Renal transplantation (Tx) represents the treatment of choice for patients with advanced chronic kidney disease (ACKD), but the shortage of available organs for those with a high level of comorbidity can significantly increase mortality in patients who are candidates for Tx. This constitutes a worrying health care problem, given the increase in incident and prevalent patients with ACKD, and is especially concerning amongst those with ACKD that is secondary to conditions with a high level of comorbidity, such as diabetes or arterial hypertension. In addition, this can increase the number of patients on the waiting list (WL) and cause the rapid raising of mortality figures. Therefore, nowadays it is relevant to identify the causes of death and the mortality risk factors in this population, to know the barriers that limit access to Tx and to apply predictive mortality models, with the aim of improving survival rates from these illnesses. In this review on the mortality of the patients on the WL, the following aspects will be addressed: 1) the magnitude of this problem and the importance of certain epidemiological data; 2) the mortality risk factors in these patients and the barriers that exist against access to Tx, which could increase mortality rates amongst this population; 3) evaluation of the risk of death in patients on dialysis from comorbidity; 4) assessment of mortality on the WL, via regression analysis of competitive risks, and the generation of a compound risk model, which includes comorbidity and other uraemic factors.
El trasplante renal (Tx) representa el tratamiento de elección para los pacientes con enfermedad renal crónica avanzada (ERC), pero la escasez de órganos disponibles para aquellos con gran comorbilidad puede incrementar significativamente la mortalidad en enfermos candidatos a Tx. Esto constituye un problema sanitario preocupante, dado el incremento de los pacientes incidentes y prevalentes con ERC, especialmente de aquellos con ERC secundaria a entidades de gran comorbilidad como la diabetes y la hipertensión arterial. Asimismo, este hecho puede incrementar el número de pacientes en lista de espera (LE) y disparar sus cifras de mortalidad. Por tanto, actualmente resulta pertinente identificar las causas de muerte y los factores de riesgo de mortalidad en esta población, conocer las barreras que limitan el acceso al Tx y aplicar modelos predictivos de mortalidad en aras de mejorar los resultados de estos enfermos en términos de supervivencia. En esta revisión sobre la mortalidad de los pacientes en LE se abordarán los siguientes aspectos: 1) la magnitud de este problema y la importancia de algunos datos epidemiológicos; 2) los factores de riesgo de mortalidad en estos enfermos y las barreras que existen para el acceso al Tx que pudieran incrementar la mortalidad en esta población; 3) evaluación del riesgo de muerte de los pacientes en diálisis a partir de la comorbilidad; y 4) valoración de la mortalidad en LE mediante análisis de regresión de riesgos competitivos y la generación de un modelo de riesgo compuesto, incluyendo la comorbilidad y otros factores urémicos.
INTRODUCTION
In general, renal transplantation (Tx) is the treatment of choice for advanced chronic kidney disease (CKD) patients, but the scarcity of available organs for a population with high comorbidity, particularly cardiovascular disease and comorbidity related to the uraemic state, may significantly increase mortality in patients who are candidates for a Tx1,2. Indeed, observational studies in a large number of patients have demonstrated a significant increase in mortality of patients on the waiting list (WL) for Tx with respect to those who received a renal graft3. This is currently a concerning health issue, given the gradual increase in incident and prevalent CKD patients eligible for renal replacement therapy, compared with a fixed level of Tx activity in recent years, as has been observed in recent data provided by the American registry4. As a result, the number of patients on the WL has remained stable in the last decade, experiencing a slight increase over the last two or three years, despite a determined effort to increase the activity of Tx5. Specifically, according to data of the National Transplantation Organisation, only 60% of prevalent Spanish patients on the WL had received a Tx as of December 20126. We must add to this the significant increase in the proportion of incident patients with advanced CKD secondary to conditions with high comorbidity such as diabetes and arterial hypertension, which could trigger the mortality rates in the WL4,5. In this review, we will address some of the evidence on mortality in patients on the WL, studying: 1) the magnitude of this problem and the importance of some epidemiological data; 2) mortality risk factors in patients on the WL and the barriers that exist to accessing Tx that could indirectly increase mortality in this population; 3) evaluation of the risk of death in dialysis patients from comorbidity; and 4) assessment of mortality in the WL through a regression analysis of competing risks and the creation of a compound risk model that includes comorbidity and other factors related to the uraemic state estimated at the start of dialysis.
MAGNITUDE OF THE PROBLEM AND EPIDEMIOLOGICAL DATA
CKD patients have significantly higher mortality than the general population and than patients with a functioning renal graft, and this effect is accentuated in individuals over 65 years of age7,8. Focussing on patients who are candidates for Tx, longitudinal studies have demonstrated that overall mortality in patients on the WL is significantly higher than that of patients with transplants, regardless of the type of Tx, although these differences are observed beyond the third month after Tx9. A similar situation is observed in older patients (>65 years of age), regardless of the cardiovascular risk and the type of Tx (standard or with expanded criteria)10. The annual mortality rate varies between 5% and 10% but increases greatly in the older population. Specifically, almost 50% of patients older than 60 years of age who are candidates for Tx in the United States die while on the WL before receiving a renal graft11. A similar overall mortality rate (≈10%) is constantly observed in Spanish dialysis patients, in which a non-negligible proportion dies while on the WL6. And, as expected, the most common cause of death in these patients during the first year of renal replacement therapy is cardiovascular disease, followed by complications from infection. Specifically, ischaemic heart disease, ventricular dysfunction and stroke, followed by infectious comorbidity are the most prevalent causes of mortality in dialysis patients with respect to individuals without CKD5,12.
