Introducción y objetivos: La enfermedad arterial coronaria es una de las principales causas de morbilidad y mortalidad en pacientes diabéticos candidatos para trasplante renal. La alta prevalencia de enfermedad coronaria en pacientes asintomáticos obliga a hacer despistaje de coronariopatía significativa. Nuestro objetivo es conocer la prevalencia y los factores pronósticos asociados a enfermedad coronaria en este grupo de pacientes en nuestro medio. Métodos: Estudio observacional, descriptivo y retrospectivo de los aspectos epidemiológicos y clínicos de los pacientes diabéticos con enfermedad renal crónica candidatos a trasplante renal entre enero de 2007 y octubre de 2011. Resultados: Se analiza una cohorte de 36 pacientes. El 65% (13) con diabetes mellitus tipo 1 y el 81,3% (13) con diabetes mellitus tipo 2 presentan enfermedad coronaria significativa. En el análisis multivariado de regresión logística, se asociaron significativamente con enfermedad coronaria el tabaquismo (odds ratio [OR] = 8,3, p = 0,048) y los niveles de hemoglobina glicosilada (OR = 9,525, p = 0,006). Factores que no se asociaron significativamente a enfermedad coronaria incluyen: edad, sexo, tipo de diabetes mellitus, duración de diabetes mellitus (años) e hipertensión arterial. Conclusión: Los pacientes diabéticos sin clínica anginosa con enfermedad renal crónica candidatos a inclusión en lista de trasplante renal presentan una alta prevalencia de enfermedad arterial coronaria significativa. El tabaquismo y los niveles de hemoglobina glicosilada se asocian de forma independiente con la presencia de enfermedad arterial coronaria.
Introduction and objectives: Coronary artery disease is a major cause of morbidity and mortality in diabetic kidney transplant candidates. The high prevalence of coronary disease in asymptomatic patients creates the need for major coronary artery disease screening. Our goal was to determine the prevalence and prognostic factors associated with coronary disease in this patient group. Method: A retrospective study of a cohort of 36 asymptomatic patients with diabetes mellitus type 1 and 2 and chronic renal failure that were candidates for renal transplantation between January 2007 and October 2011. Results: We followed a cohort of 36 patients. Significant coronary disease was found in 65% (13) of patients with type 1 diabetes mellitus and 81.3% (13) with type 2 diabetes mellitus. In the multivariate logistic regression analysis, smoking (OR=8.3, P=.048) and glycosylated haemoglobin levels (OR=9.525, P=.006) were significantly associated with coronary artery disease. Factors not significantly associated with coronary artery disease included: age, sex, type of diabetes mellitus, duration of diabetes mellitus (years) and hypertension. Conclusion: Diabetic patients without clinical angina and chronic renal failure who were candidates for inclusion in the kidney transplant waiting list have a high prevalence of significant coronary artery disease. Smoking and glycosylated haemoglobin levels were independently associated with the presence of coronary artery disease.
INTRODUCTION
Patients with diabetes mellitus (DM) have a substantially higher risk of developing end-stage chronic kidney disease (ESCKD) and cardiovascular disease, with increased associated morbidity and mortality rates.1-3 Kidney transplantation is the treatment of choice for these patients, since it effectively reduces mortality as compared to diabetic and non-diabetic patients that remain on dialysis treatment.4,5
Approximately half of all patients with ESCKD have cardiovascular disease when they start dialysis treatment.6 One third of all diabetics with ESCKD have coronary artery disease (CAD) that is clinically silent.6 Despite kidney transplantation, the percentage of deaths due to cardiovascular disease continues to be a source of concern in the initial post-transplant phase.7-9
Although current guidelines suggest a selective conservative approach for the risk stratification of this population, few specific tests exist for this sub-group of patients.8 Several studies have evaluated the sensitivity of at-rest electrocardiograms and non-invasive procedures, coming to the conclusion that these testing methods are not adequate in diabetic patients with ESCKD.9-11 As a result, there is still no consensus regarding what type of screening method is most appropriate for ruling out ischaemic pathologies in these patients and predicting post-transplant coronary complications. Catheterisation involves substantial associated morbidity rates, including contrast-induced nephropathy, and can accelerate the need for starting dialysis. However, the benefits of coronary angiography outweigh the risks in patients with ESCKD.12-15
PATIENTS AND METHOD
Study population
We carried out an observational, descriptive, retrospective study on the epidemiological and clinical aspects of diabetic patients with ESCKD. We reviewed the clinical histories of asymptomatic diabetic patients with ESCKD who underwent pre-kidney transplant examinations in the Hospital General de Ciudad Real between January 2007 and October 2011. The criteria for indicating CAD screening by cardiac catheterisation were: age (>45 years) and duration of diabetes (>15 years with type 1 DM, >10 years with type 2 DM and no other cardiovascular risk factors, >5 years with type 2 DM and other cardiovascular risk factors). A total of 36 patients were identified with DM and no history of angina, myocardial infarction, or revascularisation with percutaneous or surgical coronary angioplasty.
