The efficacy of phosphate binders is difficult to be estimated clinically. This study analyzes the changes in serum phosphate and urinary phosphate excretion after the prescription of phosphate binders (PB) in patients with chronic kidney disease stage 4–5 pre-dialysis, and the usefulness of the ratio between total urinary phosphate and protein catabolic rate (Pu/PCR) for estimating the efficacy of PB.
MethodsThis retrospective observational cohort study included adult chronic kidney disease patients. Biochemical parameters were determined baseline and after 45–60 days on a low phosphate diet plus PB (“binder” subgroup=260 patients) or only with dietary advice (“control” subgroup=79 patients).
ResultsPhosphate load (total urinary excretion) per unit of renal function (Pu/GFR) was the best parameter correlated with serum phosphate levels (R2=0.61). Mean±SD level of Pu/PCR was 8.2±2.3mg of urinary phosphate per each g of estimated protein intake. After treatment with PB, serum phosphate levels decreased by 11%, urinary phosphate 22%, protein catabolic rate 7%, and Pu/PCR 15%. In the control subgroup, Pu/PCR increased by 20%. Urinary phosphate and urea nitrogen excretion correlated strongly, both baseline and after PB or dietary advice.
ConclusionsThe proposed parameter Pu/PCR may reflect the rate of intestinal phosphate absorption, and therefore, its variations after PB prescription may be a useful tool for estimating the pharmacological efficacy of these drugs.
Es difícil estimar clínicamente la eficacia de los captores de fósforo (CP). Este estudio analiza los cambios que se producen en la fosfatemia y excreción urinaria de fósforo tras la administración de CP a pacientes con enfermedad renal crónica, y la utilidad de la relación entre la excreción urinaria de fósforo y la tasa de catabolismo proteico (Po/TCP) en la estimación de la eficacia de estos fármacos.
MétodosEstudio retrospectivo de observación en una cohorte de pacientes adultos con enfermedad renal crónica en estadios 4-5. Se compararon parámetros bioquímicos basales y 45-60 días después de un tratamiento con dieta baja en fósforo más CP (subgrupo «captor»=260 pacientes) o solo con los consejos dietéticos (subgrupo «control»=79 pacientes).
ResultadosLa carga de fósforo (excreción urinaria total) por unidad de función renal (Po/GFR) fue el parámetro mejor relacionado con la fosfatemia (R2=0,61). La cifra media de Po/TCP fue de 8,2±2,3mg de fósforo por gramo de proteína. Tras la administración de CP, la fosfatemia descendió un 11%, la fosfaturia un 22%, la tasa de catabolismo proteico un 7% y la Po/TCP un 15%. En el subgrupo control la Po/TCP se incrementó un 20%. La excreción urinaria de fósforo y de nitrógeno ureico se correlacionaron fuertemente de forma lineal antes y después del tratamiento con CP o tras los consejos dietéticos en el subgrupo control.
ConclusionesLa Po/TCP es un parámetro que podría reflejar la absorción intestinal de fósforo y, por tanto, sus variaciones tras la administración de CP podrían servir para estimar la eficacia de estos fármacos.
The alteration of phosphate metabolism associated with chronic kidney disease (CKD) is an important pathogenic factor involved in the development of metabolic bone disease, cardiovascular complications and the progression of renal disease.1–4
A decrease in the tubular reabsorption of phosphates (increase in fractional excretion) is the only compensatory mechanism to maintain the phosphate balance in the organism when the glomerular filtration rate decreases.5 However, this compensatory mechanism is limited in the most advanced stages of CKD. Reducing the dietary load and preventing intestinal phosphate absorption by the use of binders are the only treatment measures currently available to maintain the phosphate balance in advanced CKD.6
Clinically, the efficacy of phosphate binders is difficult to assess. The decrease in serum phosphate after administration of the binder could be a simple method of assess phosphate binders efficacy. However its use proves inaccurate when taking into account the many other factors that condition phosphataemia (e.g. intake, renal excretion, bone reabsorption, extraosseous exchange, treatment compliance).
If it is assumed a neutral balance in the phosphate exchanges between fluid compartments and the bone pool in patients with CKD, urinary excretion can reliably reflect the amount of phosphate ingested and absorbed in the intestine.7–11 Thus, changes in urinary phosphate after administering a binder could be used to assess its efficacy. However, this method does not take into account the possible variations in the dietary intake of phosphate. If a binder that, by causing digestive irritation, led to a decrease in the total intake of dietary phosphate due to anorexia or vomiting, could be wrongly classed as very effective.
On the other hand, estimating phosphate intake through a dietary survey may also be inaccurate, due to the dissociation between the quantity of phosphate in a food and its availability for intestinal absorption.12,13 The measurement of faecal phosphate in order to obtain an intestinal absorption balance is an unquestionably but cumbersome method in clinical practice.
A strong positive linear correlation has been observed between urinary excretions of urea nitrogen and phosphate, and a close link between the intake of proteins and the intake-intestinal absorption of phosphate has been demonstrated.11,14,15 Thus, the quotient between the urinary excretion of phosphate and the protein catabolic rate (calculated by total urea nitrogen excretion) could be a useful parameter for estimating the intestinal absorption of phosphate and the efficacy of a binder, assuming that the pharmacological effect of the binder only influences the absorption of phosphate and not of nitrogen.
The objectives of this study were: to analyze the changes that occur in phosphataemia and in urinary phosphate excretion after administering binders to patients with advanced CKD, and to determine the usefulness of different parameters in estimating the efficacy of these drugs.
Materials and methodsA retrospective observational cohort study was performed on adult incident patients in an advanced CKD outpatient clinic between February 2008 and October 2015. The selection criteria were: patients over 18 years of age, an estimated glomerular filtration rate (eGFR) of <30mL/min/1.73m2, no previous treatment with phosphate binders and the ability to properly collect urine over a 24h period.
Patients with clinical instability (heart failure or failure of other organs, cancer patients or patients in a hypercatabolic state), with recent acute kidney failure, in active treatment for glomerular disease or vasculitis, being treated with corticosteroids or with drugs with a potential influence on bone remodelling (vitamin D analogues, corticosteroids, bisphosphonates, denosumab, etc.), were excluded.
Included in the clinical routine of this advanced CKD outpatient clinic, was the determination of the conventional biochemical parameters in blood – urea, creatinine, albumin, total calcium, ionic calcium, phosphate, magnesium, bicarbonate, and PTH – concentrations of creatinine, urea, calcium and phosphate in urine collected over the 24h period were also determined in all the patients prior to drawing blood samples.
The biochemical determinations were performed using conventional laboratory methods (Advia Chemistry, Siemens Healthcare Diagnostics). PTH (molecule 7–84) levels were determined by an automated chemiluminescence immunoassay (DiaSorin, Italy).
With this biochemical data, the following parameters were calculated: (1) Urea and creatinine clearance, and approximation of glomerular filtration rate (mGFR: half of the sum of urea and creatinine clearances). (2) Total urinary urea nitrogen excretion. (3) Protein catabolic rate using the Maroni et al. formula.16 (4) Total urinary excretion of calcium and phosphate (mg/24h). (5) Fractional excretion of phosphate (in %). (6) Phosphate load relative to kidney function: total urinary phosphate excretion/measured glomerular filtration rate (mg of phosphate per mL/min/1.73m2). (7) Intestinal phosphate absorption estimation parameter: total urinary phosphate excretion/protein catabolic rate (mg of phosphate per gram of estimated protein intake).
The total group of patients studied was divided into 2 subgroups: “case” patients are those treated with phosphate binders after the initial visit, and the “control” patients were those that did not receive binders due to clinical criteria, or those that admit not to be taking the prescribed binder due to intolerance or for other reasons.