MORTALITY RISK FACTORS AND BARRIERS TO RENAL TRANSPLANTATION ACCESS
There are few clinical conditions that group together as many risk factors as uraemia. Although risk factors inherent to the uraemic state and other emerging factors not directly related to uraemia have been identified, such as age and traditional cardiovascular risk factors, such as diabetes or arterial hypertension, they play a decisive role in the increased risk of death in the framework of an older population with higher comorbidity12. In this regard, data from the American registry show that age and diabetes significantly increase the risk of death in dialysis patients, particularly in those returning from transplantation, in which cardiovascular complications and infections are the main causes of death13. In the European population, dialysis patients >60 years of age display significantly lower survival than younger patients14. Indeed, the risk of death in this older population significantly increases as their time on renal replacement therapy increases15. In the American population, some socio-demographic factors (age, race or employment status), being a smoker, diabetes and cardiovascular comorbidity (ischaemic heart disease and cerebrovascular disease and peripheral vascular disease) are independent mortality risk factors in dialysis patients, adjusting for other confounding clinical variables16. Likewise, these same factors in addition to psychiatric disorders and a history of neoplasia are also associated with a higher risk of mortality in the European dialysis population14. In line with this, an observational study on a large number of patients showed that peripheral vascular disease (PVD) was very prevalent in the dialysis population, particularly in diabetic patients (48%) and conferred three times more risk of mortality than in those who did not have ischaemic disorders in their lower limbs17. This was subsequently confirmed in the HEMO study, in which patients with more severe PVD had a greater rate of overall and cardiovascular mortality18. To further complicate this situation, comorbidity can progress over time in haemodialysis patients, including those on the WL. In this regard, an observational study carried out in haemodialysis patients in Taiwan demonstrated that prevalence of comorbidities included in the Charlson comorbidity index (CCI) increased alarmingly during the first three years of dialysis19. Therefore, the higher the comorbidity, the lower the chances are of being assessed and included on the WL, as has been demonstrated in observational studies through sensitivity and specificity analyses20.
Furthermore, significant weight loss (>5kg) or a body mass index (BMI) of <23kg/m2 in patients on the WL have been associated with a significant increase in mortality (20%), possibly due to a hypercatabolic inflammatory state21. At the same time, a high BMI (>25kg/m2 in females or >35kg/m2 in males) in patients on the WL is a barrier to accessing Tx22. Most clinical practice guidelines recommend that obese patients achieve a BMI of around 30kg/m2 before Tx23,24. In general, this leads to temporary exclusion from the WL and a longer time on dialysis for obese patients, resulting in a potential increase in morbidity and mortality in this population25.
Similarly, in Tx candidates there may be a high prevalence of suboptimal health indicators, recently established by the American health authorities (County Health Rankings Web Site. 2011. http://www.countyhealthrankings.org), such as low weight at birth, heavy smoking, obesity, mental deterioration, physical inactivity or low income, and as such, the sum of these inadequate health indicators could increase the risk of mortality on the WL or at least determine the temporary or definitive exclusion from the WL26,27.
Other factors inherent in the uraemic process may also involve a greater risk of mortality in dialysis patients, including those on the WL. A recent observational cohort study demonstrated that haemodialysis increased the risk of death with respect to peritoneal dialysis, adjusting for a propensity analysis28. In line with this, a subsequent longitudinal study of the same group showed that the excess of mortality in haemodialysis patients was mainly observed in patients with a central venous catheter (CVC) at the start of renal replacement therapy, again adjusting for confounding factors such as age or comorbidity29. Indeed, Lorenzo et al. demonstrated in an elegant multi-centre observational study that a CVC and non-scheduled start of dialysis were associated with a significant increase in mortality in incident haemodialysis patients and that these conditions had synergic effects on decreased survival30. A subsequent study on thousands of incident haemodialysis patients confirmed that the presence of a CVC was an important risk factor for mortality of any cause, including cardiovascular mortality, in the American population31.
Blood transfusions in patients on the WL may result in a greater risk of sensitivisation (20%) when the patient receives a graft, through the creation of anti-HLA antibodies32. This may obviously extend the time on the WL and favour future comorbidities. Likewise, the clinical management of hepatotropic viral infections or infection with the human immunodeficiency virus in dialysis patients generally require a complex diagnostic and therapeutic approach that may be a temporary barrier to quick access to Tx23,24,33,34.
