Objetivos: Determinar la prevalencia de insuficiencia renal en población mayor de 49 años de edad con enfermedad arterial periférica y analizar su relación con factores de riesgo y enfermedad cardiovascular. Material y métodos: Estudio epidemiológico prospectivo de 3 años de duración con selección por muestreo aleatorio simple en población general de edad superior a 49 años. Se recogieron datos demográficos, clínicos, prevalencia de factores de riesgo y enfermedad cardiovascular. Se definió enfermedad arterial periférica (EAP) por índice tobillo-brazo ≤ 0,9 e insuficiencia renal crónica (IRC) según valores de filtrado glomerular estimado por la fórmula MDRD < 60 ml/min/1,73 m2. Se presentan datos basales. Resultados: Se incluyeron 511 personas de 66,6 (9,7) años de edad media (desviación estándar), 37% varones. La prevalencia de EAP fue del 12,4% (n = 63) de la muestra, de 72,6 años de edad media, 46% varones. La presencia de enfermedad renal crónica en estadios 3-5 fue del 39,7%. Los pacientes con EAP e IRC, en comparación con los que tenían función renal normal, eran de mayor edad (75,6 vs. 70,6, p = 0,08), predominantemente mujeres (64 vs. 47,4%, p = no significativa [ns]), presentaban cifras más elevadas de presión arterial sistólica y diastólica (159,2 vs. 146,1, p = ns y 85,7 vs. 80,3 mmHg, p = 0,09), proteína C reactiva (1,23 vs. 0,38 mg/dl, p = 0,05) y cociente albúmina-creatinina (90,2 vs. 26,4 mg/g, p = ns). En este grupo, la prevalencia de factores de riesgo cardiovascular y enfermedad cardiovascular asociada fueron notablemente superiores, sin alcanzar significación estadística. Conclusiones: La insuficiencia renal está presente en el 39,7% de los pacientes con EAP y delimita un subgrupo de pacientes con elevado riesgo cardiovascular.
Objectives: To determine the prevalence of kidney failure in people older than 49 years old with peripheral arterial disease and to analyse its relationship with risk factors and cardiovascular disease. Material and method: Prospective epidemiological study 3 years in duration with selection by simple random sampling in the general population aged over 49 years. Data on demographic, clinical, prevalence of risk factors and cardiovascular disease were registered. We defined peripheral arterial disease (PAD) by ankle-brachial index (ABI) <_0.9 and chronic kidney disease (CKD) according to estimated glomerular filtration rates by the MDRD <60 ml/min/1.73m2. Baseline data are presented. Results: 511 people were included. The mean age was 66.6 (9.7) years (SD), 37% were men. The prevalence of PAD was 12.4% (N=63) of the sample, average age 72.6 years, 46% men. The presence of CKD stages 3-5 was 39.7%. Patients with PAD and CKD compared with those with normal renal function were older (75.6 vs. 70.6, p=0.08), predominantly women (64% vs 47.4%, p=ns), and showed higher values for systolic and diastolic blood pressure (159.2 vs 146.1, p=ns and 85.7 vs 80.3 mmHg, p=0.09), CRP (1.23 vs 0.38 mg/dl, p=0.05) and albumin creatinine ratio (90.2 vs 26.4 mg/g, p=ns). In this group, the prevalence of cardiovascular risk factors and associated cardiovascular disease, were significantly higher without reaching statistical significance. Conclusions: Kidney failure is present in 39.7% of patients with PAD and it defines a subgroup of patients with high cardiovascular risk.
INTRODUCTION
In clinical practice, the term "peripheral arterial disease" tends to refer to vascular disease in the lower limbs, defined as being caused by stenosis or thrombosis of leg arteries, and one manifestation of systemic atherosclerosis.1
Although the presence of intermittent claudication has traditionally been used as a Peripheral arterial disease (PAD) marker to give an approximate calculation of its prevalence in the general population, the ankle-brachial index (ABI) is currently considered to have more diagnostic precision, and it is recommended for both individual diagnoses and epidemiological studies.1,2 An ABI below 0.9 is 95% sensitive and 100% specific for the presence of stenosis in the vascular array of the lower limbs, which is indicative of atherosclerosis in that region.3,4 ABI is also recognised as a marker of cardiovascular risk, and it has an excellent correlation with the development of coronary artery complications, the incidence rate of strokes and death by cardiovascular or other causes in the general population, particularly in the geriatric population.5,6
It is well understood that the presence of end-stage kidney failure in patients with kidney replacement therapy and PAD is accompanied by increases in lower limb amputations, hospitalisation, and total and cardiovascular mortality.7-9 There is a growing interest in examining the correlation between kidney function and PAD, not only during end-stage kidney failure, but also throughout all stages of kidney disease. The NHANES 1999-2000 epidemiological study analysed the prevalence of PAD in patients with kidney disease, defined as those having an estimated glomerular filtration rate (eGFR) measured with the Cockcroft-Gault formula below 60ml/min/1.73 m2. Prevalence in this population was 24% compared with 3.7% in the population whose eGFR was higher than 60ml/min/1.73 m2 (odds ratio [OR]: 9.7, 95% confidence interval [CI]: 5.6-16.7), P<.001). This correlation was independent from other variables such as age, diabetes, hypertension, hypercholesterolaemia, coronary artery disease and ictus (OR: 2.5, 95% CI: 1.2-5.1), P<.011).10 Very similar results were obtained in NHANES 1999-2004.11 Longitudinal follow-up studies have also shown increased risk of developing PAD in patients with an eGFR below 60ml/min compared to those with a higher glomerular filtration rate (GFR).12,13
Chronic kidney disease (CKD) and PAD have more than just atherosclerosis as a common underlying factor in their anatomical pathology. They also share risk factors such as diabetes, arterial hypertension and smoking, and it is possible that gaining control over these risk factors would reduce or slow the progression of both diseases. This would explain the reciprocal relationship between both entities; that is, patients with reduced kidney function will have higher incidence and prevalence rates for arterial disease, and in turn, kidney failure will be more common in patients with chronic ischaemia of the lower limbs. However, this last postulate has not received much study.
