Acute kidney injury in the critically ill represents an independent risk factor of morbidity and mortality in the short and long terms, with significant economic impacts in terms of public health costs. Currently its diagnosis is still based on the presence of oliguria and/or a gradual increase in serum creatinine, which make the diagnosis a delayed event and to detriment of the so-called ‘therapeutic window’. The appearance of new biomarkers of acute kidney injury could potentially improve this situation, contributing to the detection of ‘subclinical acute kidney injury’, which could allow the precocious employment of multiple treatment strategies in order to preserve kidney function. However these new biomarkers display sensitive features that may threaten their full capacity of action, which focus specifically on their additional contribution in the early approach of the situation, given the lack of specific validated treatments for acute kidney injury. This review aims to analyze the strengths and weaknesses of these new tools in the early management of acute kidney injury.
La lesión renal aguda en los pacientes críticos representa un factor de riesgo independiente de la morbilidad y la mortalidad a corto y a largo plazo, con un tremendo impacto económico en cuanto a los costes en salud pública. Por el momento, el diagnóstico de la lesión renal aguda sigue basándose en la presencia de oliguria o en un aumento gradual de la creatinina sérica, hecho que retrasa el diagnóstico, en detrimento de la llamada «ventana terapéutica». La aparición de nuevos biomarcadores de lesión renal aguda podría mejorar esta situación y contribuir a la detección de la «lesión renal aguda subclínica», lo que permitiría el uso precoz de múltiples estrategias de tratamiento con el objetivo de preservar la funcionalidad renal. No obstante, los nuevos biomarcadores presentan características que podrían vulnerar su capacidad de acción, centrada concretamente en aportar un valor añadido al abordaje precoz de la enfermedad, dada la falta de tratamientos específicos validados para la lesión renal aguda. Esta revisión tiene como objetivo analizar los puntos fuertes y débiles de esta nueva herramienta para el diagnóstico temprano de la lesión renal aguda.
In the article “Dissent, Dogmatism and Belief Polarization”,1 published in The Journal of Philosophy, Thomas Kelly refers to a phenomenon called “belief polarization” by which exposure to the same evidence, far from bringing those who have different opinions closer, usually makes the disagreement between them become more pronounced: we are more demanding with anything that contradicts our belief and more permissive with what favors our own point of view.
This phenomenon may partly explain our acceptance or rejection of the use of new biomarkers in the diagnosis of acute kidney injury (AKI).
In 2005, the American Society of Nephrology Renal Research Report (ASNRRR) assigned the highest research priority to the discovery and standardization of new AKI biomarkers.2
AKI in critically ill patients is an independent risk factor that increases morbidity and mortality in the short and long term, with a tremendous financial impact in terms of health costs.3 AKI is also a gateway to chronic kidney disease (CKD).4 It is noteworthy that, after an AKI episode, 7.8 of every 100patients/year develop CKD and 4.9 per 100patients/year will develop advanced chronic kidney disease.5
There has been numerous preventive or curative strategies for AKI that have been either ineffective or insufficiently validated to be routinely recommended.4
The most important risk factor for AKI is the pre-existing CKD, which increases its risk up to 10 times.6,7,8
Some processes, such as endothelial dysfunction,9 myocardial remodeling,10 epigenetic factors11 and increased oxidative stress,12 are factors that could explain the increased risk of morbidity and mortality that persists long after the AKI episode.
Another reason that may explain the negative outcome of AKI patients is the late recognition of kidney injury leading to delayed interventions.
We must bear in mind that the diagnosis of AKI is based on indirect markers of kidney damage, that are not very sensitive or specific, to the detriment of the so-called “therapeutic window”.4
In this context, there are various limitations of serum creatinine (sCr): sCr comes into play as a functional marker when more than 50% of the glomerular filtration rate has been lost and is only useful after a stationary state has been reached. The latter may differ over time, sometimes up to 48h, especially in patients in intensive care units (ICU).12 On the other hand, the excretion of creatinine does not depend on the load filtered solely by the glomeruli, but also on that secreted by the kidney tubules, which normally varies from 5 to 20% of total excretion, and may increase to 50% as a compensatory mechanism when the glomerular filtration rate (GFR) decreases.13 Also, even if there were genuinely a fall in GFR, the sCr might not increase or increase late as a result of recruitment of the renal functional reserve; also the sCr could be “diluted” as a result of a profuse positive balance, which is frequently observed after resuscitation maneuvers, especially in patients in the ICU.14 Finally, the eGFR seems to overestimate renal function in patients admitted to ICU during a long period of time. This was well demonstrated in a secondary analysis of the EPANIC study, in which 757 patients participated and which showed that a reduction in production of sCr positively correlated with the length of stay in the ICU, probably due to loss of muscle mass.15
The other parameter of AKI is oliguria, which is neither sensitive nor specific, since it could occur as a result of a kidney injury, but may also reflect an adaptive physiological response to either intracellular dehydration or hypovolemia.16 Only a small proportion of oliguric patients in the ICU have in fact a sustained fall in GFR which is reflected in an increase in sCr.16 Similarly, Mandelbaum et al.17 showed that only pronounced (<0.3mL/kg/h) or prolonged (>12h) episodes of oliguria were associated with the need to start renal replacement therapy (RRT) or with increased hospital mortality.
