR <60ml/min/1.73m2 (GR 1A). It is currently debated whether the potential kidney damage is due to dehydration caused by these compounds more than by the phosphorus itself. For enemas, preparations without phosphate may be safer (fisioenema). For oral preparation, there are no phosphate-free preparations, and as such, it is recommended to avoid dehydration.
Vaccinations
Unless contraindicated, it is recommended to vaccinate against the flu in patients with a GFR <60mlmin/1.73m2, against pneumococcal infection in patients with a GFR <30ml/min/1.73m2 and in high risk cases such as patients with nephrotic syndrome, diabetes or those who receive immunosuppressive treatment, and against hepatitis B in cases with a GFR <30ml/min/1.73m2 and risk of progression.
REFERRAL TO THE NEPHROLOGIST
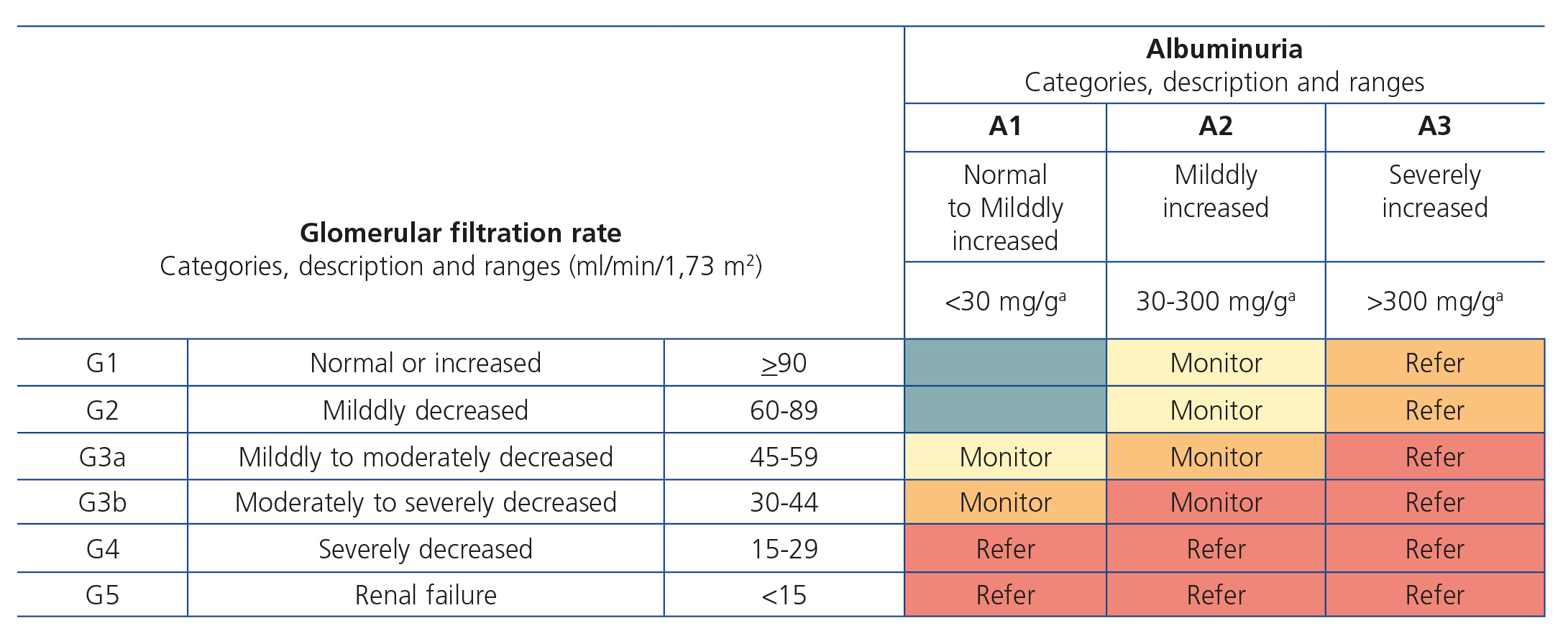
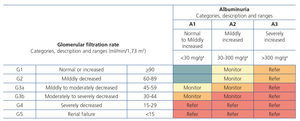
Table 7 displays the criteria for referring CKD patients to Nephrology (GR 1B). Other situations may be managed by other doctors, mainly Family and Community Medicine specialists, who should regularly monitor patients, in accordance with that stated in the corresponding section. Figure 2 indicates the recommendations for monitoring or referral in accordance with the CKD grade. These referral criteria are basically the same as those recommended in the aforementioned 2008 S.E.N.-semFYC consensus document on CKD of ten Spanish scientific societies7,12.
Conflicts of interest
The authors declare that they have no conflicts of interest related to the contents of this article.
Table 2. Classification into chronic kidney disease grades
Table 3. Prevalence of common complications of chronic kidney disease according to the glomerular filtration grades
Table 4. Key aspects of high blood pressure management in chronic kidney disease
Table 5. Indications, additional benefits, caution and combined use of the different antihypertensive drug groups in chronic kidney disease patients
Table 6. Values recommended in the guidelines of the Spanish Society of Nephrology for managing mineral and bone metabolism disorders in chronic kidney disease patients not on dialysis
Table 7. Criteria for referral to the nephrologist
Figure 1. Prognosis of chronic kidney disease according to the glomerular filtration rate and albuminuria categories.
Figure 2. Recommendations for the referral of chronic kidney disease patients to the nephrologist according to glomerular filtration rate and albuminuria categories.
GRADE system
Working group for the creation of this document
12464_19157_57549_en_ref.1246428399_12464_19115_56224_es_12464_tabla1.doc
Table 1. Chronic kidney disease diagnosis criteria