Other socio-demographic factors may represent barriers for access to Tx, which increases times on the WL and favours the potential onset of other non-desirable comorbidities during time on dialysis. Specifically, several observational studies have shown that females35, patients who live in rural areas36 or who live far from the transplantation centre37, unmarried patients38 or non-Caucasian individuals or economically disadvantaged individuals and those without adequate health coverage are less likely to be included on the WL39,40, which undoubtedly demonstrates the inequality of some healthcare policies. Lastly, the hospital itself may be an obstacle for access to Tx. Patients of hospitals with high Tx activity have a greater probability of being included on the WL than those without this healthcare activity41. As a result, survival expectations for patients on the WL in hospitals with high Tx activity are significantly higher than those of patients of other hospitals with a low Tx rate42. Furthermore, it has recently been observed that private dialysis units could delay inclusion on the WL and access to Tx compared to dialysis units in public or not-for-profit healthcare centres43.
In any case, some of the criteria of health professionals for choosing a candidate for the WL are imprecise (life expectancy, adherence to treatment, social factors, etc.) and may cause a major imbalance in access to Tx and distrust in the general population. As such, clinical practice guidelines have been developed that propose recommendations for the assessment and acceptance of patients who are candidates for Tx23,24,34. Although the guidelines have limitations, they may contribute to balancing individual equity with interest and effectiveness for the general community.
However, there may be a high overlapping in the risk profile of dialysis patients who are on the Tx waiting list and those who are not. A longitudinal study carried out on an American population showed that a third (34%) of patients on the transplant WL during the first year who apparently had lower comorbidity had a life expectancy lower than five years (mean survival time 3.8 years); on the contrary, 33% of patients who are not on the list for Tx and who presumably had higher comorbidity had a projected life expectancy >5 years (median survival 6.6 years). In other words, many dialysis patients not included on the WL showed survival higher than that of some patients who were included early, during the first year of renal replacement therapy16.