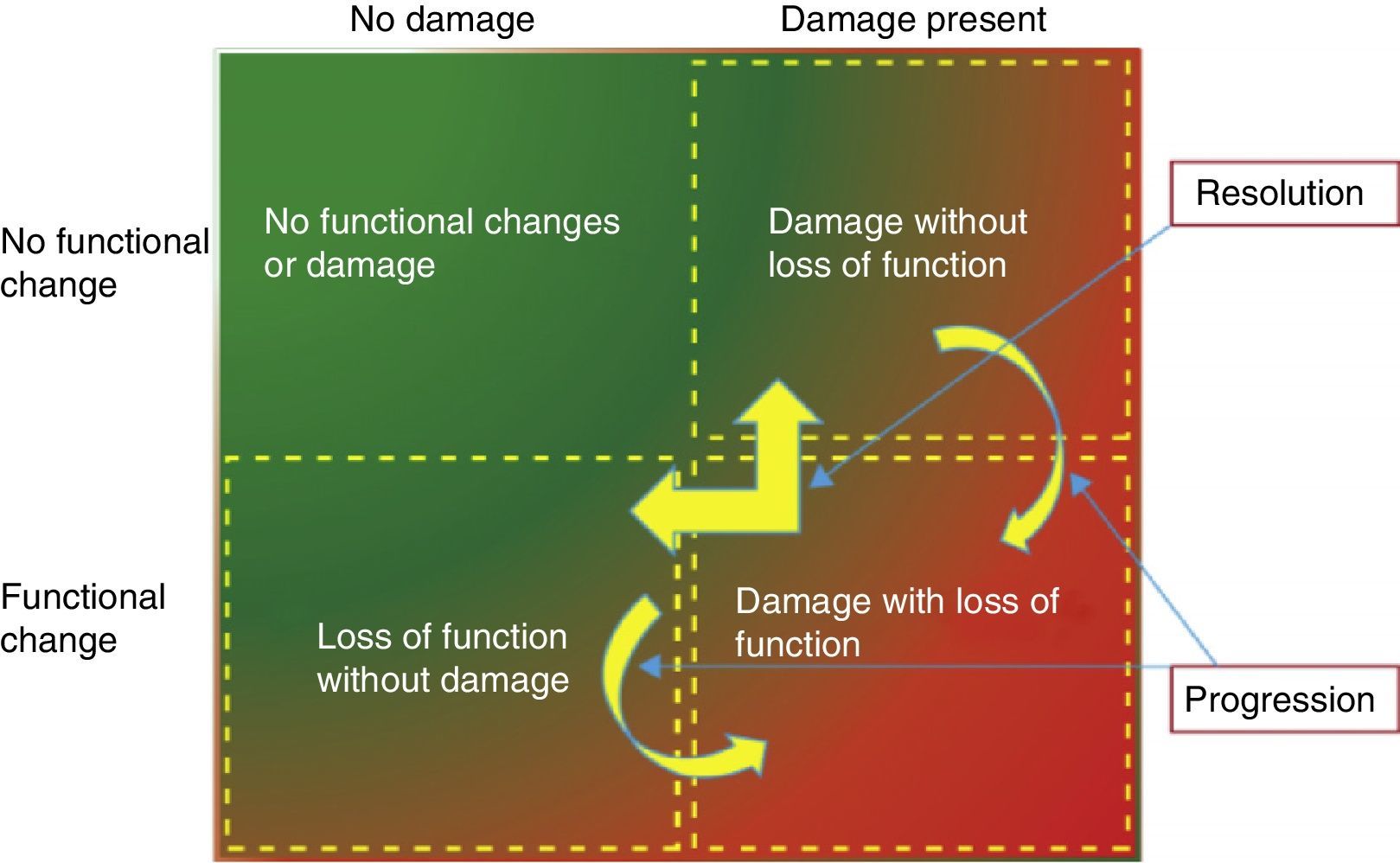
The most recent biomarkers promise: to identify early patients at risk of AKI; to diagnose AKI earlier than with other conventional tests; to indicate the need to initiate RRT and also predict the risk for progression to CKD.18,19 The objective is to accomplish more timely interventions the more favorable outcomes in patients with AKI. The availability of these new biomarkers and the evaluation of simultaneous combinations of functional and tissue damage biomarkers may help stratify patients into 4 subgroups: no change in biomarkers, only changes in functional biomarkers, only changes in biomarkers of tissue injury, or changes in the 2, functional and injury biomarkers (Fig. 1).