Clinical and demographic data
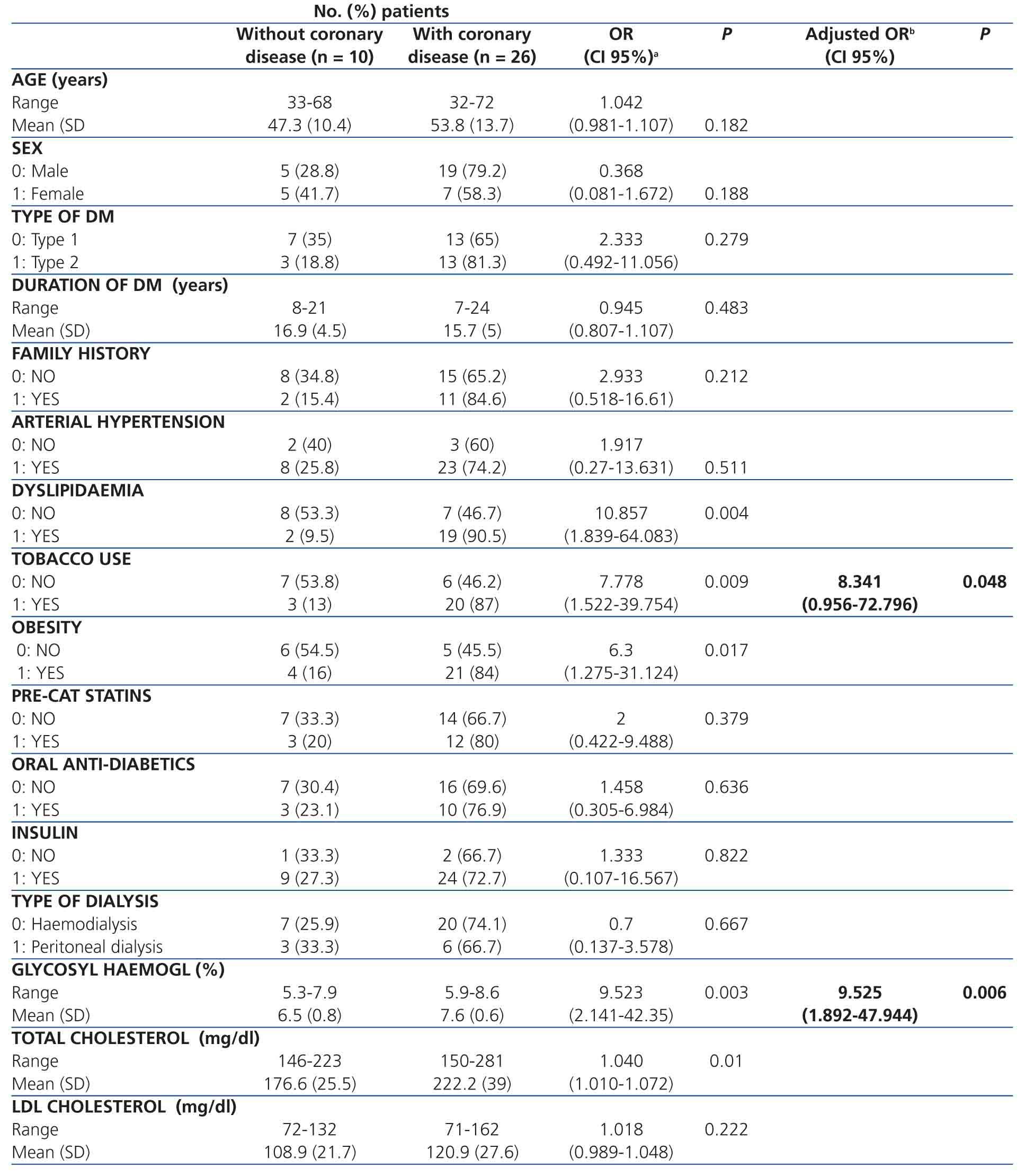
We measured the following clinical and demographic variables: age (years), gender, type of DM (type 1 or 2), duration of DM (years), family history of coronary disease, arterial hypertension, dyslipidaemia, tobacco use (including patients that quit using tobacco within the last 10 years), obesity (body mass index >25kg/m2; as such including overweight and obese patients), pre-catheterisation treatment with statins, oral anti-diabetic and/or insulin treatment, type of dialysis (haemodialysis, peritoneal dialysis), glycosylated haemoglobin (%), total cholesterol (mg/dl), and low-density lipoprotein (LDL) cholesterol (mg/dl).