Study design and statistical analysisIn this observational study, the baseline biochemical parameters were compared to the biochemical parameters 45–60 days after treatment. In the “case” subgroup, patients were treated with conventional doses of phosphate binders (see “Results” section for specific information on the binders used), and also received dietary advice for reducing phosphate intake. In the “control” group, only dietary advice was given.
To compare continuous variables, a Student's t-test was used (paired data or non-paired data). The chi-squared test was used to compare categorical variables between the subgroups.
To determine possible association between continuous variables and to represent it graphically, a simple linear regression analysis was used. The coefficient of determination (R2) was calculated to quantify the degree of fitness of the model.
The data is presented as the mean±standard deviation. A p<0.05 indicated statistical significance. Statistical analysis and graphs were produced using SPSS version 21.0 (IBM Corp., Armonk, USA).
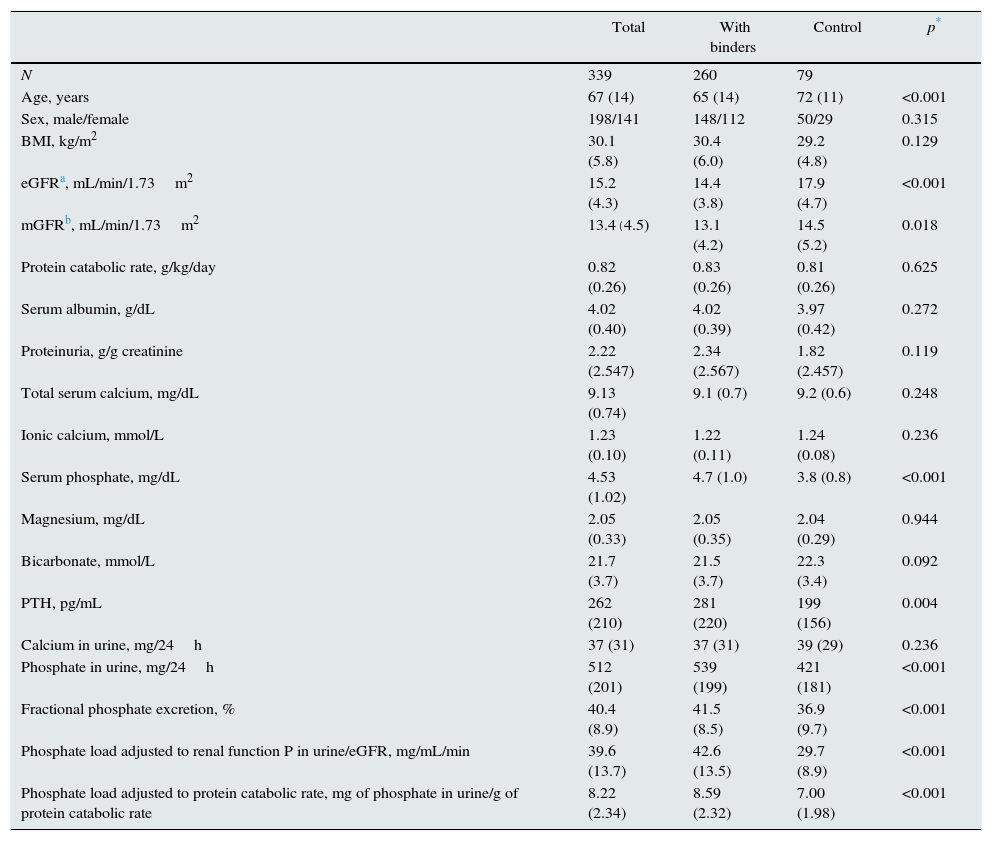
ResultsA total of 339 patients were included; demographic, clinical, and baseline biochemical characteristics are shown in Table 1.
Demographic, clinical and biochemical baseline (before treatment) characteristics of the study group and differences according to subgroups treated with or without binders.
| Total | With binders | Control | p* | |
|---|---|---|---|---|
| N | 339 | 260 | 79 | |
| Age, years | 67 (14) | 65 (14) | 72 (11) | <0.001 |
| Sex, male/female | 198/141 | 148/112 | 50/29 | 0.315 |
| BMI, kg/m2 | 30.1 (5.8) | 30.4 (6.0) | 29.2 (4.8) | 0.129 |
| eGFRa, mL/min/1.73m2 | 15.2 (4.3) | 14.4 (3.8) | 17.9 (4.7) | <0.001 |
| mGFRb, mL/min/1.73m2 | 13.4(4.5) | 13.1 (4.2) | 14.5 (5.2) | 0.018 |
| Protein catabolic rate, g/kg/day | 0.82 (0.26) | 0.83 (0.26) | 0.81 (0.26) | 0.625 |
| Serum albumin, g/dL | 4.02 (0.40) | 4.02 (0.39) | 3.97 (0.42) | 0.272 |
| Proteinuria, g/g creatinine | 2.22 (2.547) | 2.34 (2.567) | 1.82 (2.457) | 0.119 |
| Total serum calcium, mg/dL | 9.13 (0.74) | 9.1 (0.7) | 9.2 (0.6) | 0.248 |
| Ionic calcium, mmol/L | 1.23 (0.10) | 1.22 (0.11) | 1.24 (0.08) | 0.236 |
| Serum phosphate, mg/dL | 4.53 (1.02) | 4.7 (1.0) | 3.8 (0.8) | <0.001 |
| Magnesium, mg/dL | 2.05 (0.33) | 2.05 (0.35) | 2.04 (0.29) | 0.944 |
| Bicarbonate, mmol/L | 21.7 (3.7) | 21.5 (3.7) | 22.3 (3.4) | 0.092 |
| PTH, pg/mL | 262 (210) | 281 (220) | 199 (156) | 0.004 |
| Calcium in urine, mg/24h | 37 (31) | 37 (31) | 39 (29) | 0.236 |
| Phosphate in urine, mg/24h | 512 (201) | 539 (199) | 421 (181) | <0.001 |
| Fractional phosphate excretion, % | 40.4 (8.9) | 41.5 (8.5) | 36.9 (9.7) | <0.001 |
| Phosphate load adjusted to renal function P in urine/eGFR, mg/mL/min | 39.6 (13.7) | 42.6 (13.5) | 29.7 (8.9) | <0.001 |
| Phosphate load adjusted to protein catabolic rate, mg of phosphate in urine/g of protein catabolic rate | 8.22 (2.34) | 8.59 (2.32) | 7.00 (1.98) | <0.001 |
The total number of patients treated with binders was 260. These patients were prescribed conventional doses of calcium carbonate (1–1.5g/day), calcium acetate (1–1.5g/day), calcium acetate with magnesium carbonate (1305mg/705mg/day), lanthanum carbonate (1000–2250mg/day), sevelamer (2400–4800mg/day), or aluminium hydroxide (466–699mg/day alone or together with other binders). In order to estimate the total binding power of the prescription, the relative phosphate-binding coefficient described by Daugirdas et al.17 was used. Briefly, each mg of a calcium salt (carbonate or acetate) was multiplied×1; each mg of lanthanum carbonate×2; each mg of sevelamer×0.75; each mg of heavy magnesium carbonate×1.3; and each mg of aluminium hydroxide by 1.5. In this way, in each patient a reference was obtained for the efficacy of the daily prescribed doses of the binders.
At baseline, significant differences were observed between patients to be treated with phosphate binders and those who were not (Table 1). The treated subgroup was significantly younger and had less residual renal function. The concentrations of serum phosphate and PTH were higher in the treated group than controls. The urinary phosphate, the fractional excretion of phosphate, the phosphate load per unit of renal function, and the indirect estimation parameter of intestinal phosphate absorption (Table 1) were significantly greater in patients intended to receive binders than in controls. However, there were no significant differences between the subgroups in the protein catabolic rate or in the concentration of serum albumin.