Functional use of the new acute kidney injury biomarkers. From: Acute Dialysis Quality Initiative (ADQI). Available at: http://www.ADQI.org [accessed 10.01.13].
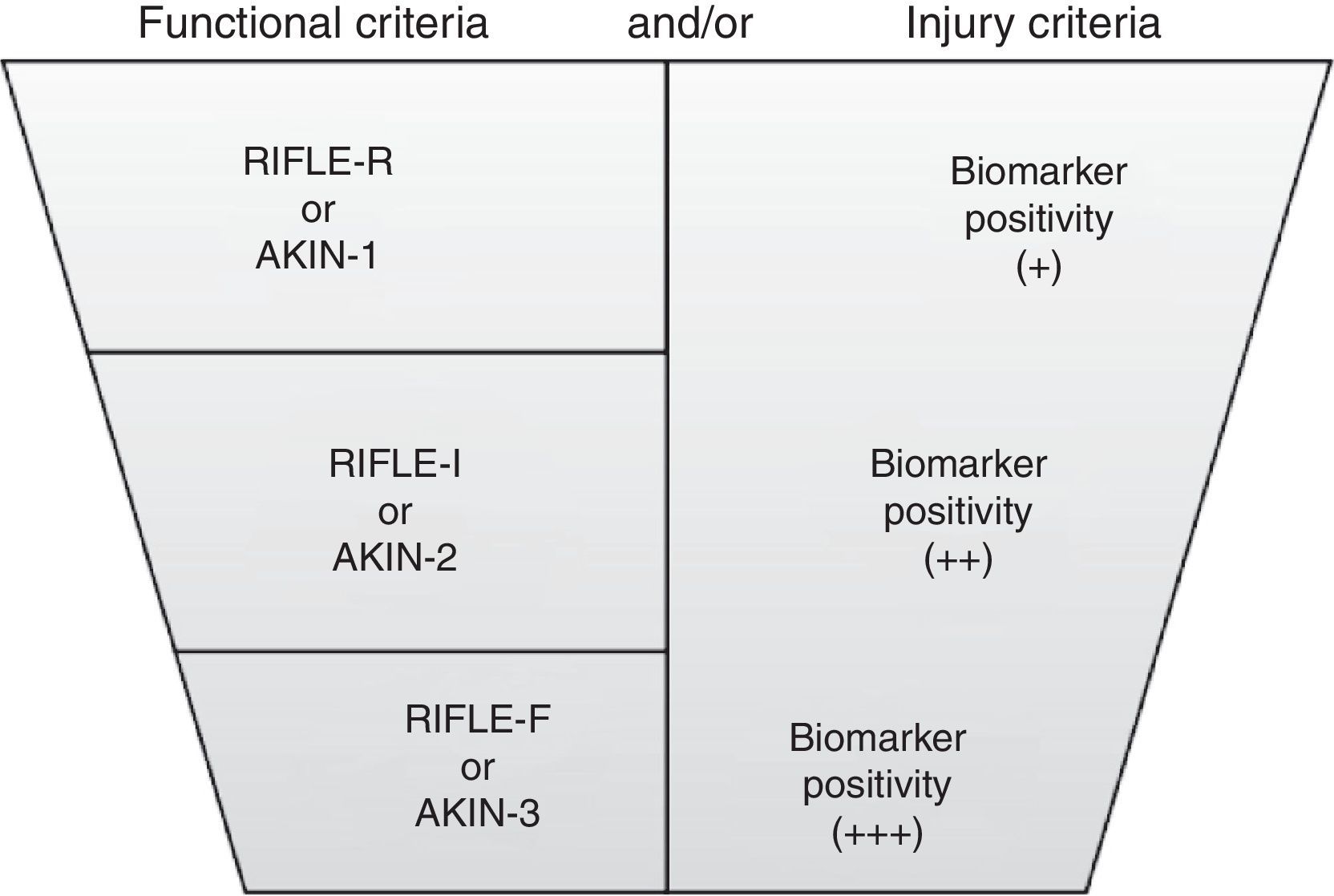
This new approach allows the identification of a new category of patients with AKI, called “Subclinical AKI”, represented by an increase in markers of tissue injury without a simultaneous reduction of kidney function. Based on this new conceptual framework, a modification of the KDIGO criteria recommend the incorporation of kidney injury markers, to sCr, GFR and diuresis (Fig. 2).20
New criteria for the diagnosis of acute kidney injury. From: Acute Dialysis Quality Initiative (ADQI). Available at: http://www.ADQI.org [accessed 10.01.13].