ASSESSMENT OF THE RISK OF DEATH IN DIALYSIS
With these premises, predicting mortality and comorbidity may be crucial in patients on the WL for making the best decisions. In other words, in these patients we should accurately assess the risk of death in order to be able to prioritise or correctly assign a Tx.
Individually, any predictive variable or subordinate survival measure may determine the prognosis of a clinical condition, but the prediction capacity may be optimised using many predictors or subordinate measures grouped into comorbidity indexes (CI).
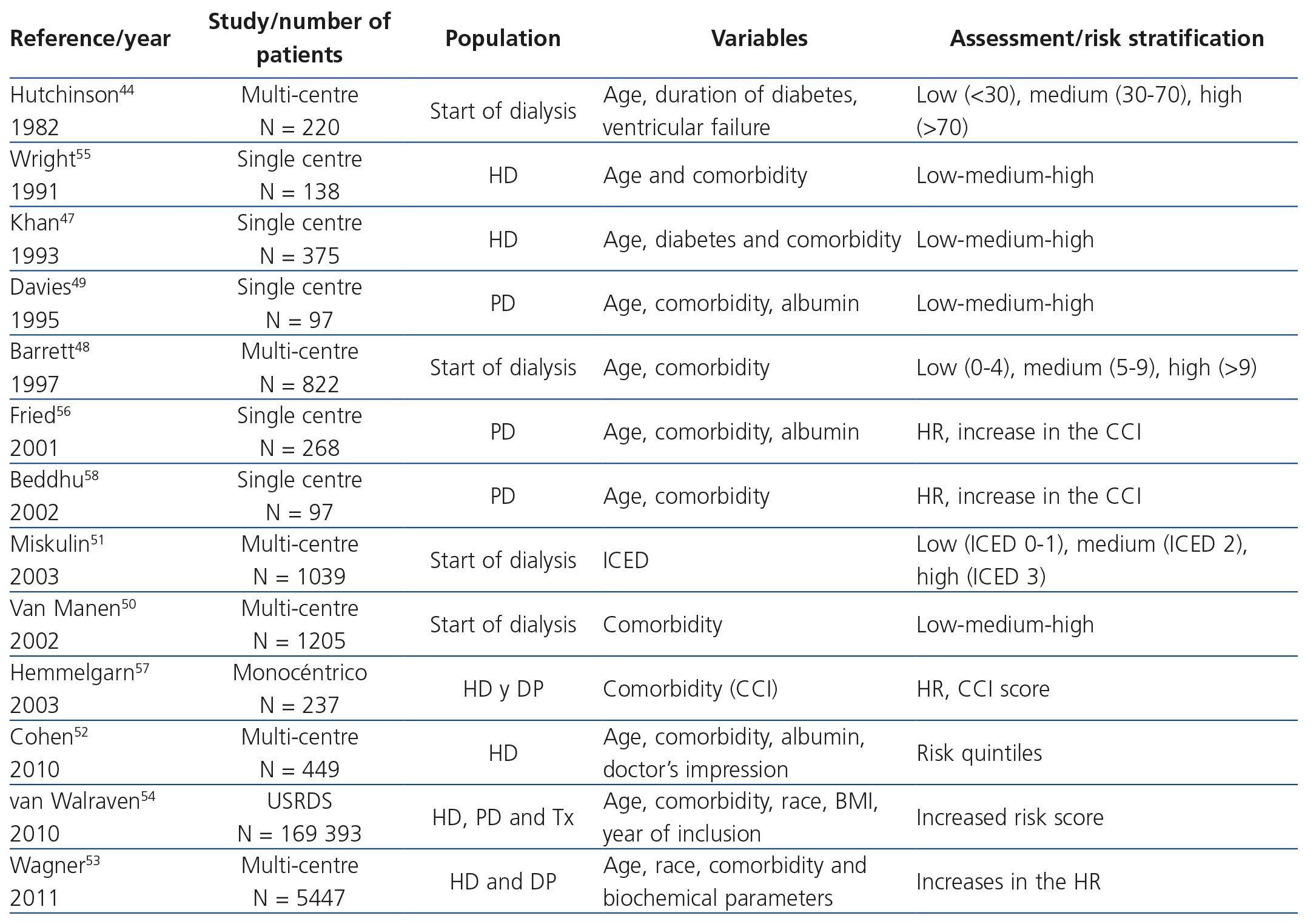
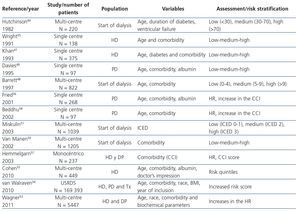
One of the first approaches in the search for a systematic CI in the uraemic population dates from 1982, when an elegant study by Hutchison et al., using age, presence of diabetes and left ventricular failure as comorbid conditions, prepared an CI using a mathematic model44. Since then, various CI have been applied in the uraemic population with satisfactory results in terms of their mortality prediction capacity (Table 1). Some of them are based on the general population, such as the index of coexisting diseases45 or the CCI46, and others are newly created from comorbidities and other variables related to the uraemic state47-58. All predict the risk of death in dialysis with a high concordance rate and they have been validated internally or externally, in different populations, but most lack a mortality analysis exclusively carried out in patients on the WL. By way of example, in a Canadian study in which 882 patients were recruited, a mortality risk scoring system was created using a multivariate logistic regression analysis from age and other comorbid situations such as ventricular dysfunction, malnutrition, PVD and neoplasia. In this system, as the score increased, the probability of death increased48. However, the sample size and the lack of randomisation of the sample may have created an overestimation bias in this index, which limits its predictive capacity. More recently, mortality prediction models have been created, after the randomisation of the sample in two subpopulations, with the variables which in the model population were associated with a higher risk of death (age, subjective assessment by the nephrologist, dementia, PVD, albumin level and comorbidity)52,53. However, this was in populations of haemodialysis patients that mainly included non-Tx candidates. With this idea, a Canadian study developed a model for predicting