OBJECTIVES
Main objective
Determine the prevalence of chronic kidney disease (CKD) defined as an eGFR <60 ml/min/1.73 m2 in a population aged 49 years and older with PAD.
Secondary objective
Analyse the relationship between patients with PAD and kidney failure and classic cardiovascular risk factors and vascular disease at other levels.
MATERIAL AND METHOD
Population and sample
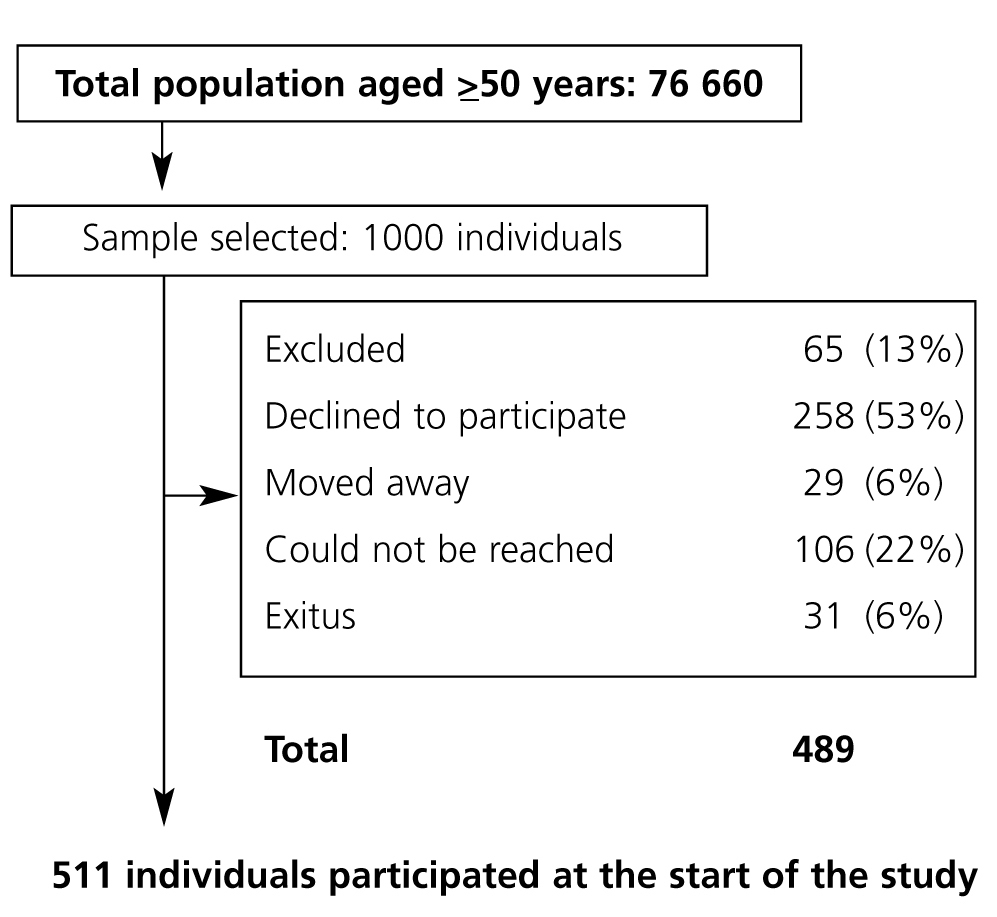
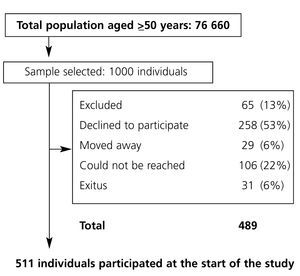
The study population includes all individuals 49 years old or older (n=76 660) residing in Oviedo, Asturias, Spain, identified in the database containing individual health card data. For the purpose of calculating PAD prevalence, 1000 individuals were selected from the initial population through simple random sampling. This number was established based on the following suppositions: estimated prevalence of 0.10 based on other studies, with a Type I error of 0.05 and desired precision 0.03, giving us a sample size (453) that was increased according to an estimate of 60% drop-outs and non-responses. The final sample size was 996 individuals. Terminal and immobilised patients were excluded. Deceased patients, those who moved their residence and those who declined to participate in the study were considered lost and they were not substituted. The study was approved by the Clinical Research Ethics Committee of Asturias.