This new approach has been recognized and recently incorporated to the definition of AKI in Australia and New Zealand regional practice guides, without clarifying what kind of biomarker to use or its cutoff value.21
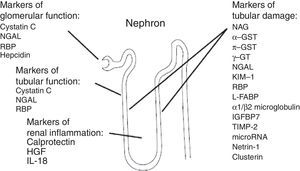
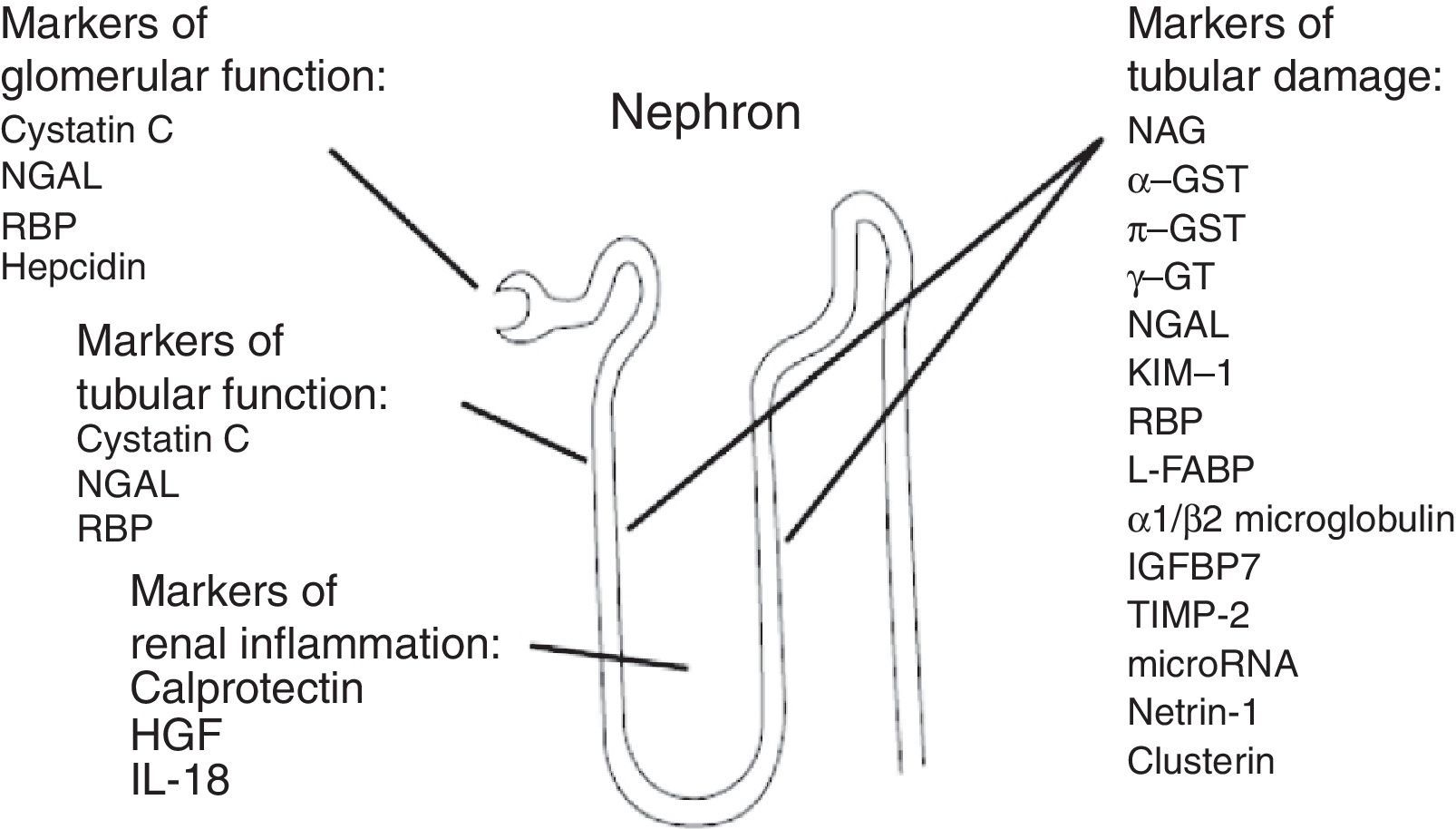
New AKI biomarkers vary in their source, in their function, in their distribution and in the time of their release after kidney injury (Fig. 3).