mortality in three patient groups (patients on the WL, patients with a Tx from a deceased donor and patients with a Tx from a living donor) from a large number of patients included in the American database (n=169,393). After the sample randomisation in two subpopulations, a score was obtained for each variable from a Cox multivariate analysis, obtaining a total risk score for each patient, which predicted the probability of death after five years54. In other words, as the risk score increases, the risk of death in these patients increases exponentially, including patients on the WL. However, other risk factors inherent to uraemia were not implemented, such as the type of dialysis, previous Tx or having a CVC at the start of dialysis, amongst others.
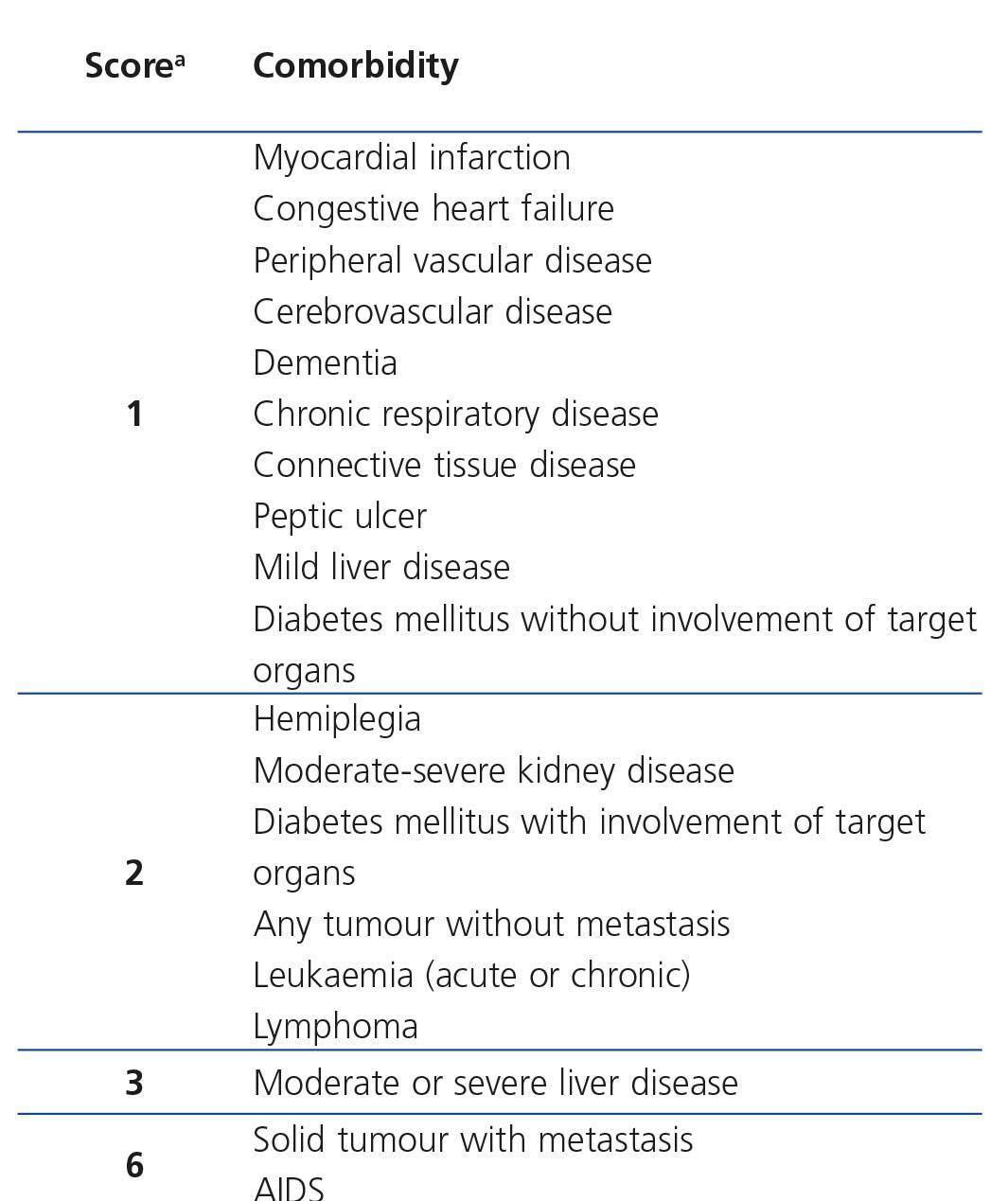
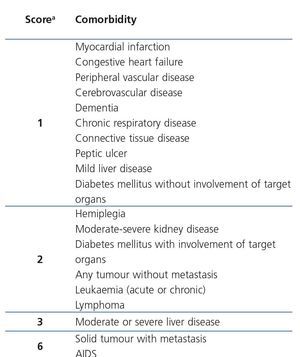
The CCI has been validated in the general population and in uraemic patients as a useful tool for predicting the risk of mortality46, particularly when they are compared with other CI validated in kidney patients, such as the Khan index, the Davies index or the van Manen index50,59. The CCI assigns a certain score to each comorbidity, including age (from 40 years of age, one point is assigned for each decade). Thus, when there is higher comorbidity, the score increases and the risk of mortality increases (Table 2). This index may be applied universally to predict mortality in patients on the WL, but it does not incorporate other mortality risk factors inherent to the uraemic state that could have a negative impact on survival, such as the presence of a CVC, early or late referral to the nephrologist, unemployment, time on dialysis or previous transplantation.
However, there is limited information on the use of the CI and of other comorbid factors inherent in uraemia in clinical practice for predicting mortality in patients who are candidates for Tx. It was therefore relevant to know the joint impact of comorbidity included in the CCI and other comorbid factors on mortality in uraemic patients on the WL.
COMPOUND RISK MODEL FOR PREDICTING MORTALITY USING COMPETING RISKS
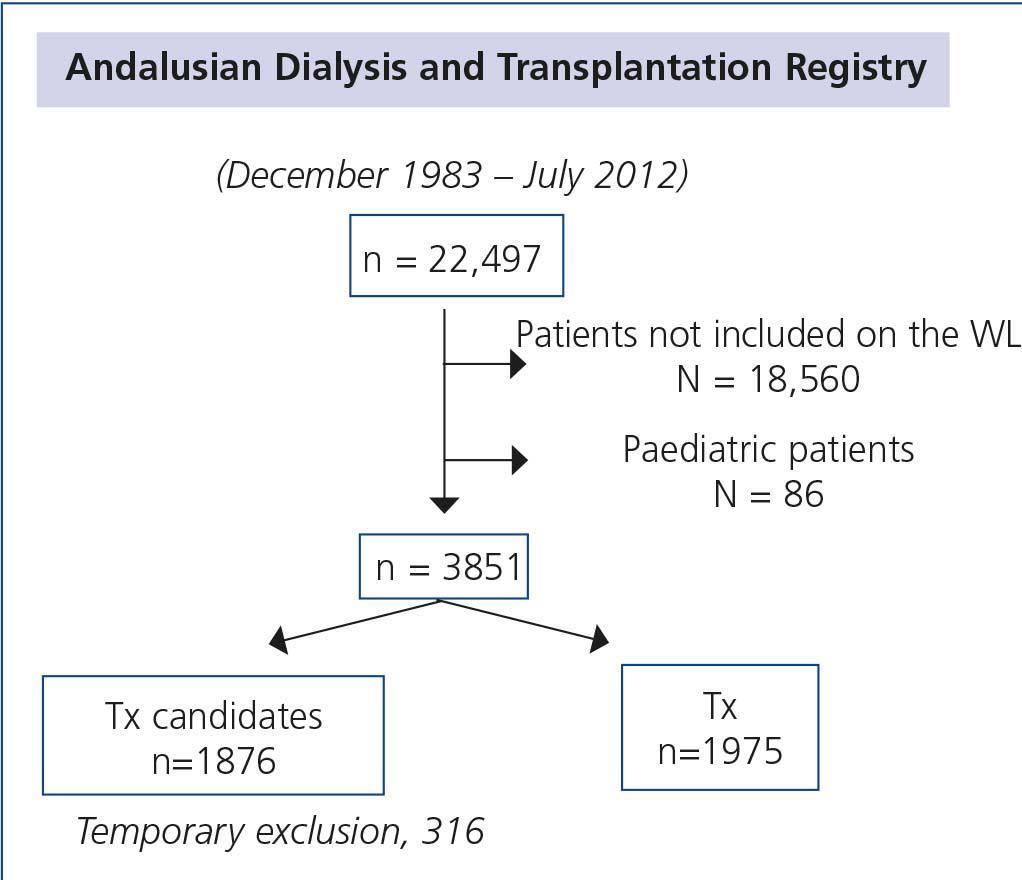
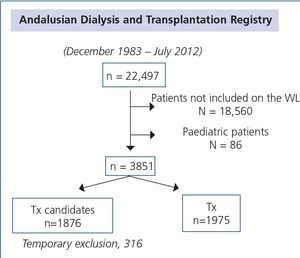