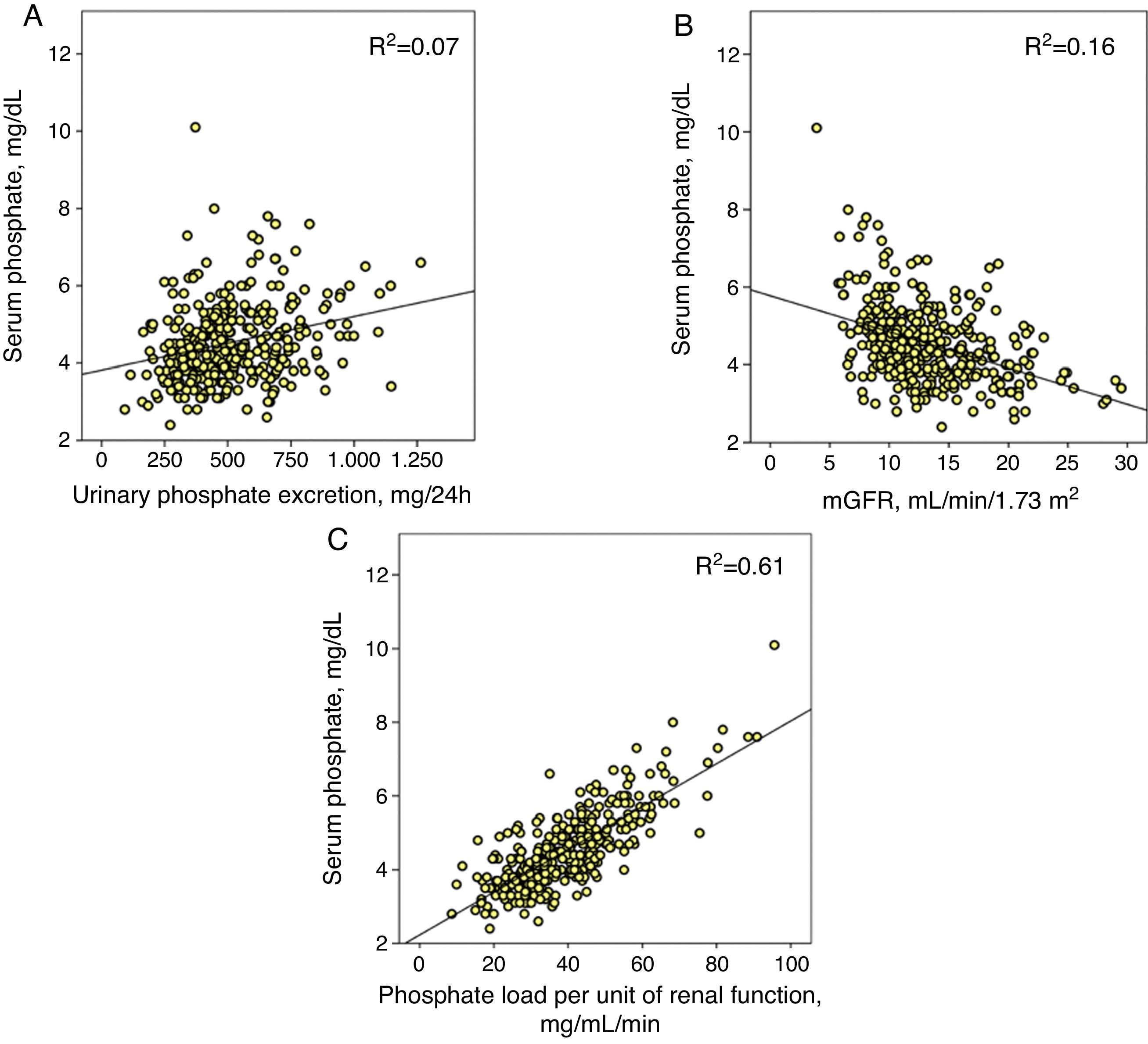
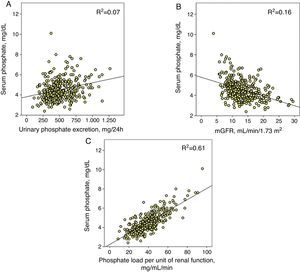
Phosphataemia determinantsIn the set of patients, the correlation between total urinary phosphate excretion and phosphataemia was adjusted with a positive linear regression model and, although statistically significant (p<0.0001), the coefficient of determination was small (R2=0.07) (Fig. 1A).
(A) Linear correlation between serum phosphate and total urinary phosphate excretion. (B) Approximation of glomerular filtration rate (mGFR) and (C) the proposed parameter as phosphate load per unit of renal function (total urinary phosphate excretion/mGFR). The three correlations have a statistical significance of p<0.0001 and each coefficient of determination is shown (R2).
The correlation between phosphataemia and kidney function (mGFR) was adjusted with an inverse linear regression (R2=0.16) (Fig. 1B).
However, the best correlation was obtained between phosphataemia and phosphate load adjusted to kidney function (R2=0.61) (Fig. 1C). A total urinary phosphate excretion of between 35 and 40mg/mL/min of glomerular filtration was the limit for the development of hyperphosphataemia (serum phosphate >4.5mg/dL).
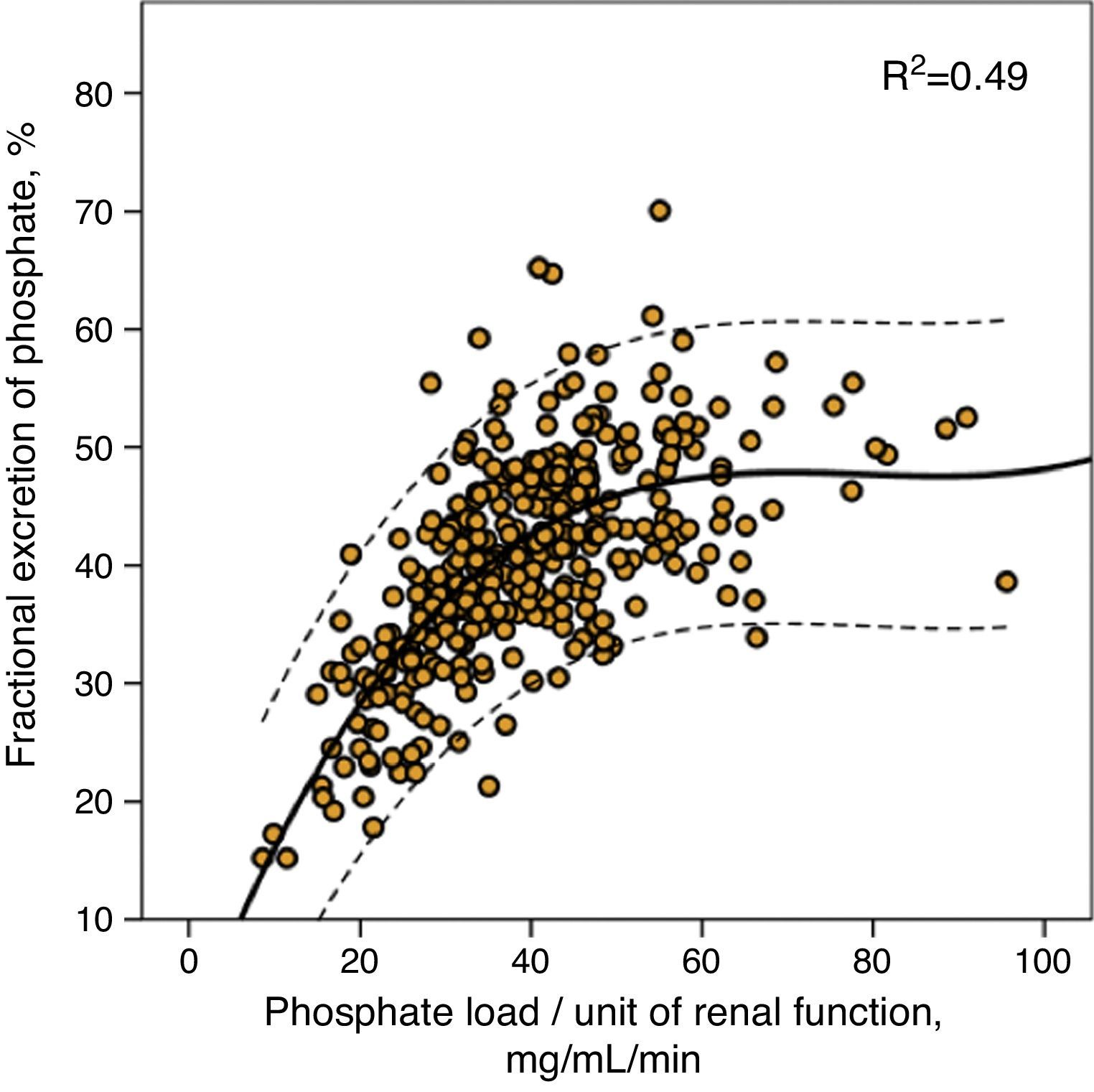
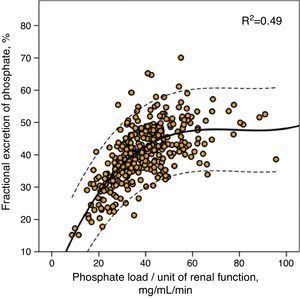
This same value of phosphate load was also that which marked the maximum limit of fractional phosphate excretion, as shown in Fig. 2, although the wide confidence intervals of this third order regression are noteworthy, which indicates that other factors existed that modified the fractional phosphate excretion in addition to the phosphate load itself.