Source of acute kidney injury biomarkers. g-GT, g-glutamyl transpeptidase; GST, glutathione S-transferase; HGF, hepatocyte growth factor; IGFBP-7, insulin-like growth factor-binding protein 7; IL-18, interleukin 18; KIM-1, kidney injury molecule; L-FABP, liver-type fatty acid-binding protein; NAG, N-acetyl-d-glucosaminidase; NGAL, neutrophil gelatinase-associated lipocalin; RBP, retinol-binding protein; TIMP-2, tissue inhibitor of metalloproteinases.38
Most evidence has focused on neutrophil gelatinase-associated lipocalin (NGAL), either urinary or plasmatic, kidney injury molecule (KIM-1), interleukin 18 (IL-18), N-acetyl-d-glucosaminidase (NAG), cystatin C (Cys-C), tissue inhibitor of metalloproteinases (TIMP-2) and insulin-like growth factor-binding protein 7 (IGFBP-7).22
In homogeneous populations of patients such as those undergoing cardiopulmonary bypass surgery, biomarkers such as NGAL, KIM-1, Cys-C and IL-18 appear to be an attractive instrument to detect AKI before changes in sCr are observed.23–27 However, in heterogeneous populations with different pathologies, such as ICU patients or patients from the emergency room, the individual performance of these biomarkers is markedly lower.23,28 The performance of most biomarkers is variable and depends on the underlying pathology affecting the patient, the ethiology of AKI, clinical aspects, associated comorbidities and the timing of the measurements.22
Some biomarkers, as for example NGAL, reflect the severity of the triggering disease rather than being specific to the kidney injury.29–31
NGAL is a gelatinase-associated human neutrophil that exists as a 25kDa monomer, as a 45kDa homodimer and as a gelatinase it conjugates with a 135kDa heterodimeric conformation.32 Monomeric and heterodimeric forms are mainly produced by tubule epithelial cells, while the homodimeric form occurs mainly in activated neutrophils.32 Circulating NGAL filters through the glomerular barrier and is fully reabsorbed by megalin-mediated endocytosis at normal proximal tubule cells level. There are commercial kits to measure uNGAL (urinary NGAL) or pNGAL (plasmatic NGAL). Under stable conditions, plasma and urine concentrations are around 20ng/mL. The marker increases 2–4h after the kidney damage occurs. The NGAL has been associated with a probable kidney protective effect, since it is released by nephron segments where they can form a complex with siderophores that binds to iron; thus, the iron released by the damaged tubular cells is chelated which prevents the formation of hydroxyl radicals and superoxide anions.
NGAL levels also rise in experimental sepsis and systemic inflammation models, suggesting that the release into the urinary system is the response of the kidney to a systemic infection or to the local urogenital infection.33 This is an important limitation to the use of NGAL, especially in terms of its diagnostic performance during AKI associated to sepsis.32
Cystatin C is a 13kDa protein produced by all nucleated cells that is released into the plasma at a constant rate, regardless of sex, race, muscle mass and hydration level. It circulates in plasma and it is not bound to any protein, is freely filtered through the glomerulus and is fully reabsorbed by megalin-mediated endocytosis at proximal tubule cells level.
Diabetes, high doses of corticosteroids, hypertriglyceridemia, hyperbilirubinemia or rheumatoid factor may affect the analysis of Cys-C.33 It is not secreted by tubule cells and is not detectable in the urine of healthy subjects. Consequently, increased urinary Cys-C values during AKI reflects decreased reabsorption at proximal tubule level.33 Urine Cys-C (u Cys-C) appears as an earlier and more sensitive marker of AKI as compared with pCys-C (plasma). However, the concentration of pCys-C is mainly correlated with GFR, even in a range where sCr is not able to detect early changes in GFR (60–90mL/min). It appears in urine 12–24h after kidney damage.
KIM-1 is a cell membrane glycoprotein whose mRNA levels increase more than any other gene after kidney injury. The 90kDa soluble ectodomain is part of the dependent metalloproteinase complexes and it is released into the tubule by epithelial cells making it readily detectable in urine. During kidney injury, KIM-1 can facilitate remodeling of the injured epithelium. It appears 12 to 24h after kidney damage.
IL-18 is an 18kDa proinflammatory cytokine produced mainly by activated neutrophils, mononuclear cells, macrophages and non-immune cells, including the proximal tubule cells. It is an important ischemic AKI mediator.33 It can be found in urine and plasma. It appears 6–24h after kidney damage.