The results of the cardiac catheterisation and the dates of the procedure were obtained from the haemodynamics department. The percentage of stenosis required for diagnosing significant CAD was: >50% in the left coronary artery (LCA) and the proximal segment of the left anterior descending (LAD) artery and >70% in all other segments. The coronary disease distribution was described based on the affected vessel: LCA, LAD artery, circumflex artery (CX), and right coronary (RCA) artery. In addition, left ventricular ejection fraction was measured using ventriculography (%) and level of ventricular dysfunction (mild: 45%-55%, moderate: 30%-45%, and severe: >30%).
Statistical analysis
We evaluated continuous variables for normal distribution using the Kolmogorov-Smirnov test. Continuous variables with a normal distribution were presented as mean ± standard deviation. Discrete variables were presented as frequencies (percentages) and compared using chi-square tests. We used Student’s t-tests for comparing continuous variables. The relationship between pairs of qualitative variables was assessed using contingency tables and Pearson’s chi-square tests, considering significant any P-value <0.05.
We also performed a multivariate analysis using multiple logistic regression analyses to evaluate which factors were associated with significant CAD in diabetic ESCKD patients on the kidney transplant waiting list. We considered the presence of significant coronary disease (1: yes; 0: no) as the dependent variable. The independent variables considered were: dyslipidaemia (1: yes; 0: no), tobacco use (1: yes, 0: no), glycosylated haemoglobin (%), and total cholesterol (mg/dl). These variables were fed into a multiple logistic regression model; using the Wald statistic, and variables with a P≥.15 (using a backwards selection procedure) were eliminated from the model. We used likelihood ratios to compare the final reduced model with the model containing all variables.
The scale of the different continuous variables was evaluated using Box Tidwell tests. We studied possible interactions between variables, and variables with a significance level greater than 0.05 were examined as possible confounding factors. They were considered confounding factors if the change coefficients were >15%. We used the Cook distance test in extreme cases. We also used the Hosmer–Lemeshow test based on percentiles to evaluate the goodness of fit of the model.
RESULTS
The demographic and clinical characteristics of the sample were: mean age was 52±13 years (range: 32-72 years); 66.7% were male, 55.6% had type 1 DM, mean time of DM was 16±4.8 years (range: 7-24 years); 75% were on haemodialysis, 36% had a family history of CAD, 86.1% had hypertension, 58.3% had dyslipidaemia, 52.8% were smokers, and 63.9% were obese. As regards the pre-catheterisation treatment, 41.7% of patients received statins, 36.1% received oral anti-diabetics, and 94.4% were on treatment with insulin. The biochemical test results showed the following mean values: glycosylated haemoglobin: 6.9%±0.8%; total cholesterol: 209.46±41.05mg/dl; and LDL cholesterol: 117.58±26.34mg/dl).