Correlation between fractional urinary phosphate excretion and phosphate load per unit of renal function (urinary phosphate excretion/mGFR). The regression curve is shown (continuous line) with 95% confidence intervals (dotted line), as well as the coefficient of determination (R2).
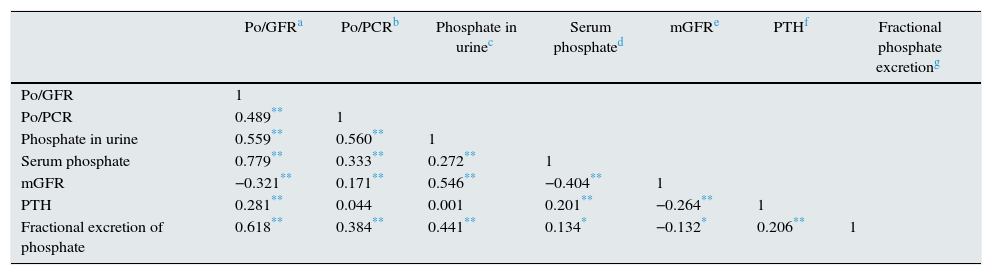
The bivariate correlation between all of the parameters of interest is shown in Table 2.
Correlation coefficient matrix between parameters of interest.
| Po/GFRa | Po/PCRb | Phosphate in urinec | Serum phosphated | mGFRe | PTHf | Fractional phosphate excretiong | |
|---|---|---|---|---|---|---|---|
| Po/GFR | 1 | ||||||
| Po/PCR | 0.489** | 1 | |||||
| Phosphate in urine | 0.559** | 0.560** | 1 | ||||
| Serum phosphate | 0.779** | 0.333** | 0.272** | 1 | |||
| mGFR | −0.321** | 0.171** | 0.546** | −0.404** | 1 | ||
| PTH | 0.281** | 0.044 | 0.001 | 0.201** | −0.264** | 1 | |
| Fractional excretion of phosphate | 0.618** | 0.384** | 0.441** | 0.134* | −0.132* | 0.206** | 1 |
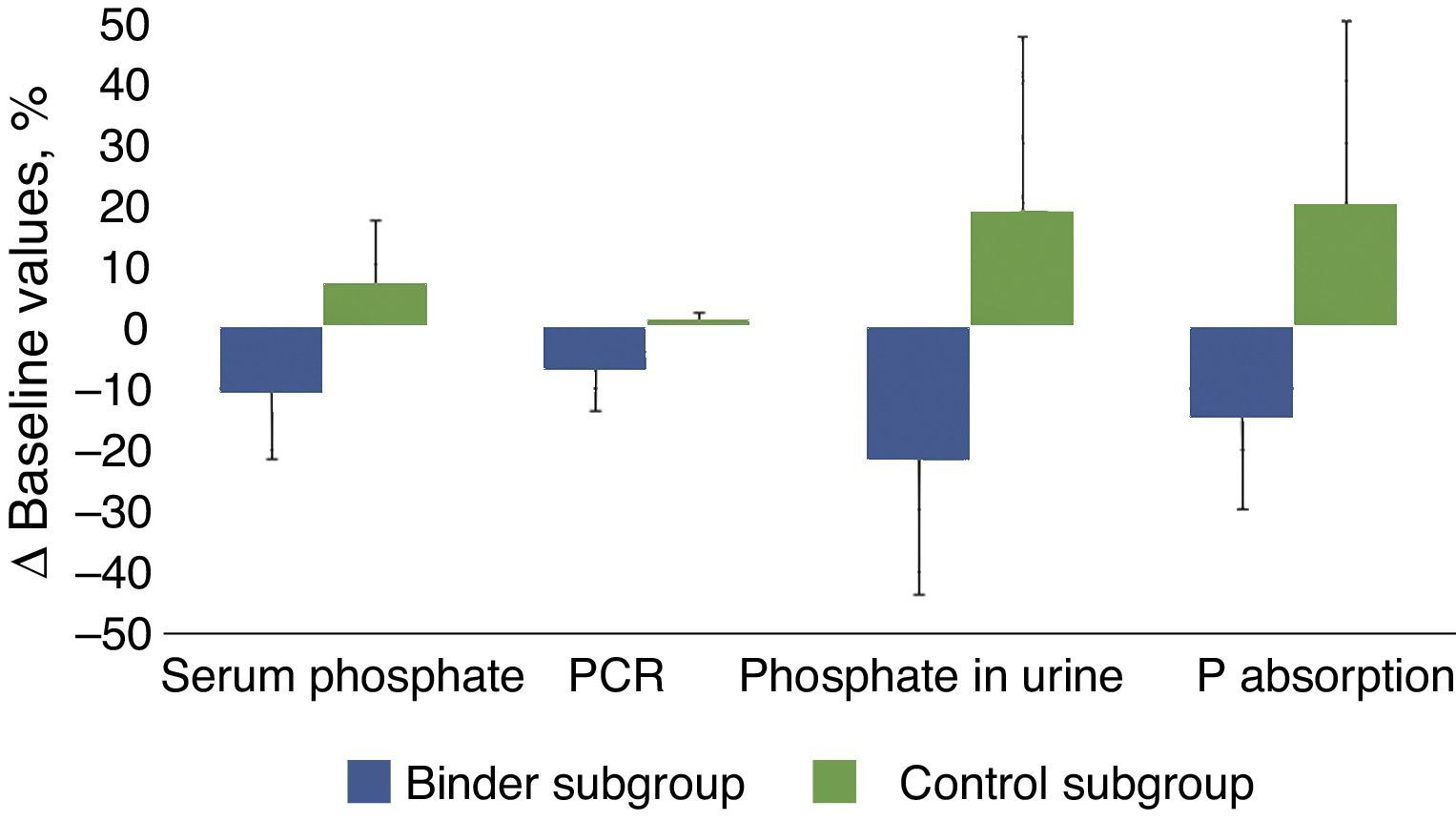
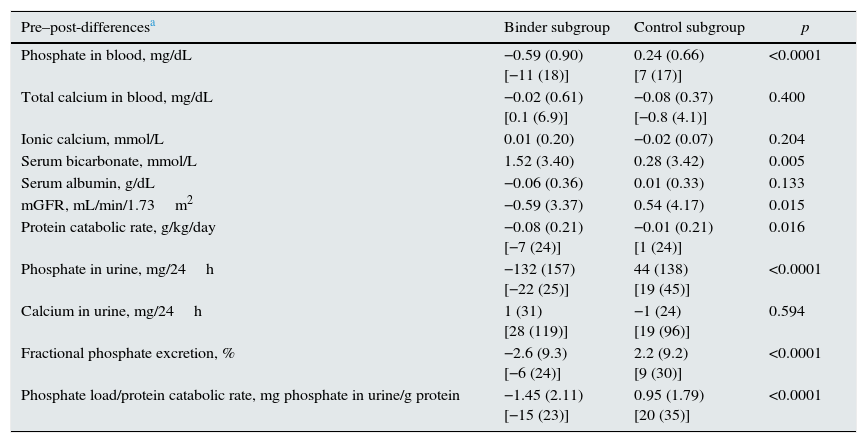
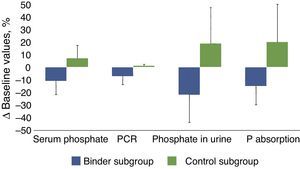
Table 3 and Fig. 3 show the changes in the parameters evaluated after the administration of phosphate binders compared with those observed in the control group. These variations are expressed as a percent change from the baseline values; phosphataemia in the group treated with binders was reduced by 11% on average, while phosphaturia decreased by 22%. Considering that the protein catabolic rate in this group of patients was reduced by 7% after treatment with binders, the efficacy that could be more directly attributed to the effect of the binders, that was quantified as the changes in phosphate excreted per gram of the protein catabolic rate, was 15% on average (Table 2, Fig. 3).