NAG is a large-sized protein (approximately 140kDa) that originates in the lysosomes of proximal tubule cells. Its high molecular weight prevents glomerular filtration and, therefore, it is unlikely that high urinary levels come from a non-renal source. NAG is correlated with histological evidence of renal proximal tubule damage. Urinary NAG at ICU admission correlates well with results in critically ill patients. However, urinary NAG has been shown to be a sensitive marker of tubular injury, and its specificity can be reduced by a low threshold for the release of the tubular enzyme. False positive values have been reported during diabetes, rheumatoid arthritis and hyperthyroidism.33 The increase in urine is observed 12h after kidney damage.
In this regard, recently the Food and Drug Administration (FDA) approved the marketing of the Nephrocheck Test and Astute 140 Meter (Astute Medical Inc., San Diego, CA, USA), a rapid test that quantitatively measures TIMP-2 and IGFBP-734 cell cycle arrest biomarkers, which block the effect of cyclD-CDK4 and CyclE-CDK2 in cell cycle promotion, making them ideal G1 cell cycle arrest markers. The Astute 140 meter automatically multiplies the concentrations of the 2 biomarkers together and divides this product by 1000, reporting a single test result with units in (ng/ml)2/1000. In clinical practice, after AKI there is activation of cell division and cell proliferation in order to repopulate the denuded tubular epithelium. Early cell cycle arrest in G0/G1 could protect the kidney from further damage, by blocking DNA damage and caspase 3/7 activation, thereby limiting the extent of damage and avoiding a potentially dangerous maladaptive process caused by poor repair. These phenomena occur 24–48h before the increase in sCr which occurs after a significant drop in the GFR, and the early detection could undoubtedly facilitate new therapeutic and protective strategies, as suggested by the latest KDIGO guidelines.35
The combination of these 2 biomarkers seems to be highly predictive of patients who ultimately develop moderate to severe AKI within the next 12–24h. The performance of these markers compared to other markers has an area under curve (AUC) with a confidence index (CI) 95% greater than 0.8, especially when combined. The application of MAKE30 (which predicts adverse kidney events within 30 days, such as death, the need for RRT or duplication of sCr) increases substantially with the use of TIMP-2/IGFBP-7 when the values are >0.3 (ng/ml)2/1000 and were doubled with values >2.0(ng/ml)2/1000.36,37
The international multicenter Sapphire study, conducted in 728 critically ill patients, showed that the increase in performance by the combination of these biomarkers was AKI-specific (i.e., it is not caused by other comorbidities such as sepsis or CKD) and provided a strong signal as a “kidney alarm” to identify patients who are at imminent risk of developing AKI.36,37 These urinary biomarkers are believed to increase in response to the kidney tubule cell stress or early injury associated with factors known to cause AKI. The primary clinical cutoff value of >0.3(ng/ml)2/1000 for the combination of the 2 biomarkers derives from the Sapphire study data, which was verified in the Opal study, on a cohort of 153 critically ill patients.37
This cutoff point was selected for its high sensitivity (92%) to detect moderate to severe AKI within the next 12h; this is to identify patients at high risk of AKI in routine clinical practice so recommended treatment can be started early following the KDIGO guidelines for high-risk patients.20
A second cutoff point was sought with high specificity >2.0(ng/ml)2/1000 in order to identify the subset of patients who are at greater risk of AKI and, therefore, could benefit from more aggressive interventions. These cutoff points between 0.3 and 2.0 (ng/ml)2/1000 were subsequently validated in a study involving 23 sites and 408 critically ill patients in the United States (Topaz study) and whose primary objective was to identify patients with moderate to severe AKI.38
The most notable properties of these urinary biomarkers, and for which they were selected from more than 300 biomarkers, include: excellent performance, and having been validated, in heterogeneous populations with various pathologies (sepsis, shock, major surgery and trauma) in 3 different cohorts of critically ill patients.37 Also, recent publications highlight the ability of these biomarkers in the ICU environment to identify patients with AKI who are at increased risk of mortality or need for RRT in the next 9 months.39
The combination of two or more biomarkers an other clinical data is an attractive strategy to refine current diagnostic capacity. Despite great advances in this field, it should be noted that there are different methodological problems in AKI biomarker research; including the following:
- (1)
The performance of the new biomarkers was judged by comparison with sCr and oliguria, in the absence of a “gold standard” in current AKI diagnosis. These 2 markers, sCr and oliguria, as already mentioned, are affected by external factors.
- (2)
Studies vary in the cutoff points chosen to set thresholds for positive and negative predictive AKI-related events.
- (3)
There is uncertainty about the exact laboratory method, test platform and sampling conditions, and whether biomarker levels should be normalized in relation to urinary creatinine.
- (4)
Like creatinine, several novel AKI biomarkers are themselves not specific for kidney pathologies and can be influenced by common comorbid conditions, such as sepsis.