The main objective of this study is to verify the relationship between PAD and cardiovascular disease in a population 49 years or older during a 3 year follow-up period. This project is a sub-study to determine whether patients with both PAD and chronic kidney disease (CKD) show a more unfavourable risk profile, both at baseline and during disease evolution. Initial data are shown here.
Data registry
We elaborated a protocol for collecting demographic data (age and sex), clinical data (weight, height, body mass index), systolic blood pressure (SBP), diastolic blood pressure (DBP), presence of cardiovascular risk factors (smoking, dyslipidaemia, hypertension, diabetes, obesity), cardiovascular disease (ischaemic heart disease, heart failure, cerebrovascular accident, peripheral arterial disease), in addition to data corresponding to hypertension, dyslipidaemia, diabetes and anti-platelet drugs. All patients underwent an electrocardiogram and a general biochemical study that included serum concentrations of glucose, uric acid, creatinine, total cholesterol, HDL cholesterol, triglycerides, fibrinogen, C-reactive protein and lipoprotein(a).
Hypertension was diagnosed in the presence of SBP figures >140mmHg and/or DBP >90mmHg on 3 separate visits, or in individuals who were already receiving dietary or pharmacological antihypertensive treatment. Arterial blood pressure figures represent average readings from three measurements taken 2 minutes apart.
All subjects who had consumed tobacco during the past month were considered smokers; those who had been smokers but who had not smoked in the past year were considered former smokers.
Subjects with baseline glycaemia figures >126mg/dl on 2 occasions, or with a an oral glucose tolerance test reading above 200mg/dl at 2 hours were considered diabetic, along with subjects already receiving anti-diabetic treatment, whether in the form of insulin or oral anti-diabetic drugs.
Hypercholesterolaemia was declared for total cholesterol scores above 240mg/dl on 2 occasions at least 3 weeks apart, and high triglyceride levels were declared for levels above 200mg/dl on 2 occasions or for subjects already receiving lipid lowering treatment.
Subjects were considered obese if their body mass index (weight in kilograms divided by height in metres squared) was greater than or equal to 30kg/m2.
Presence of cardiovascular disease was considered for ischaemic heart disease, heart failure, cerebrovascular accident and abdominal aortic aneurysm if events were documented by either hospital admission or by a specialist study. There was no active search for subclinical cardiovascular disease (lacunar stroke, silent ischaemic heart disease, etc.).
To determine ABI, each subject remained in dorsal decubitus during 5 minutes, and the ratio of the systolic pressure at the ankle (posterior tibial artery) to that at the dominant arm (humeral artery) was calculated separately for each of the limbs. We used a pocket non-directional Doppler ultrasound (Mini/Audio/Aqua/Dopplex®) with an 8MHz VP8 transducer. PAD was considered to be present if the index was less than or equal to 0.90.
The abbreviated or modified Modification of Diet in Renal Disease formula (MDRD) was used to estimate GFR, and a rate of less than 60ml/min/1.73m2 was considered indicative of kidney failure.14,15 CKD stages are those described in K/DOQI guidelines, and they included the modifications recently suggested by Levey et al. and Tonelli et al.16,17
Albuminuria was measured as the albumin-creatinine ratio (mg/g) in a sample collected during morning hours. It was classified as optimal for values <10 mg/g; normal-high, 10-29.9mg/g; high, 30-299mg/g; very high, 300-1999mg/g; and nephrotic for values >2000mg/g.
Abbreviated or modified MDRD formula:
GFR (ml/min/1.73 m2) = 186 × [serum creatinine (mg/dl)] –1.154 × (age) –0.203 × (0.742 if female) x (1.212 if black).
Quantitative variables are given as a mean and 95% CI. Qualitative variables are given with their frequency distributions. Association between variables was assessed using χ2 or Fisher’s exact test for qualitative variables, and Student's t-test for quantitative variables. In all cases, values of P<.05 were considered statistically significant. Statistical analysis was performed using the software suite SPSS 15.0.