Changes in the biochemical parameters before and after treatment with binders (binder subgroup) or dietary advice (control subgroup). The absolute values of these changes are shown in percentages with respect to the baseline values.
| Pre–post-differencesa | Binder subgroup | Control subgroup | p |
|---|---|---|---|
| Phosphate in blood, mg/dL | −0.59 (0.90) [−11 (18)] | 0.24 (0.66) [7 (17)] | <0.0001 |
| Total calcium in blood, mg/dL | −0.02 (0.61) [0.1 (6.9)] | −0.08 (0.37) [−0.8 (4.1)] | 0.400 |
| Ionic calcium, mmol/L | 0.01 (0.20) | −0.02 (0.07) | 0.204 |
| Serum bicarbonate, mmol/L | 1.52 (3.40) | 0.28 (3.42) | 0.005 |
| Serum albumin, g/dL | −0.06 (0.36) | 0.01 (0.33) | 0.133 |
| mGFR, mL/min/1.73m2 | −0.59 (3.37) | 0.54 (4.17) | 0.015 |
| Protein catabolic rate, g/kg/day | −0.08 (0.21) [−7 (24)] | −0.01 (0.21) [1 (24)] | 0.016 |
| Phosphate in urine, mg/24h | −132 (157) [−22 (25)] | 44 (138) [19 (45)] | <0.0001 |
| Calcium in urine, mg/24h | 1 (31) [28 (119)] | −1 (24) [19 (96)] | 0.594 |
| Fractional phosphate excretion, % | −2.6 (9.3) [−6 (24)] | 2.2 (9.2) [9 (30)] | <0.0001 |
| Phosphate load/protein catabolic rate, mg phosphate in urine/g protein | −1.45 (2.11) [−15 (23)] | 0.95 (1.79) [20 (35)] | <0.0001 |
Bar graph showing percentage changes of the following parameters: serum phosphate, protein catabolic rate (PCR), urinary phosphate excretion (phosphate in urine) and the proposed intestinal phosphate absorption estimation parameter (total urinary excretion/protein catabolic rate), in patients treated with phosphate binders (binder subgroup) or dietary advice (control subgroup). The statistical significance of these comparisons is shown in Table 2.
In the control group, despite the dietary advice, it was observed an increase in the phosphate concentrations in blood and urine excretion and, taking into account the minimal variations in the protein catabolic rate, the parameter of intestinal phosphate absorption increased by 20% on average.
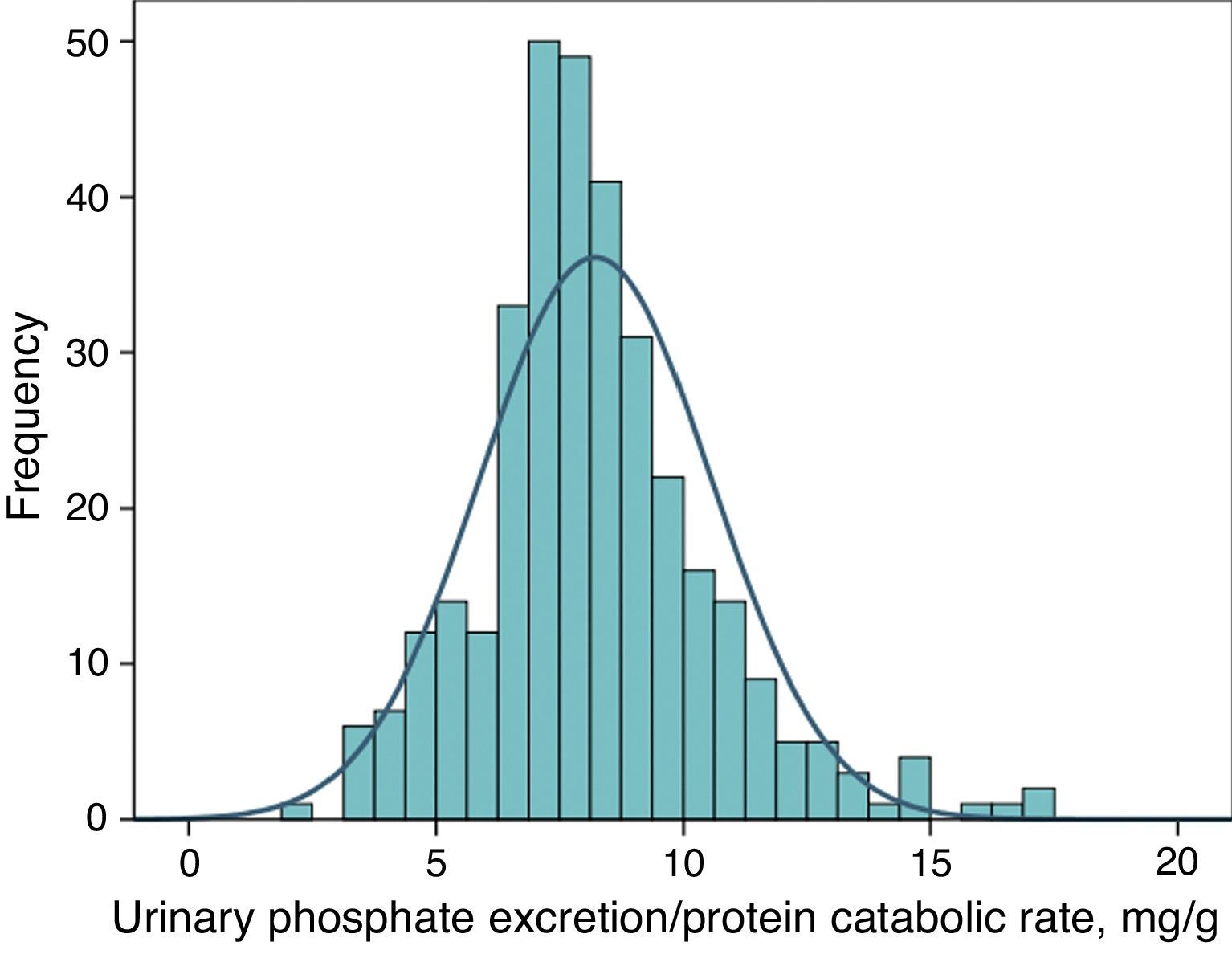
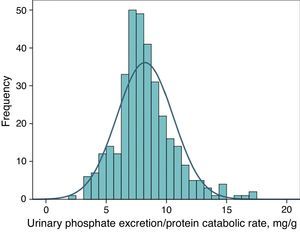
Urinary phosphate excretion adjusted to protein intake as a marker of a binder's efficacyFig. 4 shows the frequency distribution of urinary excretion of phosphate/protein catabolic rate at baseline (before treatment) in the patients evaluated. The average value was 8.2mg per gram of the protein catabolic rate, with confidence intervals of 95% between 4.5 and 12.6.