With these premises, we set out to identify mortality risk factors through the CI score and other comorbid factors inherent to uraemia in 22,497 patients on renal replacement therapy who were included in the Andalusian Registry of Kidney Patients between December 1983 and July 2012. After the exclusion of 18,560 patients who were never candidates for a Tx and paediatric patients (n=86), we analysed a final sample of 3851 patients (20% of the total) who were initially assessed as Tx candidates (Figure 1). Of these, 1876 remained on the WL at the time of their last follow-up and 1975 had received a functioning Tx and never returned to dialysis, being censored in the initial analysis.
The variables used in survival analyses included clinical and demographic data, the CCI score and other comorbid factors inherent to the uraemic state not included in the CCI such as the presence of a CVC, previous transplantation, employment status, late referral to the nephrologist, time on dialysis, the method of dialysis, the year of inclusion and all causes of death. Survival on the WL was assessed using a conventional Cox proportional regression model and a regression analysis of competing risks60. A competing event is that which modifies or prevents the event of interest (mortality) from occurring. In the estimation of survival of patients on the WL, receiving a functioning Tx is a competing event. In these cases, the probability of the event of interest occurring could be overestimated, particularly in the presence of many competitive risks, as occurs in patients on the WL61. Therefore, the estimation of the survival of patients on the WL was assessed by a competing risk regression model due to the presence of many competing events as in our population (51% received a Tx). An important characteristic of this method is that subjects who experience a competing episode remain in the risk group (instead of being censored), despite the fact that they are no longer at risk of the event of interest62.
Likewise, to avoid a confounding bias due to indication, we developed a propensity analysis through a multivariate logistic regression model, using as the dependent variable the receiving or not of a Tx, and as independent variables all the variables listed in the registry that could predispose the patient to receiving a Tx, including the year of inclusion of the WL. In this way, for each subject, we obtained a score that corresponds to the probability of receiving a Tx based on the demographic and clinical characteristics of the patients. This allows us to balance the clinical characteristics between those who remained on the WL and those who received a Tx at each level of probability63. The final score was divided into propensity quartiles that were introduced as a covariate in the multivariate survival models.