RESULTS
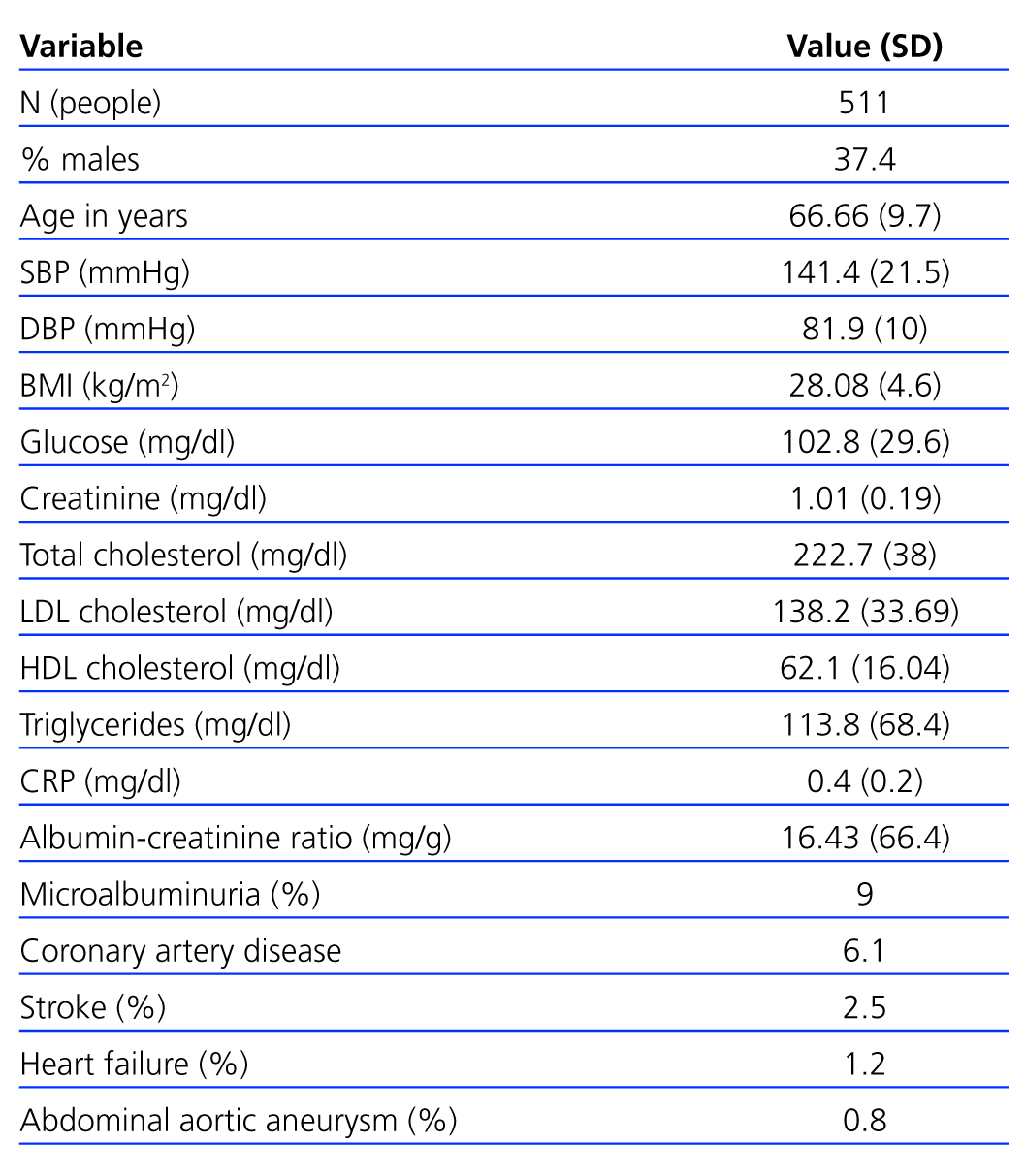
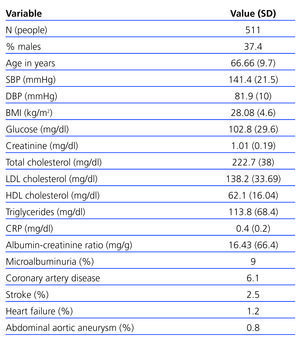
There were 511 subjects at the start of the study (exclusion criteria are listed in Figure 1); 37.4% were male and the mean age was 66.6 years. From their medical histories, we concluded that the prevalence of hypertension was 31.2%, 16.6% were smokers, 7.8% had diabetes and 26.7% had a lipid metabolism disorder. General data are described in Table 1.