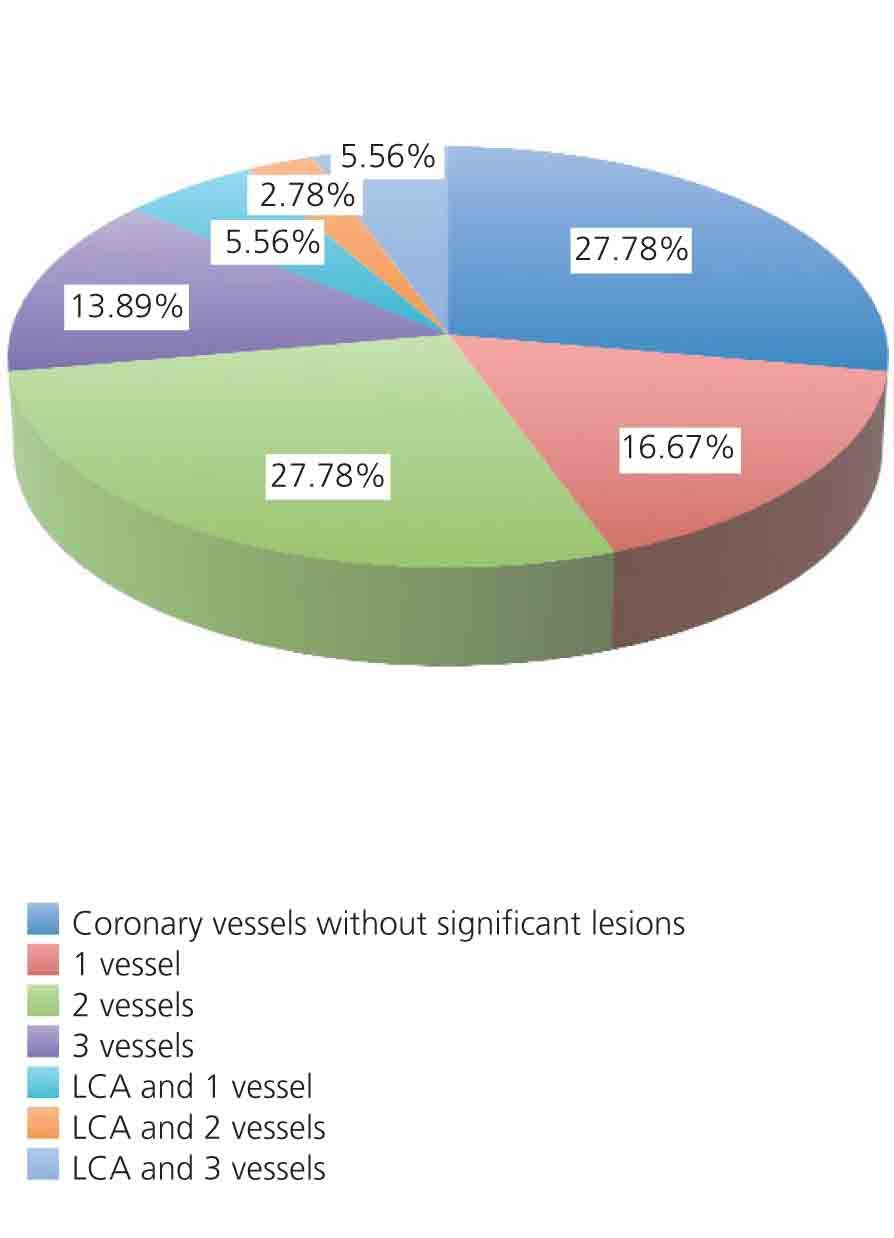
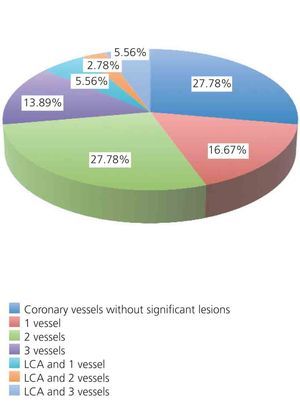
The distribution of coronary vessels with significant damage (>50% in proximal LAD and LCA; >70% in all other segments) is shown in Figure 1. Of the 36 patients in the study, 26 (72.2%) had significant CAD in one or more vessels. According to the number of vessels, we found that: LCA disease and 1 vessel in 2 patients (5.6%), LCA and 2 vessels in 1 patient (2.8%), and LCA and 3 vessels in 2 patients (5.6%). Coronary disease different from LCA (LAD, RCA, CX) were as follows: disease in 1 vessel in 6 patients (16.7%), 2 vessels in 10 patients (27.8%), and 3 vessels in 5 patients (13.9%). The distribution of significant lesions by coronary artery was as follows: significant LAD disease in 23 patients (63.9%), CX in 18 patients (50%), and RCA in 10 patients (27.8%). Mean ventricular ejection fraction (by ventriculography) was 50.28%±12.12% (range: 20-65), and ventricular dysfunction was distributed in the following manner: no ventricular dysfunction: 20 patients (55.6%); mild dysfunction: 8 patients (22.2%), moderate dysfunction: 3 patients (8.3%); and severe dysfunction: 5 patients (13.9%).