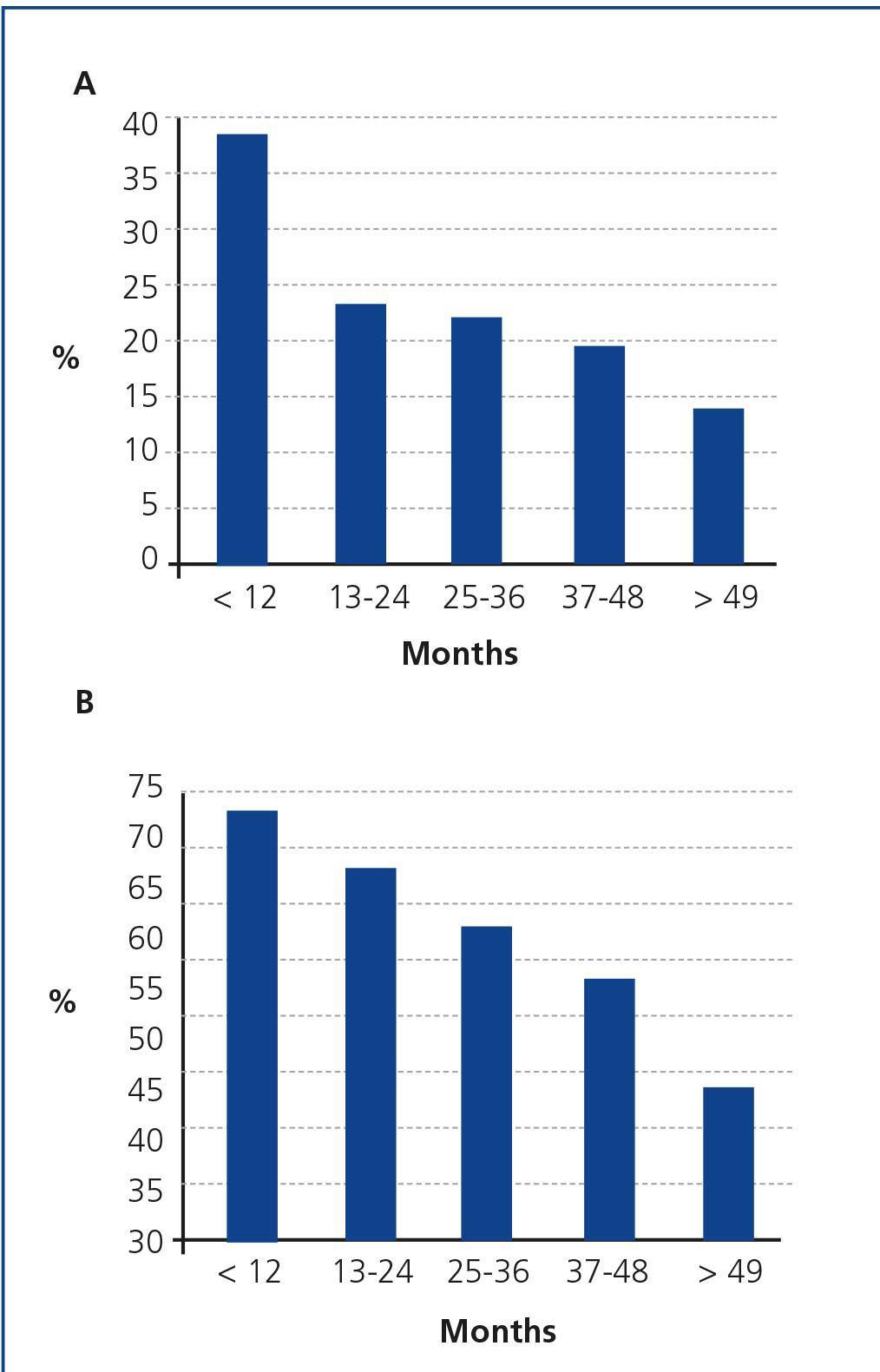
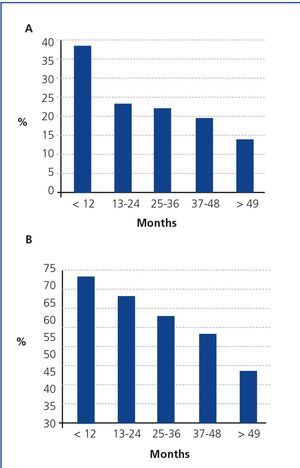
The median follow-up time was 22 months (interquartile range 12-48 months) and a total of 446 patients (24%) died while they were on the WL, with cardiovascular disease being the most common cause (25%) of this mortality. Surprisingly, 62% of exitus occurred in the first two years after inclusion on the WL (Figure 2) and a large proportion of these patients had higher comorbidity (CCI >3). In other words, the patients who remained longer on the WL had lower comorbidity than those who died early. Indeed, overall survival of younger Tx candidates (<50 years) was significantly higher than that of older candidates, despite younger candidates having spent a longer time on the WL. This may have explained why the time on the WL was not associated with mortality in our analysis. In line with our findings, an analysis of Organ Procurement and Transplantation Network data showed that around 50% of Tx candidates die within the first two years of inclusion on the waiting list, in which a high proportion of patients are older than 50 years of age and have higher comorbidity64. The fact that only 7% of dialysis patients older than 65 years of age in the United States receive transplants after three years on renal replacement therapy supports this view10. By contrast, time on the WL has been documented as an important factor associated with mortality in a retrospective study by the Scientific Registry of Transplant Recipients, but time on the WL was estimated indirectly by the proportion of patients who received a Tx during the first three years on the WL and this time was not introduced in the multivariate analyses as an independent variable42.
The regression model of competing risks showed that age >50 years (subhazard ratio [SHR] 1.4, 95% confidence interval [CI] 1.1-1.9), the presence of a CVC at the start of renal replacement therapy (SHR 1.8, 95% CI 1.4-2.2), unemployment (SHR 1.7, 95% CI 1.3-2.2) and a CCI>3 (SHR 2.8, 95% CI 2.1-3.7) were risk factors significantly associated with mortality, adjusting for other confounding variables, including the propensity analysis and the year of inclusion on the WL. In quantitative terms, the risk of mortality increased 52% for each unit the CCI increased when the score of this index was included in the multivariate analysis as a continuous variable. Similar results were observed when patients who were temporarily contraindicated were excluded.