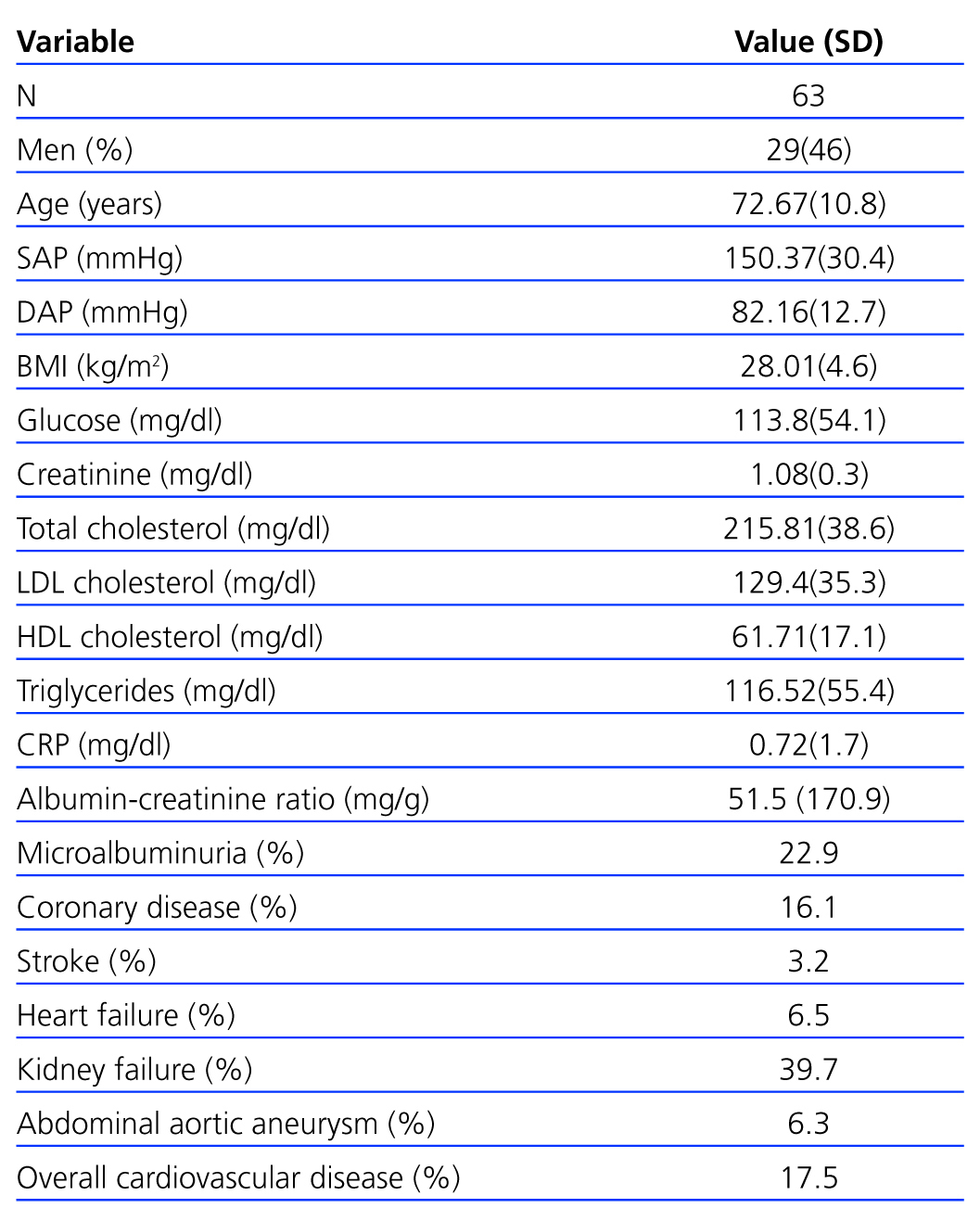
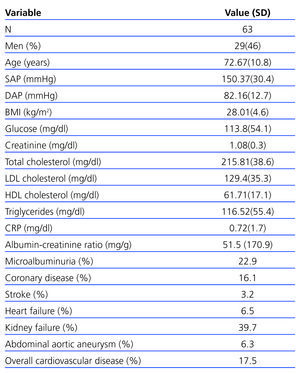
In total, 63 cases (12.4%) in the sample had PAD as defined by an ABI<0.9. The PAD patient group was 46% male with a mean age of 72.6 years. This group presented slightly elevated SBP (150.3mmHg), baseline glycaemia (113.8mg/dl) and microalbuminuria (51.46mg/g). Their cardiovascular risk factors included hypertension (40.3%), dyslipidaemia (24.2%), microalbuminuria (22.9%), smoking (19.4%) and diabetes mellitus (12.9%). The microalbuminuria section also includes 1 patient with urinary albumin values above 300mg/g (Table 2).
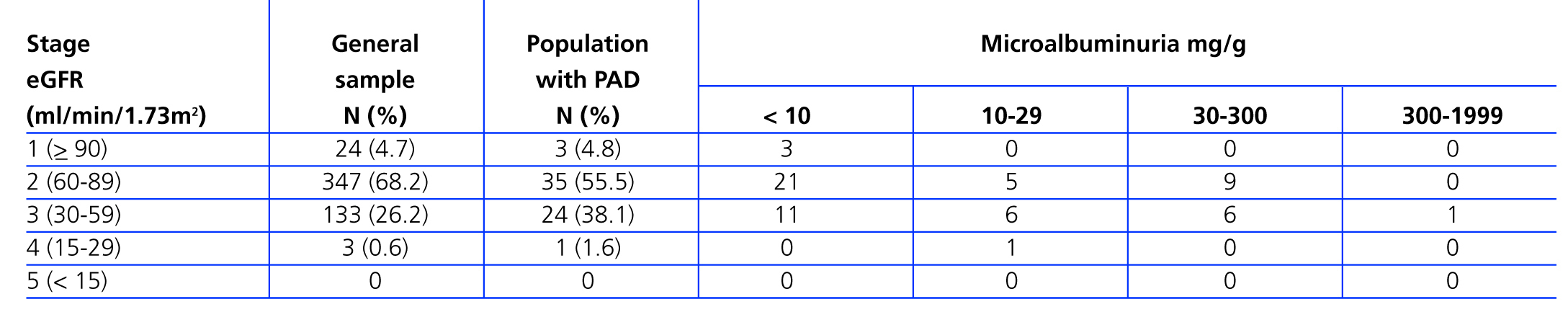
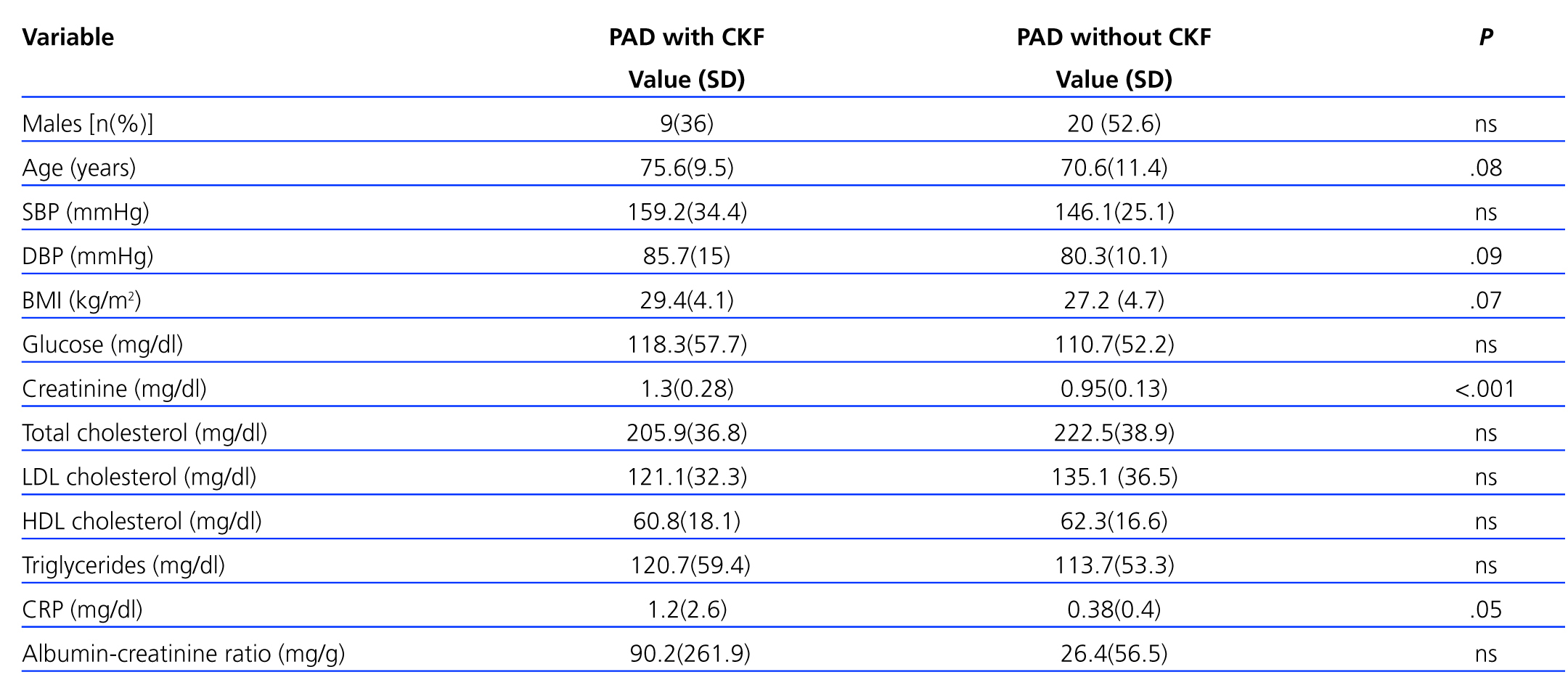
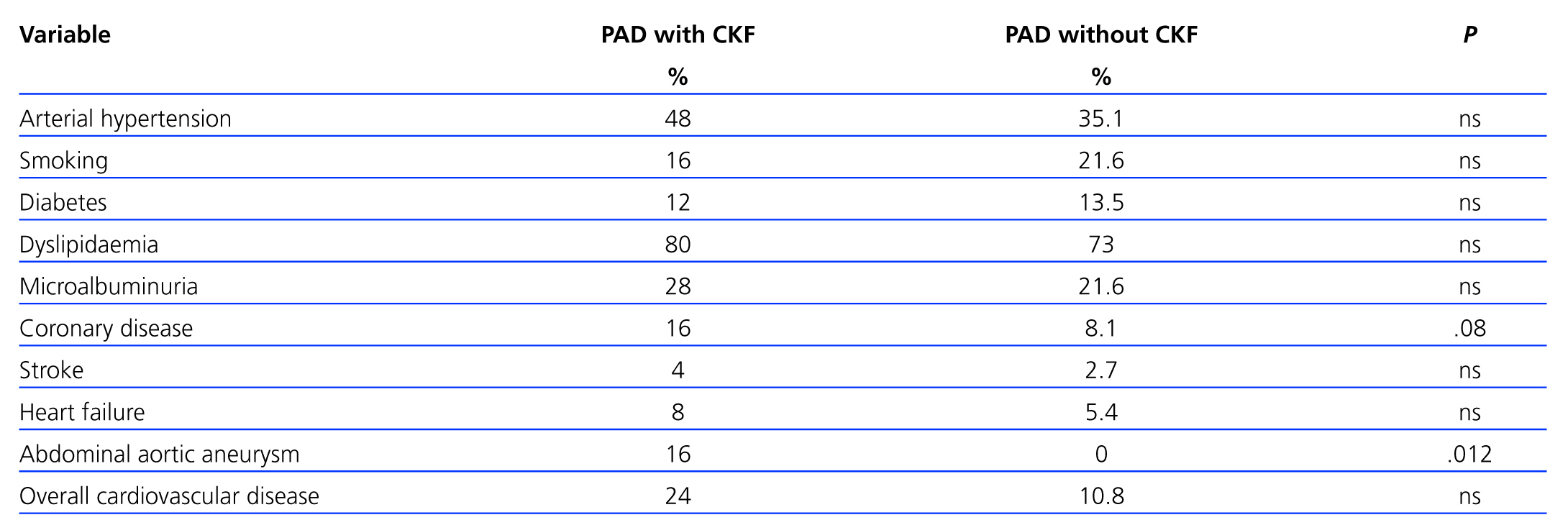
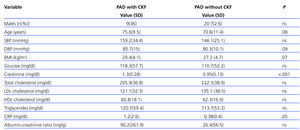
A total of 25 (39.7%) PAD patients had an eGFR<60ml/min/1.73 m2. Table 3 shows distribution by different CKD stages according to eGFR and urinary excretion of albumin for both the general sample and for patients with PAD. Patients with both PAD and kidney failure were older, with higher values for SBP and DBP, baseline glycaemia, C-reactive protein and urinary albumin excretion, although there were no cases in which differences were significant (Table 4). The group with CKD had a higher prevalence of abdominal aortic aneurysm and a tendency towards a higher presence of coronary artery disease, heart failure and global cardiovascular disease, but this was not statistically significant (Table 5).
DISCUSSION
This is the first study in our country to analyse the prevalence of CKD in the population with PAD. In PAD patients aged ≥50 years, the prevalence of CKD (defined by eGFR, MDRD formula, <60ml/min/1.73 m2) was 39.7%.