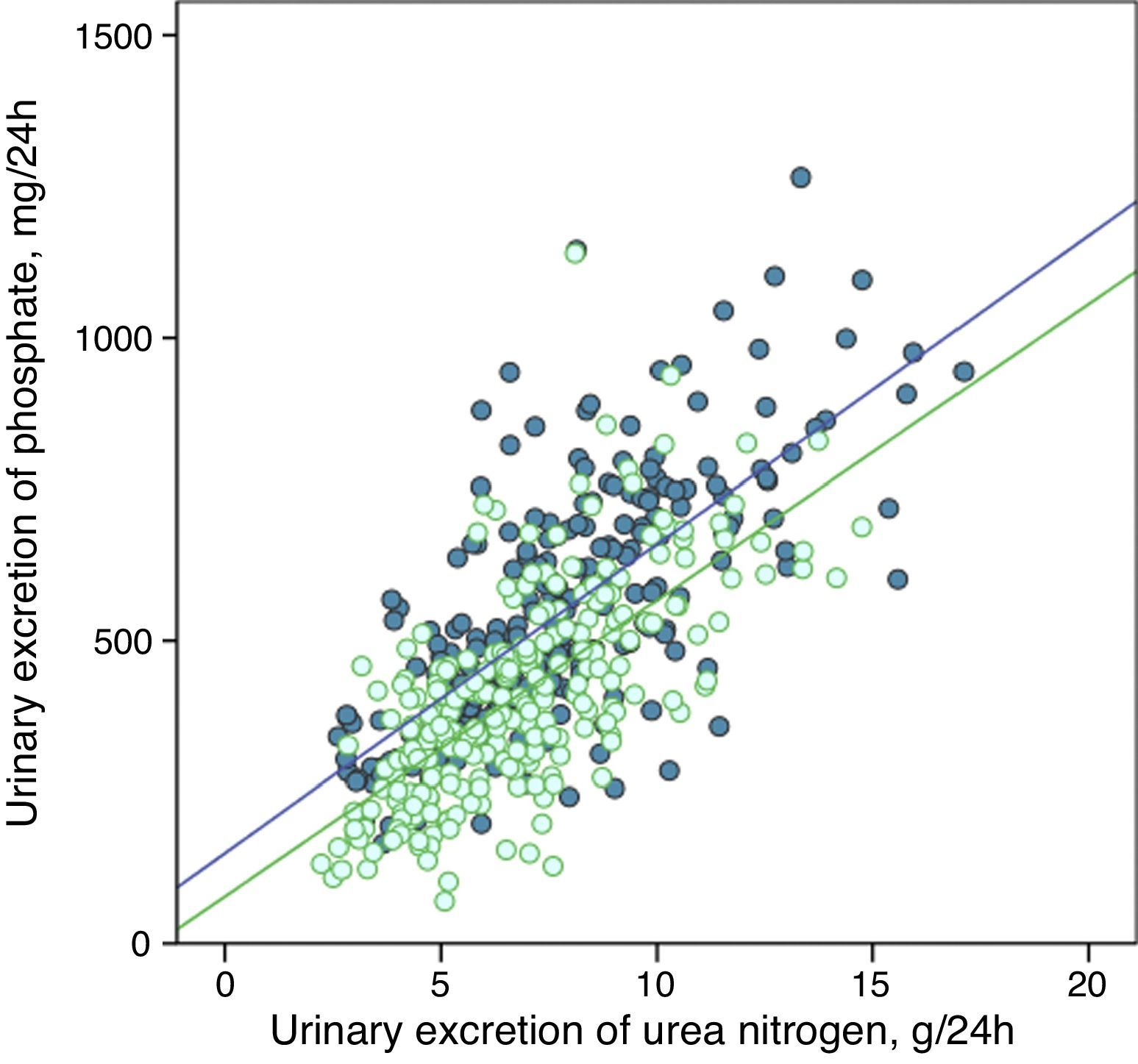
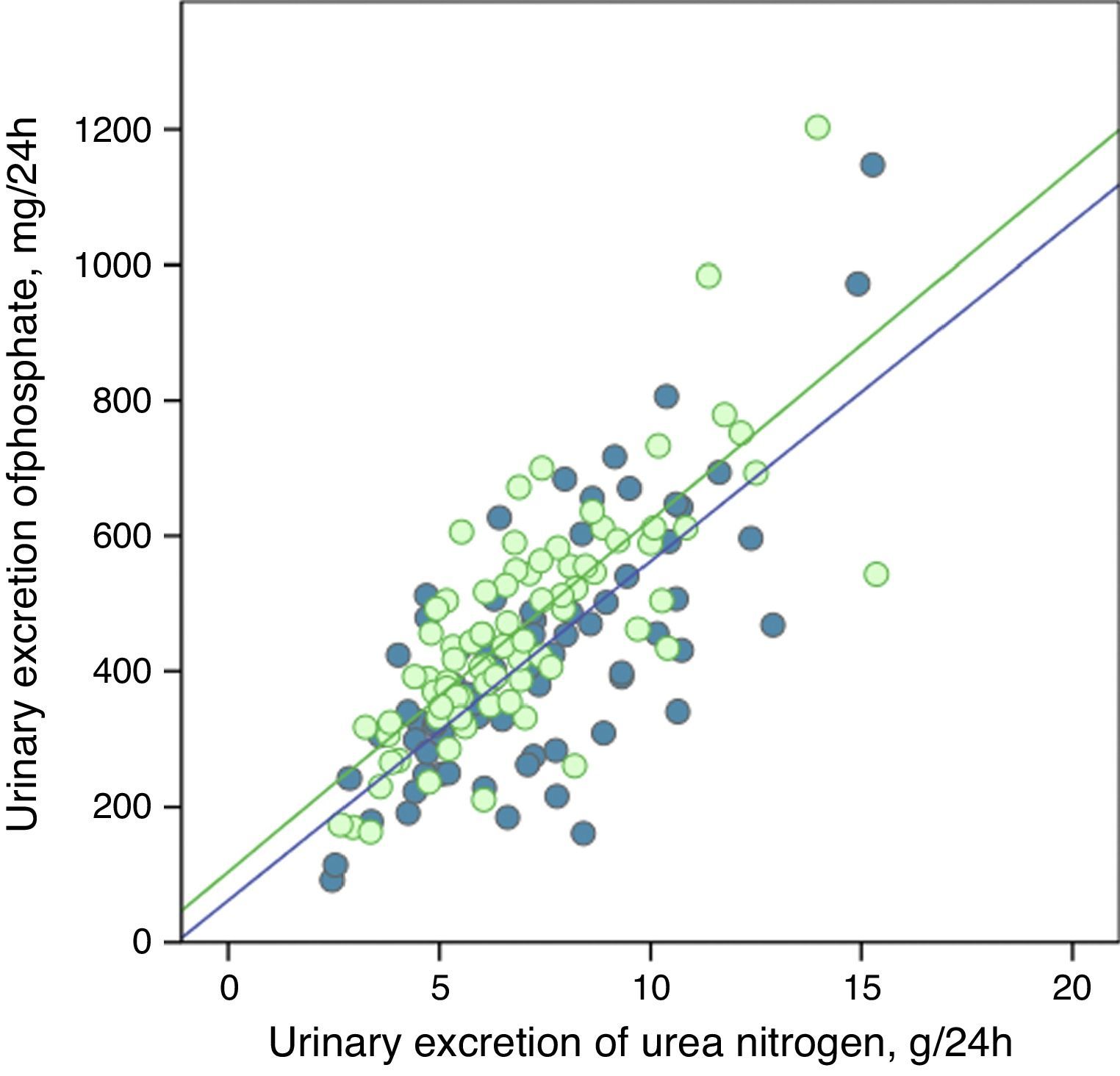
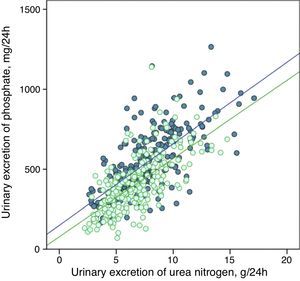
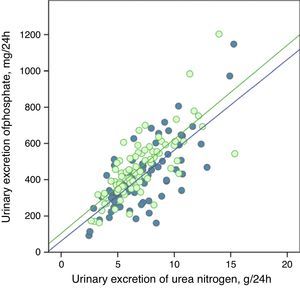
The urinary excretion of phosphate and urea nitrogen was strongly correlated in a linear manner before and after treatment with phosphate binders (Fig. 5), or after the dietary advice in the control subgroup (Fig. 6).