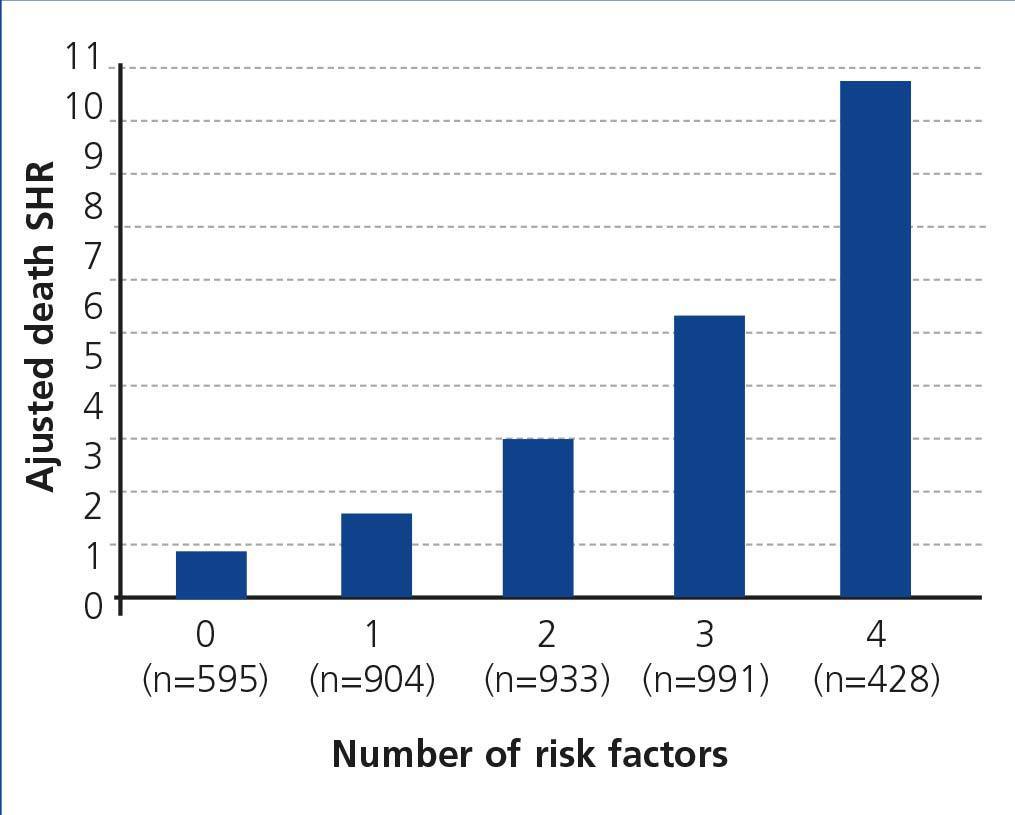
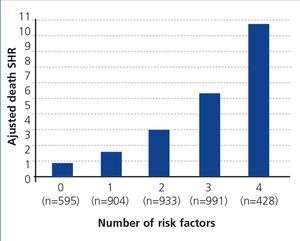
Lastly, a compound risk model was developed using risk factors associated with mortality in the adjusted competing risk regression model. Patients on the WL without risk factors were compared with those with one or more risk factors, again adjusting for confounding variables. The risk of death significantly increased at each risk level (Figure 3). By way of example, the presence of two risk factors increased the risk of death by approximately three times, while the combination of four factors increased the risk of death on the WL by more than ten times. In other words, as the number of risk factors grows, the accumulative incidence of mortality during time on the WL increases.
With the limitations inherent in registries and retrospective studies, in which major risk factors are not recognised, this assessment of mortality in patients on the WL could help prioritise patients at risk of early death on the WL, in order that they may receive a renal graft from a deceased donor of a similar age. This undoubtedly will contribute to improving the results of patients who are candidates for a Tx in terms of long-term survival.
Acknowledgements
This study has been partially funded by the Spanish Ministry of Science and Innovation, Instituto de Salud Carlos III (FIS PI10/01020), RETIC RedInRen RD12/0021/0015 and by the Ministry of Health of the Government of Andalusia (PI-0590/2012). The authors would like to thank all members of the Regional Transplant Coordination of Andalusia and all participants in each centre for their support in compiling information. Likewise, they wish to thank the Nephrology department of the Hospital Universitario Carlos Haya of Malaga for its assistance and Carmen Vozmediano and Armando Torres for their comments in the preparation of the manuscript. Lastly, we would also like to thank Víctor Abraira and Javier Zamora for their statistical advice in the data management.
Conflicts of interest
The authors declare that they have no conflicts of interest related to the contents of this article.
KEY CONCEPTS
Table 1. Different comorbidity rates for predicting mortality in kidney patients
Table 2. Charlson comorbidity index score
Figure 1. Flow diagram of assessed patients of the Andalusian Dialysis and Transplantation Registry for the analysis of mortality on the waiting list
Figure 2.
Figure 3.