The prevalence of PAD in our population (12.4%) corresponds to that reported in other population studies that used ABI<0.9 as a criterion. According to the National Health and Nutrition Examination Survey (NHANES 1999-2004), prevalence in the population aged 60 years and older was 12.2%; it was 19.1% in an epidemiological study in the Netherlands, but this was in a general population aged 55 years and older.18,19 However, a recent Spanish study of a population aged 49 years and older in 28 different health centres describes a prevalence of 7.6% for PAD. This is justified when we recall that Spain has low figures for cardiovascular risk according to international risk tables.20 Explaining this difference in prevalence is difficult, because when we compare both populations, we observe that age (72.67 vs 70.7 years), hypertension prevalence (40.3% vs 11.9%), smoking (19.4% vs 11.9%) and hypercholesterolaemia (24.2 vs 10.2%) are much higher in our study. The opposite is true for diabetes (12.9% vs 17%), obesity (1.6 vs 8.2%) and cardiovascular disease, especially myocardial infarction (6.1% vs 29%) and ictus (3.2% vs 24%), which were higher in the other study.
Nearly 40% of the patients with PAD suffered from CKD, which was higher than the 29.7% given by the EROCAP study and the 35% given by the YP Liew study which contained more than 1000 patients.21,22 The association of the two diseases is very common. O’Hare analysed the prevalence of PAD in 2229 subjects aged 40 years and older and established 2 groups according to eGFR (greater