Linear correlation between urinary phosphate excretion and urinary urea nitrogen excretion at baseline and after treatment with phosphate binders (“binder” subgroup). Regression lines corresponding to the two study periods (baseline and post-treatment) are depicted. R2 in baseline stage=0.50; p<0.0001; R2 post-treatment=0.47; p<0.0001.
Linear correlation between urinary excretion of phosphate and urinary excretion of urea nitrogen at baseline stage and after dietary advice (“control” subgroup). Regression lines corresponding to the two study periods (baseline and post-treatment) are depicted. R2 in baseline stage=0.55; p<0.0001; R2 post-treatment=0.56; p<0.0001.
The regression lines were parallel in both cases and had very similar coefficients of determination (Figs. 5 and 6). In the group of patients treated with binders, at any given value of urinary urea nitrogen excretion, phosphate excretion was less after treatment.
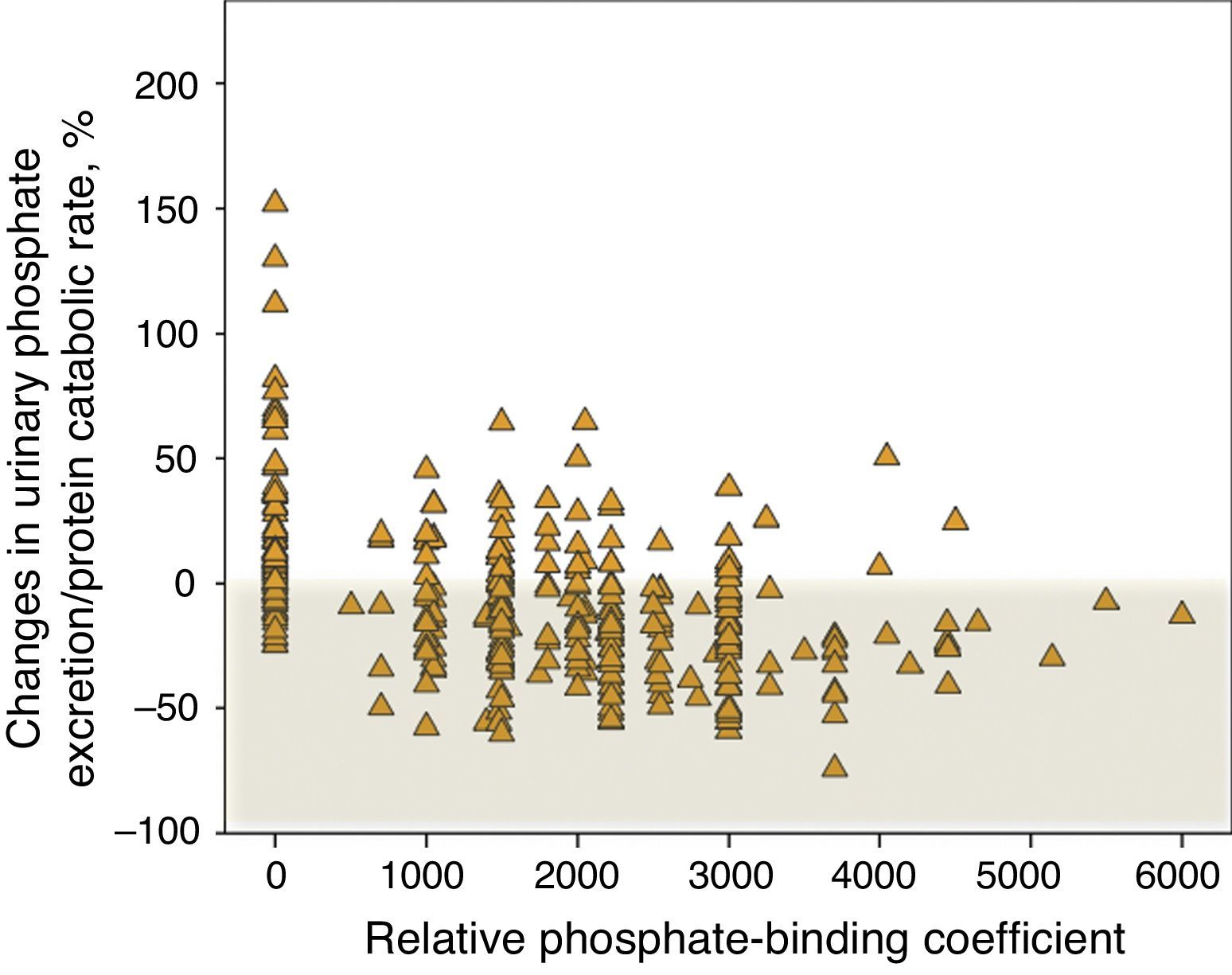
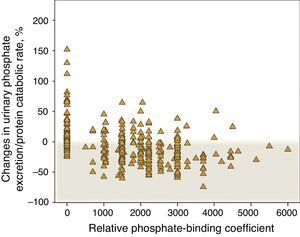
Fig. 7 shows the percent change in urinary phosphate excretion per gram of the protein catabolic rate in control patients that did not receive binders and in those treated with binders, specifying the binding power. An increase in this parameter was very common in the control patients and it decreased in the majority of patients treated with binders. However, according to this parameter, a lack of response to the treatment with phosphate binder is observed in 22% of patients, which is remarkable; and there is progressive loss in response with the highest doses of binding power prescribed.
Percentage changes in urinary phosphate excretion/protein catabolic rate according to the phosphate-binding coefficient described by Daugirdas et al.17 A binding coefficient of 0 corresponds to patients that were not treated with binders (“control” subgroup”).
The percentage changes of phosphataemia after binder treatment strongly correlated (R2=0.47) with the percentage changes in the “phosphate load” (urinary phosphate excretion per unit of renal function), but in a much weaker form (R2=0.09) with the percentage changes in non-adjusted urinary phosphate excretion.
DiscussionThe main results of this study are: (1) Phosphate load per unit of renal function is the parameter that best correlates with phosphataemia in CKD. (2) Maximum fractional excretion of phosphate is reached with an average phosphate load of approximately 40mg/mL/min of glomerular filtration. (3) There is a strong linear correlation between the urinary excretion of phosphate and urea nitrogen, which is maintained after treatment with binders. (4) Urinary phosphate excretion per gram of the protein catabolic rate could be used to estimate a binder's efficacy. (5) According to this later parameter, the efficacy of the binders at conventional doses was only an average of 15% reduction in the intestinal absorption of phosphate in our study population with advanced CKD.