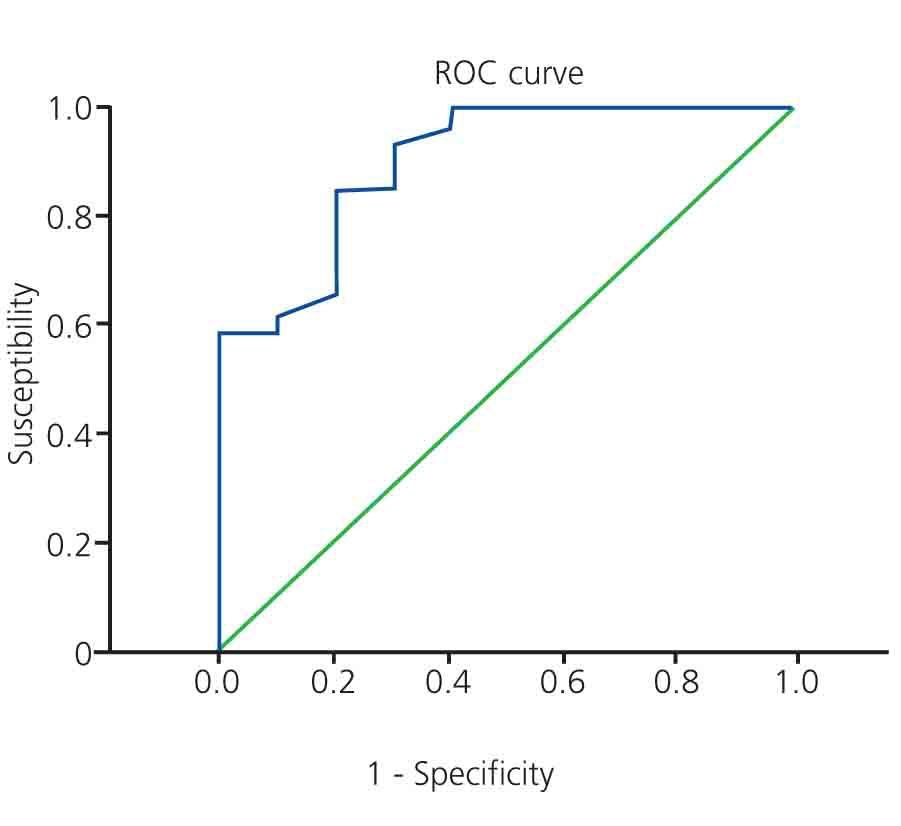
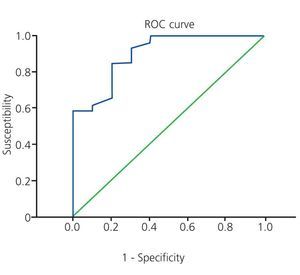
The summary results for the multiple logistic regression analysis are shown in the Table. The univariate analysis of factors associated with significant CAD revealed that 65% of patients with type 1 DM and 81.3% of patients with type 2 DM had significant coronary artery lesions, with no significant difference between these two groups (P=.279). We did not observe a significant difference based on the following variables: age (odds ratio [OR] =1.042; P=.182), female sex (OR=0.368; P=.188), family history of coronary disease (OR=2.933; P=.212), arterial hypertension (OR=1.917; P=.511), or LDL cholesterol levels (OR=1.018; P=.222). The only variables that were significantly associated with significant coronary artery disease were: dyslipidaemia (OR=10.857; P=.004), tobacco use (OR=7.778; P=.009), glycosylated haemoglobin (OR=9.523; P=.003), and total cholesterol (OR=1.040; P=.01). The latter variables were then included in the multiple logistic regression model. Dyslipidaemia and total cholesterol were excluded from the model (likelihood ratio =1.556, degrees of freedom [DF] =2). All variables had a linear scale. We evaluated all possible interactions which were not significant. No patients had a Cook distance greater than 1. The final result showed that the only variables significantly associated with CAD were: tobacco use (adjusted OR=8.341; P=.048) and percentage of glycosylated haemoglobin (adjusted OR=9.535; P=.006). For every 1% increase in the value of glycosylated haemoglobin, the risk that an asymptomatic diabetic patient with ESCKD will develop significant CAD is 9.525 times higher if all other variables are the same (for a 95% confidence interval, this difference would be 1.892-47.944). The ROC curve for this calculation is shown in Figure 2.