or less than 60ml/min/1.73 m2). PAD prevalence in the group with normal kidney function was 3.7%, and reached 24% of subjects with a low eGFR. The OR was 3.0 (95% CI [1.7-5.3], P<.001), and 2.5 (95% CI [1.2-5.1], P=.011), after adjusting for risk factors such as cholesterol, age, ATH and coronary artery disease.10 The same tendency can be found in a sub-analysis of the ARIC study in which the relative risk of developing PAD, comparing patients with eGFR >90ml/min/1.73 m2 and eGFR ≤60ml/min/1.73 m2, was 1.04 (95% CI, 0.91-1.18) and 1.82 (95% CI, 1.13-2.14) respectively.12
Analysing known risk factors that are shared by both diseases shows that the group with PAD and CKD was made up of older subjects, predominantly women (64%), and for all variables except the lipid profile, values were higher than in patients with a normal kidney function. The only parameters for which there were statistically significant differences were plasma creatinine and C-reactive protein. Various studies undertaken in patients with stage III, IV or V kidney failure presented results similar to our own with regard to the prevalence of cardiovascular risk factors.23,24
The differences in serum creatinine are logical due to the correlation between creatinine and kidney function, and this is consistent with findings from other studies in which increased serum creatinine is accompanied by increase in the PAD incidence rate.21,25
Differences in the lipid profile may be explained by the possibility of pharmacological treatment currently being used, given that the prevalence of dyslipidaemia is higher in patients with renal conditions.