Total urinary phosphate excretion is considered a reliable method of dietary phosphate intake-absorption in humans and experimental animals.7–11 However, unlike the concentrations of serum phosphate, the dietary phosphate load in patients with CKD has not demonstrated an association with variables, which is a result of major interest (mortality, CKD progression)8,18,19: these studies also show a weak relationship between phosphataemia and total phosphate urinary excretion.
In this study, we have provided some data that could explain these paradoxical findings in a simple manner. It is true that the relationship between phosphataemia and total urinary phosphate excretion, although statistically significant, is rather weak as reflected by the low coefficient of determination, and as being observed in other studies.8,18,19 However, the parameter that proves to be strongly correlated with phosphataemia is the phosphate load (total urinary phosphate excretion) per unit of renal function. According to these findings, the most important factor is not the total amount of phosphate ingested, but rather the amount of phosphate ingested/absorbed, which can be managed by a specific renal function. A person who absorbs 1000mg of phosphate daily with a glomerular filtration rate of 100mL/min, would have a phosphate load of 10mg/mL/min per unit of renal function, while in a patient with a glomerular filtration rate of 10mL/min, the same phosphate absorption would induce a load of 100mg/mL/min. With the same amount of phosphate absorbed, the consequences are different, as the phosphate excretion capacity increases in a linear manner, but up to a limit that is reached at approximately 35–40mg of phosphate per mL/min of glomerular filtration, as shown in Fig. 2. Fractional phosphate excretion reaches its maximum level of compensation (an average of around 50%) with these phosphate loads and, in this way, higher loads could cause a positive phosphate balance, as reflected in our study, by the development of hyperphosphataemia.
The parameter proposed in this study as an estimator of intestinal phosphate absorption and, thus, as a potential control parameter of the efficacy of the binders, is the relationship between urinary phosphate excretion and the protein catabolic rate, i.e. how much phosphate is excreted in the urine per gram of the estimated protein intake.
The potential advantages of this parameter are: (1) Simple and cost-effective measurement of all the biochemical data contained. (2) Compensation for possible errors in collecting the 24-h urine sample by calculating the parameter as a relation between two determinations having the same error. (3) Excellent correlation between urinary excretion of phosphate and urea nitrogen, which is maintained after treatment with binders, and which allows a reduction in dietary intake (protein) to be valued as a non-desirable response to the prescription of a binder. (4) Better identification and assessment of non-compliance with treatment. This determination allows the precise identification of those patients that do not comply with the binder treatment, but had decreased the phosphate intake, which would result in decreases in phosphataemia. Needless to say, these decreases must not be attributed to the pharmacological effect of the binder. In these patients, a decrease in total urinary phosphate excretion is observed, but without significant variations in the relationship between urinary phosphate and the protein catabolic rate.
The main disadvantages of this parameter of estimation binder efficacy are: (1) Need to measure these parameters in urine, meaning that this is not applicable in dialysis patients. (2) Errors in sample collection, which is very dependent on the patient's cooperation, although, as noted previously, the use of the relation between two points of biochemical data subject to the same inaccuracy cancels out the possible error. (3) Variation in the ratio of phosphate absorbed per gram of protein ingested, which depends on the characteristics of the diet12,13 or the inter-individual differences in intestinal absorption of phosphate.20,21 (4) Inaccuracy in the interpretation of this parameter in clinical situations associated with an increase in endogenous phosphate generation (high remodelling, osteolysis, cell lysis), or in non-dietary urea nitrogen generation (hypercatabolic state or treatment with corticosteroids).
In this study, a baseline Gaussian distribution of this parameter was observed, and these differences between patients could be due to these individual factors. As the differences used to determine the efficacy of a binder are calculated between two samples from the same patient, the individual characteristics of intestinal phosphate absorption should not influence the result.
The estimation of the overall efficacy of the binders used in our study through the proposed parameter was only 15%, although it should be noted that this was the result in standard clinical conditions (“real life”), without taking into account the patient's compliance. As shown in Fig. 7, all patients treated with binders had a reduction in urinary phosphate excretion per gram of protein intake, bar 22% who could be classified as having failed to comply, due to the similarity that the biochemical data of compliant patients showed in relation to those of the explicitly non-compliant patients. In this way, the proposed parameter could also be used as a treatment compliance control.
With the exception of those who, in theory, could be considered non-compliant, the efficacy of the binders studied would reach 24±14%.
Although a significant inverse linear correlation existed between the phosphate absorption parameter and the prescribed binding power, this relationship was attenuated in the high binding power extreme, a finding that would support the hypothesis proposed in other studies on the negative effect between the amount of medication prescribed and effective treatment compliance.22
Another unexpected and surprising finding in this study was the significant increase in the parameter of urinary phosphate per gram of the protein catabolic rate (Po/PCR) in patients with a low-phosphate diet without binders. This result could be explained by non-compliance with the dietary recommendations, which is unlikely if there is no a concordant increase in urea nitrogen; another possible explanation could be a significant qualitative change in the phosphate content of foods, especially those processed industrially with added phosphates, this is also unlikely, taking into account the habits and culinary customs of the rural society to which most of the patients belong to. An alternative hypothesis that could help explain this finding would be related to the efficacy of intestinal phosphate absorption if patients are on a low phosphorus diet. In test animals, it was proven that a diet low in phosphate increases the specific intestinal transporters (NaPi-IIa and NaPi-IIb) by vitamin D-independent mechanisms.23 More studies on CKD patients would be necessary to confirm if, as in the test animals, reducing dietary phosphate increases its absorptive efficacy and, if this is confirmed, strategies to control phosphate may need to be seen from different angle and it would be necessary to search on new drugs to better control phosphate in uraemia.
The practical clinical application of this binder efficacy estimation parameter is limited to pre-dialysis patients with advanced CKD. According to our experience, this parameter is a very useful tool for adjusting treatment and differentiating between hyperphosphataemia due to diet, intestinal absorption or non-compliance with the treatment.
This parameter could also be used in clinical studies to analyze the efficacy of phosphate binders, as well as to determine the minimum effective doses to obtain a specific therapeutic effect.
With the data collected in this study, a question could be raised in relation with what would be the minimum efficacy of a binder to be useful in controlling the phosphate balance in a dialysis patient. For example, in an anuric haemodialysis patient of 70kg, with a daily protein intake of 0.8g/kg and an intestinal absorption of 8mg of phosphate per gram of protein (extrapolation from the average values observed in this study), the resulting total daily phosphate load would be 448mg. Thus, the weekly load of phosphate would be 3136mg; subtracting 1600–2600mg of phosphate as amounts removed with the 3 weekly conventional haemodialysis sessions,24,25 there would be necessary a reduction of 536–1536mg of intestinal phosphate absorption (17–49% less than the baseline absorption). This numbers underscores the importance of maintaining an adequate clearance and negative balance of phosphate through extrarenal purification an adequate protein intake, and not to expect to achieve this therapeutic objective exclusively by using the phosphate binders available.
This study has certain limitations. It is a retrospective data analysis in which a large variety of phosphate binding agents were used, without efficient control of compliance, at a single site, and in a group of patients who were solely representative of the local population as regards their dietary peculiarities. The study was also not designed to compare the specific efficacy of each binder prescribed, which is why it was decided to present the theoretical power of the binders and their associations, but not the commercial preparations.
In conclusion, urinary phosphate excretion adjusted to glomerular filtration rate or protein intake are two parameters that could reflect phosphate load and intestinal phosphate absorption, respectively. In addition to their clinical uses in pre-dialysis CKD patients, they could be useful tools for assessing the role of phosphate in CKD complications, and for estimating the pharmacological efficacy of phosphate binders.
Conflicts of interestThe authors declare that there are no conflicts of interest.
Please cite this article as: Caravaca F, Caravaca-Fontán F, Azevedo L, Luna E. Cómo estimar la eficacia de un captor del fósforo. Nefrologia. 2017;37:311–319.