- (5)
Most of the biomarkers show a reflect a dynamic pattern of molecular and cellular events occurring during the clinical or subclinical AKI phase. It is therefore likely that a panel of different biomarkers and multiple measurements combined is better than a single test.
The general concept is that in AKI at least a part of the kidney tissue can be protected by early detection and intervention. This may be especially true in the early stages of the renal injury, which would prevent further damage and would preserve renal functional reserve.
According to KDIGO standards small increases in sCr are needed to reach AKI stage i and this reflect a profound and prolonged decrease in GFR. There is a need for AKI biomarkers that rapidly detect changes that allow the identification of subclinical kidney injuries.4
Early detection of such patients would allow a stratification by risk according to increased biomarkers or a combination of them that could ultimately help in validating preventive strategies and future therapies. Beyond early diagnosis and risk stratification, biomarkers have improved our understanding of the pathophysiological mechanisms associated with AKI. They have helped to challenge the up to now obsolete dichotomy between the so-called “prerenal AKI” and “renal AKI” (misnamed “acute tubular necrosis”).40 Patients formally identified as “prerenal AKI” or “transient AKI” and/or with “low urinary sodium excretion” also have evidence of tubular injury or damage, so that the current definition has become obsolete, especially at the subcellular and molecular level.40
This change in our global vision of the pathophysiological mechanism of AKI will without doubt in the near future help us in the therapeutic design of alternative strategies and in focusing on patients most likely to benefit from these strategies with an individualized approach driven by the biomarkers, a diagnosis and personalized treatment.40,41
The assessment of the kidney prognosis, and specifically kidney recovery after AKI, are issues that have recently arisen with direct implications for all health systems. The objective is prevention of progression to CKD, either by reducing the level of the injury or facilitating the healing and recovery of the damaged kidney and the control of risk factors. The injury, however, reduces the functioning renal mass and functional reserve. This process is key for the development of CKD.
The validation of biomarkers is insufficient to recommend its clinical use until now,13,16,17,42 and some results have even been disappointing.29,43
One of the reasons for these negative results may be based in erroneous population of patients studied. The type of ICU patients who may require the use of these biomarkers is still unknown. The population of interest could theoretically influence the diagnostic performance of the tests,44 as well as their clinical relevance.
Future efforts in this area should probably focus on evaluating the best current biomarkers in well-defined groups with high pretest likelihood of AKI and in contexts in which biomarkers can alter the clinical decision making.
Biomarkers: evidenceIt is important to identify patients at high risk of AKI, especially if this provides an opportunity to intervene. In the specific case of AKI, recommended preventive strategies currently include the optimization of the hemodynamic status, volume status and avoiding nephrotoxic drugs, contrast media and/or starches.4 It could easily be argued that this should be done in any critical patient, regardless of whether or not the patient is at high risk of developing AKI, and therefore the information that a biomarker can provide us may be redundant.
Ideally, the test to identify high risk patients should be as reliable as possible to avoid false negative and false positive results. Based on the experience from other clinical areas, the unselected use of biomarkers such as prostate specific antigen (PSA) or d-dimer in an unselected population produces a high number of false positive results and can potentially lead to harmful and unnecessary interventions.45 The performance of most biomarkers for AKI is better in selected groups of patients at high risk of AKI. For example, IGFBP7 and TIMP-2 that were evaluated and validated for use by the FDA in critically ill patients in ICU units.18,39
Can the new biomarkers diagnose acute kidney injury earlier than traditional tests?Several biomarkers have been shown to indicate the onset of AKI before increases of sCr occur (subclinical AKI). The results are more solid in pediatric cohorts without comorbidities suffering from a disease with a defined onset of AKI, such as for example in children after heart surgery.39 In more heterogeneous populations, where no occurrence of kidney injury is known (i.e., patients with septic shock), the performance of some biomarkers for early detection of AKI was equivalent to the clinical assessment and standard laboratory measurements and was not significantly better.29,30,40
It is generally assumed that an earlier diagnosis of AKI would translate directly into an earlier RRT and a better outcome for the patient. Unfortunately, there is currently no evidence that this actually happens, since there is no specific treatment for AKI.
Can the new acute kidney injury biomarkers identify patients who need kidney replacement therapy?Prediction of whether patients will need RRT is similar to predicting who will develop AKI. To date, there are no interventions beyond treatment of the underlying disease, attention to detail and good health care that prevent progression of AKI or induce recovery. Although it may seem appealing to know which patient will need RRT, it is very unlikely that this information will change clinical practice, especially since there is no evidence that early RRT is more beneficial.46 However, knowing whether a patient with RRT has recovered kidney function can be very useful.