Urinary excretion of albumin is higher in the group of subjects with kidney disease [90.16 (261.9) vs 26.36 (56.53)], although the difference is not statistically significant. CKD is not defined solely on the basis of decreased GFR; it also entails the presence of persistent kidney injury during at least 3 months, demonstrated by direct methods (kidney biopsy) or indirectly by the presence of albuminuria or proteinuria or abnormalities in imaging tests or in urine sediment.14 Baber states that the association between decreased GFR and decreased microalbuminuria has to do with a high prevalence of PAD, and this could be useful for identifying a subgroup with vascular disease, although this could not be verified in our study.11
In the present study, the prevalence rates of ischaemic heart disease, ictus and heart failure were all listed separately; all of these entities were far more prevalent in the group with concomitant kidney and vascular disease, although prevalence figures were considerably lower than those published in other studies.26,27
We have not found literature analysing the association of abdominal aortic aneurysms (AAA) in the population with PAD and kidney failure. Our results may be consistent with the findings of Nakamura et al. in a small study that observed poorer kidney function in patients with AAA than in the hypertensive population, and with those of Barba et al. documenting a much higher prevalence of AAA in the PAD population than in the general population. Chue postulates that CKD, in addition to being related to atherosclerosis, might increase arterial rigidity through a series of complex processes related to uraemia. In conjunction with other risk factors, especially hypertension, this could be associated with an increased risk of aneurysm. However, the incidence and prevalence rates of AAA in CKD patients are unknown.28-31
The presence of PAD is a sign of generalised atherosclerosis, and it is associated with higher cardiovascular morbidity and mortality rates. Several population studies corroborate this finding; some well-designed studies with long follow-up periods show a close relationship between decreased kidney function (identified by serum creatinine levels or eGFR), and total/cardiovascular morbidity and mortality. 26,27 It would therefore seem that this group is a target population for treatment to prevent cardiovascular events, and a detection strategy must therefore be implemented in order to identify the at-risk population.
Unfortunately, the exact mechanisms contributing to the high prevalence of CKD among patients with peripheral arterial disease are still unknown. Classic cardiovascular risk factors, which are very prevalent in CKD patients, may contribute aetiologically to PAD development, but they only explain a small part of this association.32 Atheromatous lesions are not restricted to a single area; rather, they become more widespread and severe, and therefore PAD patients frequently suffer from renal artery stenosis, as described in the prospective study by Marin et al. which monitored 418 patients admitted to hospital with severe PAD. An unsuspected stenosis was discovered in the renal artery in 27% of the cases, together with decreased kidney function and increased prevalence of ischaemic heart disease. Since they share aetiopathogenesis, differing stages of ischaemic nephropathy and nephroangiosclerosis might coexist, which would explain the decrease in GFR.33 The role which alterations in the calcium-phosphorus metabolism may play in causing increased vascular calcification and therefore increased PAD and/or kidney disease, especially in the subgroup of patients with both CKD and PAD, is beyond the scope of this study.
A number of authors cite other, less traditional and less commonly-studied risk factors, such as inflammatory markers, increase in lipoprotein(a), oxidative stress, endothelial dysfunction and hyperhomocysteinaemia as factors that could contribute to atherosclerosis development in these patients.32 We found high lipoprotein(a) and C-reactive protein values in the first group, which supports this theory.
Our study has certain limitations. Firstly, the sample size could be called small; it was appropriate for estimating PAD prevalence, but probably insufficient for finding statistically significant differences between the two groups with regard to the risk factor and cardiovascular disease distributions. Secondly, its objective was to measure CKD prevalence in patients with PAD, and not vice versa (prevalence of PAD in CKD patients). This made it difficult to compare it with major studies that did not have the same design. Limitations include the cross-sectional design; since associations are not prospective, we cannot establish a causal relationship between PAD and kidney failure in either direction. Due to these limitations, and particularly the sample size, we may only venture the hypothesis that CKD in the population with vascular disease poses a larger cardiovascular risk. We may be able to confirm this with follow-up data from the study.
CKD is present in nearly 40% of patients with PAD. The presence of decreased kidney function must be taken into account not only for proper adjustment and use of a number of drugs, but also because the copresence of the two conditions is likely to delimit a patient subgroup with a high risk of cardiovascular events. This group may benefit from early diagnosis and treatment, and studies providing an in-depth analysis of the relationship between both conditions are therefore necessary.
Conflicts of interest
The authors affirm that they have no conflicts of interest related to the content of this article.
Acknowledgments
The study was made possible by a grant from the Spanish Society of Family and Community Medicine (semFYC).
Figure 1. Study population flow
Table 1. General sample data
Table 2. Characteristics of patients with peripheral arterial disease
Table 3. Estimated glomerular filtration rate and urinary albumin excretion by chronic kidney disease stages
Table 4. Comparison of clinical variables among patients with peripheral arterial disease with and without associated kidney failure
Table 5. Prevalence of cardiovascular risk factors in patients with peripheral arterial disease with and without associated chronic kidney disease