DISCUSSION
Ischaemic cardiopathy is a common cause of mortality and morbidity in patients with ESCKD.15 The prevalence of asymptomatic CAD in diabetic patients with ESCKD was approximately 70% in our study. In addition, these patients have a lower survival rate after an acute coronary event.15 The diagnosis of CAD in patients with ESCKD, especially in diabetics, continues to be a substantial challenge. Previous studies have shown that non-invasive methods for diagnosing CAD, including clinical histories and at-rest/stress electrocardiograms, are not reliable for detecting coronary disease in diabetic patients with ESCKD.16,17 Studies of ischaemia using stress echocardiograms and radionuclide methods differ in terms of sensitivity and predictive power, although in general they show a low negative predictive value.18
As regards risk factors, it has been well established that diabetic patients with ESCKD have multiple associated cardiovascular risk factors. Despite the high prevalence of CAD, many diabetic patients with ESCKD do not show angina clinically as a consequence of the advanced diabetic nephropathy, which could be explained by the high level of inactivity and reduced myocardial demand in these patients. Additionally, the constitutional symptoms associated with ESCKD may lead to an erroneous interpretation of symptoms. These factors increase the complexity of the clinical evaluation of these patients prior to kidney transplantation.19
Grupa et al. evaluated the clinical factors associated with mortality following kidney transplants. Their study revealed that this population is at high risk of cardiovascular events and has higher mortality and morbidity rates that other populations.20
Cardiac catheterisation is the standard procedure for a diagnosis of CAD. However, this is a costly examination and may also cause progression of renal failure, among other complications. Despite this, the benefits obtained from the diagnostic and therapeutic potential of this procedure outweigh the risks to the patient. In many hospitals, such as ours, cardiac catheterisation is indicated in diabetic patients with ESCKD due to the low sensitivity of non-invasive cardiac tests. Our study confirms the high prevalence of silent CAD in this population. Silent CAD was found in 65% of type 1 diabetics and 81.3% of type 2 diabetics.
Tobacco use and high levels of glycosylated haemoglobin during the pre-transplantation evaluation are associated with CAD in these patients. The univariate analysis also showed that obese patients, patients with dyslipidaemia, and those with high total cholesterol levels should be closely evaluated.
Our study had several limitations. Firstly, we used a retrospective study design, based on reviewing clinical histories and reports from cardiac catheterisations. Secondly, the small sample size greatly limited the statistical power of our tests, the inclusion of variables, and the analysis of these variables in the multiple logistic regression model. Thirdly, we did not include information regarding patient evolution and prognosis. Finally, all patients in our study underwent direct cardiac catheterisation. While our study did not intend to validate non-invasive tests, a comparative study would be useful for constructing an algorithm for the evaluation of coronary disease in diabetic patients.
In conclusion, we have demonstrated the high prevalence of significant CAD in asymptomatic diabetic patients with no history of ischaemic heart disease who are candidates for kidney transplants. We identified a sub-population of greater risk: patients with a history of tobacco use with high levels of glycosylated haemoglobin.
Conflicts of interest
The authors affirm that they have no conflicts of interest related to the content of this article.
Table 1. Factors associated with coronary disease in asymptomatic pre-kidney transplant diabetic patients
Figure 1. Percentage of vessels with significant disease
Figure 2. ROC curve