Does the use of the new acute kidney injury biomarkers improve patient outcomes?The efficacy of a specific intervention based on increased biomarkers has not been demonstrated. The only intervention study using biomarkers to guide treatment was negative.41
Therefore, the claim that the use of biomarkers benefits patients and improves the outcome remains unproven.
When should acute kidney injury biomarkers be measured?It has been difficult to identify which patients would benefit more with the use of the biomarkers in situations in which the outcome already seems predictable based on standard parameters, such as clinical diagnosis and oliguria. Clearly in this situation, there is little additional benefit, just as with an application in patients with low risk of AKI, which leads to a useless use of the biomarkers, with an unnecessary increase in health care costs.
ConclusionIt has been suggested that AKI should be called “kidney attack”,46 in order to draw an analogy with a heart attack, and that a “kidney troponin” could be used to detect AKI early, as in acute myocardial infarction (AMI). Probably the analogy between the 2 conditions crumbles when potential interventions are considered. AMI has specific treatments. Yet even if a reliable biomarker showed AKI early, there would be no currently specific treatments that could be offered for the treatment of tubular ischemia or septic nephropathy.
While the results regarding the ability of biomarkers to improve the predictive value of AKI47–50 are contradictory and have not yet made the transition to clinical routine, recent publications of the Acute Dialysis Quality Initiative (ADQI)20 should be noted, which promote the search for early identification of damage or risk of AKI, especially in those patients in whom sCr is negative and biomarkers are positive; the promotion is based on the potential for the latter to transform the manner of diagnosis and treatment of AKI. However, these same recommendations, together with the KDIGO AKI guidelines,4 clearly state that so far sCr and diuresis represent the best markers with clinical applicability of AKI for use in diagnosis and monitoring. Thus we believe that the “belief polarization” must be oriented to the evidence, based on the development of new paradigms and use of new biomarkers in AKI.
Key phrases- •
Acute kidney injury (AKI) in critically ill patients is an independent risk factor that increases morbidity and mortality in the short and long term, with a tremendous financial impact in terms of health costs. AKI is also a first step toward chronic kidney disease (CKD).
- •
The diagnosis of AKI is based on indirect markers of kidney damage (sCr and diuresis), which are very insensitive and unspecific, to the detriment of the so-called “therapeutic window”.
- •
The new AKI biomarkers promise early identification of patients at risk of AKI, early diagnosis of AKI, indicating the need to initiate RRT and predicting the risk of progression to CKD.
- •
The ADQI proposes 4 possible combinations between functional and tissue damage biomarkers: no change in biomarkers, only changes in functional biomarkers, only changes in damage biomarkers, or changes in the 2 biomarkers, functional and damage.
- •
The FDA approved the use of TIMP-2 and IGFBP-7 cell cycle arrest biomarkers in a heterogeneous population such as the ICU population, which appear to be highly predictive of the development of moderate to severe AKI in the following 12–24h.
- •
Future efforts in this area should probably focus on evaluating the best current biomarkers in well-defined groups with high likelihood of AKI and in contexts in which biomarkers can alter clinical decision making.
The authors declare no conflict of interest.
Please cite this article as: Lombi F, Muryan A, Canzonieri R, Trimarchi H. Biomarcadores en la lesión renal aguda: ¿ paradigma o evidencia? Nefrología. 2016;36:339–346.


![Functional use of the new acute kidney injury biomarkers. From: Acute Dialysis Quality Initiative (ADQI). Available at: http://www.ADQI.org [accessed 10.01.13]. Functional use of the new acute kidney injury biomarkers. From: Acute Dialysis Quality Initiative (ADQI). Available at: http://www.ADQI.org [accessed 10.01.13].](https://static.elsevier.es/multimedia/20132514/0000003600000004/v1_201612030031/S2013251416300931/v1_201612030031/en/main.assets/thumbnail/gr1.jpeg?xkr=ue/ImdikoIMrsJoerZ+w94GCRvdQBB6xyQjMrWMzrts=)
![New criteria for the diagnosis of acute kidney injury. From: Acute Dialysis Quality Initiative (ADQI). Available at: http://www.ADQI.org [accessed 10.01.13]. New criteria for the diagnosis of acute kidney injury. From: Acute Dialysis Quality Initiative (ADQI). Available at: http://www.ADQI.org [accessed 10.01.13].](https://static.elsevier.es/multimedia/20132514/0000003600000004/v1_201612030031/S2013251416300931/v1_201612030031/en/main.assets/thumbnail/gr2.jpeg?xkr=ue/ImdikoIMrsJoerZ+w94GCRvdQBB6xyQjMrWMzrts=)